Advanced Simvastatin Manufacturing: High-Yield Synthesis via Optimized Hydrolysis and Acylation Catalysis
Advanced Simvastatin Manufacturing: High-Yield Synthesis via Optimized Hydrolysis and Acylation Catalysis
The global demand for effective lipid-lowering agents continues to drive innovation in the manufacturing of HMG-CoA reductase inhibitors, with simvastatin remaining a cornerstone therapeutic agent. Patent CN1283633C discloses a transformative process for the preparation of highly pure simvastatin that addresses critical bottlenecks in traditional synthetic routes. This technical breakthrough leverages a strategic combination of potassium hydroxide-mediated hydrolysis and quaternary salt-catalyzed acylation to achieve superior yields and purity profiles under significantly milder conditions. For pharmaceutical manufacturers and procurement leaders, this methodology represents a pivotal shift towards more sustainable and cost-effective production paradigms. By optimizing the conversion of lovastatin to simvastatin, the process eliminates the need for cryogenic temperatures and expensive reagents, thereby enhancing the overall economic feasibility of large-scale operations. The following analysis details the mechanistic advantages and commercial implications of adopting this refined synthetic strategy for reliable simvastatin supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of simvastatin from lovastatin has been plagued by severe operational constraints that hinder scalability and inflate production costs. Traditional methodologies, such as those disclosed in earlier patents, often rely on direct C-methylation of the side chain using metal amide bases under extremely low temperatures ranging from -75°C to -30°C. These cryogenic conditions necessitate specialized equipment and substantial energy consumption, creating a significant barrier to efficient commercial scale-up of complex pharmaceutical intermediates. Furthermore, alternative routes utilizing lithium hydroxide or potassium tert-butoxide for hydrolysis frequently require prolonged reaction times extending up to 56 hours, which drastically reduces throughput capacity. The reliance on anhydrous conditions and strong bases also promotes the formation of undesired by-products, complicating downstream purification and ultimately compromising the purity of the final active pharmaceutical ingredient. These inefficiencies collectively result in lower overall yields and higher manufacturing expenses, posing challenges for supply chain stability.
The Novel Approach
In stark contrast, the methodology outlined in patent CN1283633C introduces a robust and streamlined pathway that circumvents these historical limitations through intelligent reagent selection and catalytic enhancement. The process initiates with the treatment of lovastatin using potassium hydroxide dissolved in a water-methanol mixture, a modification that allows hydrolysis to proceed at moderate temperatures between 50°C and 70°C. This shift not only accelerates the reaction kinetics, reducing the hydrolysis timeframe to merely 5-12 hours, but also simplifies the operational requirements by eliminating the need for rigorous anhydrous environments. Subsequent steps involve relactonization and protection of the lactone ring hydroxyl group, followed by a highly efficient acylation step. As illustrated in the reaction scheme below, the integration of quaternary ammonium or phosphonium halide catalysts during acylation facilitates rapid side-chain installation, completing the transformation in just 6-8 hours compared to the 3-4 days required by conventional methods.
