Advanced Manufacturing of Neratinib Intermediates via Green Reductive Amination
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the synthesis of complex oncology drugs like Neratinib. Patent CN103265530A discloses a significant advancement in this domain, presenting a preparation method that diverges from traditional, hazard-heavy synthetic routes. This innovation focuses on a convergent strategy involving the condensation and subsequent reduction of two key precursors: 6-[(E)-4-(dimethylamino)-2-butenamide]-7-ethoxy-4-amino-3-quinolinecarbonitrile and 3-chloro-4-[(pyridine-2-yl)methoxy]-benzaldehyde. By shifting the synthetic logic towards reductive amination, this method addresses critical pain points regarding toxicity and process complexity that have long plagued the manufacturing of ErbB receptor tyrosine kinase inhibitors.
For R&D directors and process chemists, the implications of this patent are profound. It offers a tangible solution to the challenges of impurity control and waste management associated with legacy processes. The ability to construct the final molecule through a mild coupling reaction rather than harsh substitution opens new avenues for optimizing yield and purity profiles, which are paramount for regulatory approval and commercial success in the competitive landscape of breast cancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Neratinib and its analogues has relied heavily on classical heterocyclic chemistry that, while robust, carries significant environmental and operational burdens. As detailed in prior art such as WO2004/032909 and related documents, the conventional pathway typically involves the formation of a 4-oxo-quinoline derivative followed by a chlorination step to activate the 4-position for nucleophilic substitution. This chlorination step notoriously requires the use of phosphorus oxychloride (POCl3) or similar aggressive chlorinating agents like phosphorus trichloride.
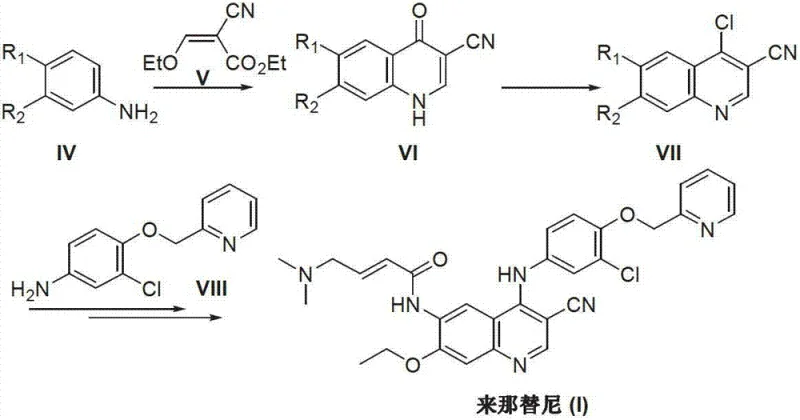
The reliance on POCl3 presents a multifaceted challenge for modern manufacturing. Firstly, it generates substantial amounts of acidic and phosphorous-containing waste, necessitating expensive and complex effluent treatment protocols to meet increasingly stringent environmental regulations. Secondly, the reaction conditions often demand high-temperature reflux for extended periods to drive the cyclization and chlorination to completion, which increases energy consumption and poses safety risks on a large scale. Furthermore, the handling of such corrosive reagents requires specialized equipment and rigorous safety measures, adding to the overall capital and operational expenditure of the production facility.
The Novel Approach
In stark contrast, the method disclosed in CN103265530A introduces a streamlined and greener alternative that bypasses these hazardous steps entirely. The core innovation lies in the direct coupling of a pre-functionalized amino-quinoline intermediate with a substituted benzaldehyde. Instead of activating the quinoline ring with chlorine, the process utilizes the inherent nucleophilicity of the 4-amino group on the quinoline scaffold to attack the electrophilic carbonyl carbon of the aldehyde. This condensation forms an imine intermediate, which is subsequently reduced to the stable secondary amine linkage found in the final Neratinib structure.
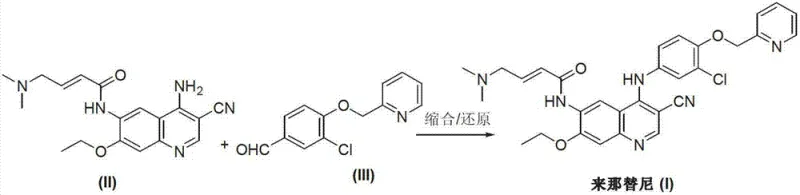
This reductive amination strategy offers several distinct advantages. It operates under significantly milder conditions, typically utilizing solvents like methanol and bases such as triethylamine at moderate temperatures around 50-55°C. The elimination of the chlorination step not only removes the need for toxic reagents but also simplifies the purification process, as there are no chlorinated by-products to separate. This results in a cleaner reaction profile, higher atom economy, and a process that is inherently safer and more sustainable for industrial application.
