Advanced Manufacturing of Olanzapine Intermediates for Commercial Scale-Up
Advanced Manufacturing of Olanzapine Intermediates for Commercial Scale-Up
The pharmaceutical landscape for antipsychotic medications continues to evolve, with Olanzapine remaining a cornerstone therapy for schizophrenia and bipolar disorder. As detailed in patent CN102942573A, significant advancements have been made in the synthetic methodology for producing this critical active pharmaceutical ingredient (API). The disclosed technology addresses long-standing challenges in impurity profiling and yield optimization, offering a robust pathway for industrial application. By integrating specific water-absorbing agents and precisely tuning reagent concentrations, this method effectively mitigates the formation of stubborn byproducts that have historically plagued manufacturers. For R&D directors and procurement specialists alike, understanding these mechanistic improvements is vital for securing a reliable supply chain of high-purity olanzapine intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Olanzapine has relied on routes that either utilize expensive N-methylpiperazine directly or employ a two-step process involving piperazine followed by methylation. While the latter approach offers cost benefits by using cheaper starting materials, it introduces significant technical hurdles regarding impurity control. Specifically, the presence of moisture during the condensation step can lead to the formation of hydrolytic impurities, often identified as Formula VI in related literature. These impurities are notoriously difficult to remove in downstream processing, thereby compromising the final API quality. Furthermore, the subsequent methylation step often suffers from lack of selectivity, leading to over-methylation or acylation on the secondary nitrogen atom, generating impurities such as Formula VII. These structural defects not only reduce the overall yield but also necessitate complex and costly purification protocols to meet regulatory standards.
The Novel Approach
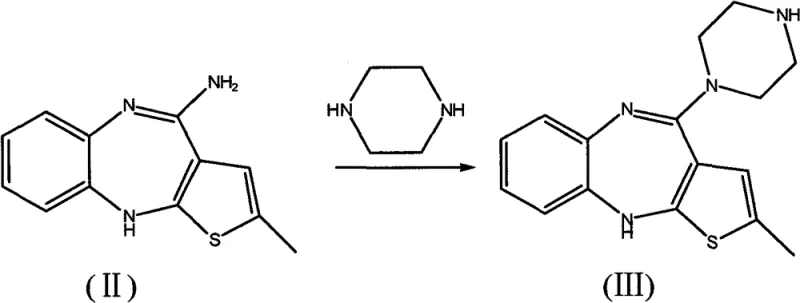
The innovative methodology presented in the patent data fundamentally alters the reaction environment to favor the desired product while suppressing side reactions. The core of this advancement lies in the strategic addition of water-absorbing agents, such as calcium oxide, during the initial reaction between the thienobenzodiazepine precursor and piperazine. This simple yet effective modification creates an anhydrous environment that drives the equilibrium towards the formation of the desmethyl intermediate (Formula III) while preventing hydrolysis. Additionally, the process optimizes the methylation conditions by utilizing a specific concentration of formic acid (80% to 90%) and formaldehyde (20% to 25%). This precise control ensures selective mono-methylation, effectively blocking the formation of di-methylated or acylated byproducts. The result is a streamlined process that delivers superior purity and yield without the need for exotic catalysts.
Mechanistic Insights into Water-Scavenged Cyclization and Selective Methylation
To fully appreciate the technical value of this synthesis, one must examine the mechanistic role of the additives. In the first stage, the reaction between 2-methyl-4-amino-thieno-benzodiazepine hydrochloride and piperazine is a nucleophilic substitution that releases water or is sensitive to ambient moisture. The introduction of calcium oxide acts as a chemical sink, irreversibly binding water molecules. This shifts the reaction equilibrium according to Le Chatelier's principle, ensuring near-quantitative conversion to the desmethyl intermediate. From a kinetic perspective, the absence of water prevents the competitive hydrolysis of the nitrile or amine functionalities, which are common degradation pathways in polar aprotic solvents like DMSO. This mechanistic intervention is critical for maintaining the integrity of the heterocyclic core throughout the synthesis.