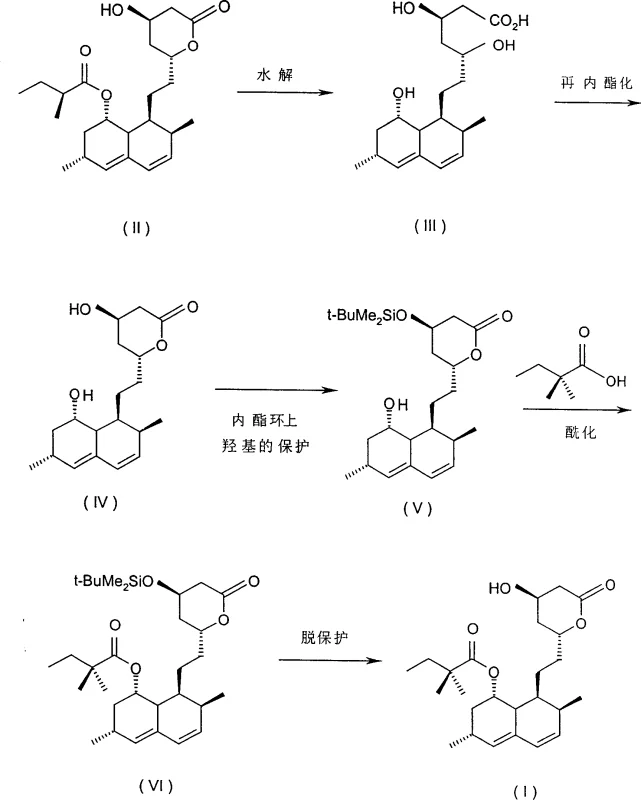
Mechanistic Insights into Quaternary Salt-Catalyzed Acylation
The core innovation of this synthetic route lies in the mechanistic efficiency of the acylation step, which is driven by the presence of phase-transfer catalysts such as quaternary ammonium halides or quaternary phosphonium halides. In traditional acylation reactions without such catalysts, the nucleophilic attack of the hydroxyl group on the acylating agent is often sluggish, requiring excessive heat or prolonged durations that degrade product quality. However, the introduction of catalysts like benzyltri-n-butylammonium bromide or tetra-n-butylphosphonium bromide creates a microenvironment that enhances the nucleophilicity of the substrate. These catalysts function by solubilizing the reactive species in the organic phase, effectively lowering the activation energy barrier for the acylation reaction. This catalytic acceleration ensures that the reaction reaches completion within a fraction of the time typically observed, while simultaneously suppressing side reactions that lead to impurity formation. The result is a cleaner reaction profile that yields the acylated intermediate with purity levels exceeding 98%, providing a solid foundation for the final deprotection step.
Furthermore, the hydrolysis mechanism employing potassium hydroxide in a aqueous-methanolic system offers distinct advantages in terms of impurity control and stereochemical integrity. The use of KOH, a cheaper and more accessible base compared to lithium hydroxide or t-BuOK, ensures that the opening of the lactone ring and the removal of the 2-methylbutyryl side chain occur with high selectivity. The specific solvent ratio of water to methanol is critical in maintaining the solubility of the intermediate triol acid while preventing degradation. By carefully controlling the pH during the subsequent acidification and precipitation stages, the process minimizes the co-precipitation of inorganic salts and organic by-products. This rigorous control over the reaction environment is essential for achieving the high-purity simvastatin required for regulatory compliance, demonstrating how fundamental chemical engineering principles can be leveraged to optimize pharmaceutical manufacturing processes.
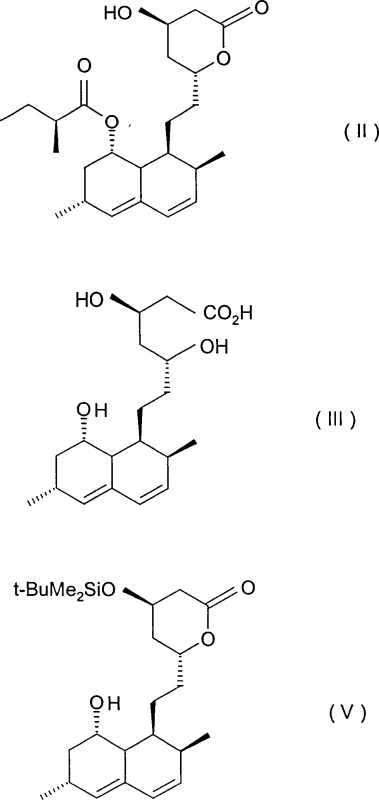
How to Synthesize Simvastatin Efficiently
The implementation of this optimized synthetic route requires precise adherence to reaction parameters to maximize yield and purity. The process begins with the hydrolysis of lovastatin using potassium hydroxide in a water-methanol mixture, followed by acidification to isolate the triol acid intermediate. This intermediate is then subjected to relactonization and silyl protection to generate the protected lactone substrate. The critical acylation step utilizes 2,2-dimethylbutyryl chloride in the presence of a quaternary salt catalyst under reflux conditions, followed by a final deprotection step to yield the target molecule. Detailed standard operating procedures regarding stoichiometry, temperature gradients, and workup protocols are essential for reproducibility. For a comprehensive guide on executing these steps with precision, please refer to the standardized synthesis instructions provided below.
