Advanced Electroreduction Technology for High-Purity Olanzapine Intermediate Manufacturing
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable methods for producing critical active pharmaceutical ingredient (API) precursors. Patent CN110656347B introduces a groundbreaking electroreduction preparation method for Olanzapine intermediates, specifically targeting the synthesis of 4-amino-2-methyl-10H-thieno[2,3-b][1,5]benzodiazepine hydrochloride. This technology represents a significant paradigm shift from traditional chemical reduction techniques, utilizing electrons as clean reagents within a diaphragm electrolytic cell to achieve high conversion rates and exceptional selectivity. By operating under mild conditions with precise control over electrode potential, this process effectively mitigates the risks associated with toxic reducing agents and heavy metal contamination, offering a compelling solution for reliable pharmaceutical intermediate supplier networks aiming to enhance their green manufacturing credentials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of nitro groups in complex heterocyclic systems has relied heavily on stoichiometric chemical reductants or catalytic hydrogenation, both of which present substantial operational and environmental challenges. Traditional methods utilizing stannous chloride often result in expensive reagent costs and the formation of tenacious tin-complexes that are notoriously difficult to separate, severely compromising the purity of the final intermediate. Similarly, iron powder reduction generates massive quantities of iron sludge, creating a significant environmental burden and risking the adsorption of valuable aniline compounds, while hydrazine hydrate poses severe genotoxicity and safety hazards. Furthermore, catalytic hydrogenation using Raney Nickel or Palladium on Carbon introduces the risk of heavy metal residues exceeding strict regulatory limits, necessitating costly and complex purification steps to ensure patient safety.
The Novel Approach
In stark contrast, the electroreduction methodology described in the patent offers a streamlined, catalyst-free alternative that fundamentally alters the reaction landscape. By employing a diaphragm electrolytic cell with specific cathode and anode configurations, the process utilizes electrical energy to drive the reduction of the nitro group directly, eliminating the need for external chemical reducing agents entirely. This approach not only simplifies the downstream processing by avoiding metal contamination but also allows for precise tuning of the reaction selectivity through voltage control, ensuring high yields of the desired amino-intermediate. The ability to telescope the electroreduction directly into the subsequent cyclization step further enhances efficiency, as demonstrated by the overall reaction pathway which minimizes unit operations and solvent usage.
Mechanistic Insights into Electrochemical Nitro Reduction
The core of this innovation lies in the controlled electron transfer at the cathode surface, where the nitro group of 2-(2-nitroanilino)-5-methylthiophene-3-carbonitrile undergoes a multi-step reduction sequence. Under acidic conditions facilitated by the inorganic ammonium salt electrolyte, the nitro group accepts electrons and protons sequentially, passing through nitroso and hydroxylamine intermediates before finally forming the stable amine functionality. This mechanistic pathway is highly dependent on the electrode potential, which is maintained between 1.20V and 2.50V relative to a reference electrode to prevent over-reduction or side reactions. The use of a cation exchange membrane effectively separates the anodic and cathodic compartments, preventing the oxidation of the sensitive amino product at the anode and ensuring that the protons generated at the anode can migrate to support the cathodic reduction process.
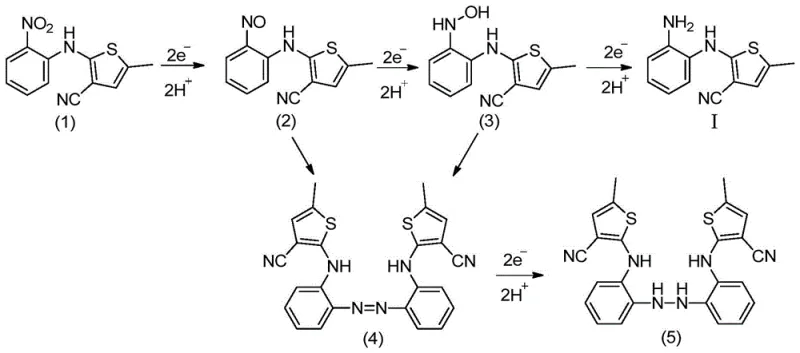
Impurity control is inherently built into this electrochemical system through the precise regulation of current density and temperature. Unlike chemical reductions where local concentration gradients of reducing agents can lead to hotspots and byproduct formation, the uniform distribution of current across the electrode surface promotes homogeneous reaction kinetics. The patent highlights that by optimizing parameters such as current density (25.0 mA/cm² to 250.0 mA/cm²) and temperature (25°C to 80°C), the formation of azo or hydrazo coupling byproducts—common pitfalls in nitro reductions—is significantly suppressed. This results in a crude product profile that is exceptionally clean, reducing the burden on crystallization and purification stages and ultimately delivering a high-purity intermediate suitable for direct progression to the final API synthesis.