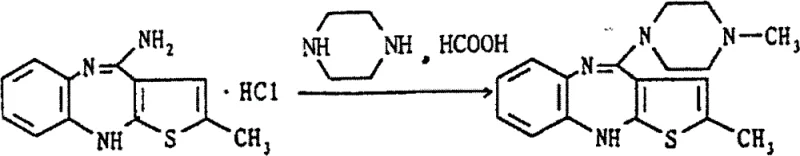
In the second stage, the reductive alkylation requires a delicate balance of acidity and reducing power. The use of 85% formic acid serves a dual purpose: it provides the protons necessary to activate the iminium intermediate formed with formaldehyde, and its specific water content modulates the reactivity to prevent over-alkylation. If the acid concentration is too high or too low, the selectivity for the tertiary amine formation drops, leading to the aforementioned Formula VII impurities. The patent data highlights that using 23% formaldehyde in conjunction with this specific acid concentration creates an optimal kinetic window. This ensures that the methylation occurs exclusively at the piperazine nitrogen intended for substitution, leaving the other secondary nitrogen untouched. This level of control is essential for minimizing the impurity burden before the final crystallization step.
How to Synthesize High-Purity Olanzapine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, solvent ratios, and crystallization triggers. The process begins with the reflux of the starting materials in a toluene and DMSO mixture, followed by a controlled quench into a saturated sodium bicarbonate solution. A critical innovation in this workflow is the addition of methanol during the crystallization phase, which significantly enhances the precipitation of the intermediate, improving both recovery and physical form. The subsequent methylation is conducted at a moderate temperature of 80°C to balance reaction rate with selectivity. For a comprehensive breakdown of the operational parameters and safety considerations, please refer to the standardized guide below.
- Condense 2-methyl-4-amino-thieno-benzodiazepine hydrochloride with anhydrous piperazine in toluene/DMSO using calcium oxide as a water absorbent under reflux.
- Perform reductive methylation on the resulting desmethyl intermediate using 23% formaldehyde and 85% formic acid at 80°C to prevent over-methylation.
- Purify the crude product via recrystallization in ethanol with activated carbon treatment to achieve pharmaceutical grade specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthesis route offers tangible benefits beyond mere technical elegance. The primary advantage lies in the substantial cost reduction achieved by substituting expensive N-methylpiperazine with commodity-grade piperazine. This raw material swap drastically lowers the Bill of Materials (BOM) cost without sacrificing quality. Furthermore, the enhanced selectivity of the reaction means that less material is lost to byproduct formation, effectively increasing the throughput of existing manufacturing assets. This efficiency gain translates directly into improved margin potential for the final API product.
- Cost Reduction in Manufacturing: The elimination of expensive methylating agents in the first step and the use of cost-effective formic acid for the second step create a highly economical process. By avoiding the formation of difficult-to-remove impurities, the need for extensive chromatographic purification or multiple recrystallizations is minimized. This reduction in downstream processing time and solvent consumption leads to significant operational expenditure savings, making the final product more competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as piperazine, calcium oxide, and formic acid ensures a stable supply chain不受 geopolitical or niche market fluctuations. Unlike processes dependent on specialized catalysts or rare reagents, this method utilizes commodities that can be sourced from multiple vendors globally. This diversification of supply risk guarantees continuity of production, a critical factor for maintaining long-term contracts with major pharmaceutical companies.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like reflux, filtration, and crystallization that are easily transferred from pilot plant to commercial scale. The avoidance of heavy metal catalysts simplifies waste stream management and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with hazardous waste disposal and treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications for industrial adoption.
Q: How does this method control impurities compared to traditional routes?
A: By introducing a water-absorbing agent like calcium oxide during the initial condensation, the formation of hydrolytic impurities (Formula VI) is significantly suppressed. Furthermore, optimizing the formic acid concentration to 80-90% prevents unwanted acylation or over-methylation on the secondary nitrogen.
Q: What are the key advantages for large-scale production?
A: The process utilizes cost-effective piperazine instead of expensive N-methylpiperazine. The specific crystallization protocol involving methanol addition ensures high recovery rates and consistent particle size, facilitating easier filtration and drying on a multi-ton scale.
Q: Is the synthesis suitable for GMP manufacturing?
A: Yes, the method avoids toxic heavy metal catalysts and uses standard organic solvents like toluene and DMSO which are easily removed. The final purification step using activated carbon ensures the removal of colored bodies and trace organic impurities, meeting stringent pharmacopoeia standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olanzapine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against global pharmacopoeia standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can drive value for your organization.