- Hydrolyze lovastatin using potassium hydroxide in a water-methanol mixture at moderate temperatures (50-70°C) to form the triol acid intermediate efficiently.
- Perform relactonization followed by selective protection of the lactone ring hydroxyl group using silyl protecting agents to prepare the substrate for acylation.
- Execute the critical acylation step using 2,2-dimethylbutyryl chloride in the presence of quaternary ammonium or phosphonium halide catalysts to install the side chain rapidly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the process described in patent CN1283633C offers tangible strategic benefits that extend beyond mere technical superiority. The transition from cryogenic, long-duration reactions to mild, catalytic processes fundamentally reshapes the cost structure and reliability of the supply chain. By eliminating the dependency on expensive reagents like potassium tert-butoxide and reducing the energy load associated with maintaining sub-zero temperatures, manufacturers can achieve substantial cost savings in raw material procurement and utility consumption. Moreover, the drastic reduction in reaction times—from days to hours—significantly increases asset utilization rates, allowing production facilities to generate more batches within the same timeframe. This enhanced throughput capability directly translates to improved supply continuity and the ability to respond more agilely to market fluctuations in demand for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of costly lithium hydroxide and t-BuOK with economical potassium hydroxide, combined with the use of efficient quaternary salt catalysts, drastically lowers the bill of materials. Additionally, the elimination of energy-intensive cryogenic cooling systems and the reduction in heating duration for acylation contribute to significant operational expenditure reductions. These factors collectively enhance the profit margin potential for generic drug manufacturers seeking cost reduction in statin manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the KOH hydrolysis method and the rapid acylation kinetics ensure consistent batch-to-batch performance, minimizing the risk of production delays caused by reaction failures or extended processing times. The use of readily available reagents reduces the vulnerability of the supply chain to shortages of specialized chemicals. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream formulation partners receive their materials on schedule.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures facilitate easier scale-up from pilot plants to multi-ton commercial production lines. The process generates fewer hazardous by-products and avoids the use of volatile or toxic solvents associated with older methods, aligning with increasingly stringent environmental regulations. This environmental compatibility not only reduces waste disposal costs but also strengthens the sustainability profile of the manufacturing operation, a key metric for modern corporate responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced simvastatin synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical application of the technology. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: How does the new catalytic method improve upon traditional simvastatin synthesis?
A: The novel method replaces cryogenic conditions and expensive bases like t-BuOK with mild KOH hydrolysis and quaternary salt catalysts, reducing reaction times from days to hours while significantly improving yield and purity profiles.
Q: What specific catalysts are utilized in the acylation step of this process?
A: The process employs quaternary ammonium halides such as benzyltri-n-butylammonium bromide or quaternary phosphonium halides like tetra-n-butylphosphonium bromide to accelerate acylation under reflux conditions.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the elimination of extreme low-temperature requirements and the use of robust, inexpensive reagents like potassium hydroxide make this route highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the KOH-mediated hydrolysis and quaternary salt-catalyzed acylation are seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of simvastatin adheres to the highest international standards. Our infrastructure is designed to support the complex chemistry required for HMG-CoA reductase inhibitors, providing a secure and reliable source for your API needs.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall production costs. Let us be your partner in delivering high-quality, cost-effective pharmaceutical solutions to the global market.