Mechanistic Insights into Reductive Amination and Cyclization
To fully appreciate the technical merit of this synthesis, one must examine the mechanistic details of the key transformations. The formation of the crucial intermediate (II), 6-[(E)-4-(dimethylamino)-2-butenamide]-7-ethoxy-4-amino-3-quinolinecarbonitrile, involves a sophisticated cyclization cascade. This step typically employs a Lewis acid catalyst, such as aluminum chloride (AlCl3), to facilitate the condensation of an aniline derivative with triethyl orthoformate and malononitrile. The Lewis acid activates the orthoformate, promoting the formation of an imine ether species that undergoes intramolecular electrophilic aromatic substitution to close the quinoline ring. The precise control of temperature, ideally between 120-140°C, is critical here to ensure complete cyclization without degrading the sensitive amide side chain.
Following the isolation of intermediate (II), the final assembly of Neratinib proceeds via a classic reductive amination mechanism. In the presence of a base like triethylamine, the primary amine of the quinoline attacks the aldehyde group of the pyridine-methoxy-benzaldehyde component. This reversible reaction forms a Schiff base (imine). The subsequent addition of a reducing agent, such as sodium borohydride (NaBH4) or via catalytic hydrogenation using Pd/CaCO3, irreversibly reduces the C=N double bond to a C-N single bond. This mechanism is highly selective; under the optimized conditions described (e.g., 0-5°C for NaBH4 addition), the reduction targets the imine exclusively, leaving the nitrile group and the conjugated alkene in the side chain intact, thereby ensuring high chemical purity and minimizing the formation of over-reduced impurities.
How to Synthesize Neratinib Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize yield and quality. The process begins with the preparation of the amino-quinoline core, followed by the convergent coupling step. Operators must maintain strict control over the addition rates of reagents, particularly during the exothermic reduction phase, to prevent thermal runaways. Detailed standard operating procedures regarding solvent quality, catalyst loading, and work-up protocols are essential for reproducibility. For a comprehensive guide on the specific experimental parameters and step-by-step instructions derived from the patent examples, please refer to the standardized synthesis protocol below.
- Prepare the key intermediate 6-[(E)-4-(dimethylamino)-2-butenamide]-7-ethoxy-4-amino-3-quinolinecarbonitrile via cyclization using aluminum chloride.
- Perform condensation between the quinoline amine intermediate and 3-chloro-4-[(pyridine-2-yl)methoxy]-benzaldehyde in the presence of a base like triethylamine.
- Execute the reduction step using sodium borohydride or catalytic hydrogenation to yield the final Neratinib API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible business value and risk mitigation. The shift away from hazardous reagents like phosphorus oxychloride fundamentally alters the cost structure of manufacturing. By eliminating the need for specialized corrosion-resistant reactors and complex waste neutralization systems, the capital expenditure required for setting up production lines is significantly reduced. Moreover, the simplified downstream processing—owing to fewer by-products and cleaner reaction profiles—lowers the operational costs associated with purification and solvent recovery, leading to a more economically efficient production model overall.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the removal of expensive and hazardous reagents. Traditional chlorination methods incur high costs not just for the reagents themselves, but for the disposal of the resulting toxic waste streams. By replacing this with a reductive amination using common bases and reducing agents, the direct material costs are lowered. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, further contributing to substantial cost savings in utility expenses without compromising the quality of the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials. The key building blocks, such as the substituted anilines and benzaldehydes, are commodity chemicals with robust global supply networks, unlike some specialized chlorinating intermediates that may face supply bottlenecks. The simplicity of the process also means that manufacturing can be easily transferred between different facilities or scaled up rapidly to meet market demand, ensuring a continuous and reliable supply of this critical oncology ingredient to downstream drug product manufacturers.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this green chemistry approach positions the manufacturer favorably. The reduction in hazardous waste generation aligns with global trends towards stricter environmental compliance, reducing the risk of regulatory fines or shutdowns. The process is inherently scalable; the exothermic nature of the reduction can be managed effectively in large reactors through controlled dosing, allowing for seamless transition from pilot plant batches to multi-ton commercial production runs while maintaining consistent product quality and safety standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the feasibility, safety, and quality aspects of this patented method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a clear picture of what partners can expect when adopting this technology for their supply chains.
Q: How does this new Neratinib synthesis method improve environmental safety?
A: The novel route eliminates the use of phosphorus oxychloride (POCl3) and other harsh chlorinating agents required in traditional methods, significantly reducing toxic waste generation and improving operator safety.
Q: What are the critical reaction conditions for the final coupling step?
A: The condensation typically occurs at mild temperatures (50-55°C) using solvents like methanol and bases such as triethylamine, followed by reduction with sodium borohydride at room temperature.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials, avoids hazardous reagents, and operates under mild conditions, making it highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Neratinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic methodologies like the one described in CN103265530A to stay ahead in the competitive pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We are committed to delivering high-purity Neratinib intermediates and APIs that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch-to-batch consistency.
We invite potential partners to engage with us to explore how this greener, more efficient synthesis route can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term goals for delivering life-saving breast cancer treatments to patients worldwide.