How to Synthesize 4-Amino-2-Methyl-10H-Thieno-Benzodiazepine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology at scale, emphasizing the preparation of a biphasic or mixed catholyte system to ensure solubility and conductivity. The process begins with the dissolution of the nitro-precursor in an organic solvent such as ethyl acetate or propanol, which is then mixed with an aqueous solution of an inorganic ammonium salt to form the catholyte. Following the electrolysis, the reaction mixture is treated directly with concentrated hydrochloric acid and refluxed, facilitating the intramolecular cyclization to form the benzodiazepine ring system without the need to isolate the unstable amino-intermediate. This telescoped approach not only saves time but also minimizes material loss, as illustrated in the comprehensive process flow below.
- Prepare the catholyte by dissolving 2-(2-nitroanilino)-5-methylthiophene-3-carbonitrile in an organic solvent and mixing with an aqueous inorganic ammonium salt solution.
- Perform electrolysis in a diaphragm cell using a DSA anode and metal cathode at a current density of 25.0-250.0 mA/cm² and voltage of 1.20-2.50V.
- Add concentrated hydrochloric acid to the cathode product and reflux for 3.0-5.0 hours to cyclize and obtain the final benzodiazepine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electroreduction technology translates into tangible strategic benefits regarding cost stability and supply continuity. By removing the dependency on volatile commodity chemicals like hydrazine and expensive precious metal catalysts, manufacturers can insulate their production costs from market fluctuations and supply disruptions. The elimination of heavy metal catalysts also removes the need for specialized scavenging resins and extensive testing for residual metals, streamlining the quality control workflow and accelerating the release of batches for downstream processing. Furthermore, the simplified waste profile, devoid of heavy metal sludge and toxic organic byproducts, significantly reduces the complexity and cost of environmental compliance and waste disposal.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of stoichiometric chemical reductants with electricity, which is generally more cost-effective and easier to source reliably than specialized reducing agents. Additionally, the removal of palladium or nickel catalysts eliminates a major capital expense and the recurring cost of catalyst replacement or regeneration. The high selectivity of the electrochemical method leads to improved yields, meaning less raw material is wasted on side products, directly enhancing the overall material efficiency and reducing the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Relying on electricity and common inorganic salts like ammonium chloride creates a much more resilient supply chain compared to one dependent on specialized organic reductants which may have limited suppliers. The robustness of the electrochemical cell design allows for continuous operation and easier scaling, ensuring that production volumes can be ramped up quickly to meet surging demand without the lead times associated with sourcing bulk quantities of hazardous chemicals. This reliability is crucial for maintaining uninterrupted supply to API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable through the addition of electrode surface area or the use of filter-press type cells, making the transition from pilot to commercial scale straightforward and predictable. From an environmental perspective, the 'green' nature of using electrons as reagents aligns perfectly with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The absence of toxic sludge and the potential for solvent recycling within the closed-loop electrolytic system position this method as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this electroreduction technology, derived directly from the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the feasibility of adopting this method for large-scale production and to highlight the specific advantages over legacy synthetic routes.
Q: How does electroreduction improve the purity of Olanzapine intermediates compared to catalytic hydrogenation?
A: Electroreduction eliminates the use of transition metal catalysts like Palladium or Nickel, thereby preventing the formation of difficult-to-remove metal complexes and ensuring the final product meets stringent heavy metal specifications required for API manufacturing.
Q: What are the environmental advantages of this electrochemical method?
A: This method replaces hazardous chemical reducing agents such as hydrazine hydrate, iron powder, and stannous chloride, significantly reducing toxic waste generation and aligning with green chemistry principles for sustainable pharmaceutical production.
Q: Can the electroreduction and cyclization steps be combined?
A: Yes, the patent describes a telescoped process where the electroreduction product containing the amino-intermediate can be directly subjected to acid reflux cyclization without isolation, streamlining the workflow and improving overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olanzapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110656347B are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of Olanzapine intermediate meets the highest international standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your transition to greener, more efficient manufacturing is seamless and commercially viable.
