Revolutionizing Enol Silyl Ether Production: A Room-Temperature Phase Transfer Strategy for Industrial Scale-Up
Introduction to Advanced Enol Silyl Ether Synthesis
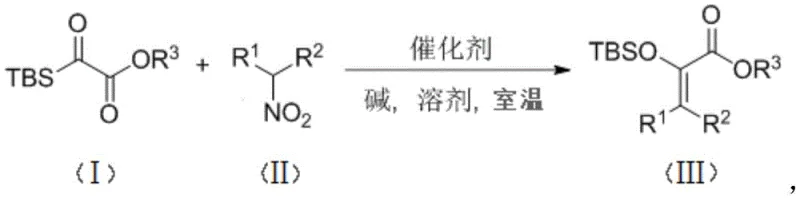
The synthesis of enol silyl ethers represents a cornerstone capability in modern organic chemistry, particularly for the construction of complex pharmaceutical architectures and natural products. These compounds serve as vital surrogates for unstable enol anions, effectively enhancing reaction yields and suppressing undesirable side pathways during carbonyl functionalization. However, traditional methodologies have long been plagued by operational complexities, often necessitating stringent anhydrous conditions and cryogenic temperatures that hinder efficient manufacturing. A groundbreaking advancement in this field is detailed in patent CN113387976A, which discloses a highly efficient method for synthesizing ester-containing enol silyl ether compounds. This innovation leverages a phase transfer catalytic system to drive a tandem nucleophilic addition-Brook rearrangement-elimination sequence at ambient temperature. By utilizing inexpensive nitroalkanes as nucleophiles and silyl acylates as substrates, this technology offers a transformative route for reliable pharmaceutical intermediates supplier networks seeking to optimize their synthetic portfolios.
The significance of this patent lies not merely in the chemical transformation itself, but in its profound implications for process chemistry and industrial scalability. Conventional routes often rely on the capture of enolate anions generated under basic conditions using silicon-halogen bonds, a process that can be capricious and sensitive to substrate electronics. In contrast, the disclosed method employs tetrabutylammonium chloride as a catalyst alongside cesium carbonate as a base, creating a robust reaction environment. This shift allows for the direct synthesis of valuable intermediates with high atom economy, addressing critical pain points related to waste generation and energy consumption. For R&D directors and process engineers, this represents a pivotal opportunity to streamline workflows, reduce reliance on hazardous reagents, and achieve consistent quality in the production of high-purity OLED material precursors or active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of enol silyl ethers, especially those containing ester functionalities, has been fraught with significant technical hurdles that impact both cost and throughput. Traditional protocols typically involve the deprotonation of carbonyl compounds containing alpha-active protons using sterically hindered, non-nucleophilic strong bases such as lithium diisopropylamide (LDA). These reactions invariably require cryogenic conditions, often ranging from -70°C to -20°C, to prevent uncontrolled side reactions and ensure regioselectivity. Such extreme thermal requirements impose a heavy burden on manufacturing infrastructure, necessitating specialized cooling equipment and substantial energy inputs. Furthermore, the subsequent trapping of the generated enolate with chlorosilanes introduces additional complexity, as these reagents are moisture-sensitive and can generate stoichiometric amounts of salt waste. The combination of harsh conditions, expensive reagents, and difficult workup procedures renders many conventional methods economically unviable for large-scale commercial application.
The Novel Approach
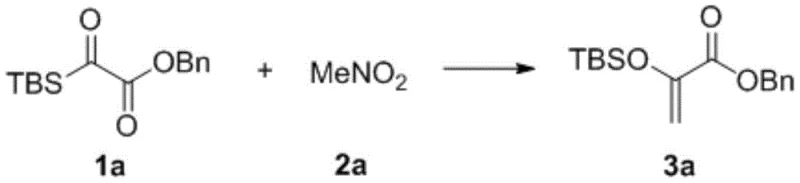
The methodology described in patent CN113387976A fundamentally disrupts this status quo by introducing a mild, phase-transfer catalyzed pathway that operates efficiently at room temperature. Instead of relying on pre-formed enolates and cryogenic cooling, this novel approach utilizes silyl acyl compounds as substrates and nitroalkanes as nucleophiles. The reaction proceeds through a sophisticated cascade mechanism involving nucleophilic addition, followed by a Brook rearrangement and final elimination, all facilitated by a quaternary ammonium salt catalyst. This strategic redesign of the synthetic route eliminates the need for extreme temperatures and aggressive bases, thereby drastically simplifying the operational protocol. As demonstrated in specific embodiments, such as the synthesis of compound 3a shown below, yields can reach impressive levels, validating the efficacy of this greener alternative.
Mechanistic Insights into Phase Transfer Catalyzed Brook Rearrangement
To fully appreciate the technical superiority of this synthesis, one must delve into the intricate mechanistic details that govern the transformation. The reaction initiates with the activation of the nitroalkane nucleophile by the base, cesium carbonate, within the organic phase, a process significantly accelerated by the phase transfer catalyst, tetrabutylammonium chloride. The resulting nitronate anion attacks the electrophilic carbonyl carbon of the silyl acylate substrate, forming a tetrahedral intermediate. This is the critical juncture where the unique chemistry of the silicon atom comes into play. The proximity of the silyl group to the newly formed alkoxide triggers a rapid 1,4-Brook rearrangement, wherein the silyl group migrates from the carbon atom to the oxygen atom. This migration is thermodynamically driven by the formation of a strong silicon-oxygen bond, effectively stabilizing the intermediate and setting the stage for the subsequent elimination step.
Following the rearrangement, the system undergoes an elimination of the nitro group, restoring the double bond character and yielding the final enol silyl ether product. This tandem sequence is remarkably efficient because each step is coupled to the next, preventing the accumulation of high-energy intermediates that could lead to decomposition or polymerization. From an impurity control perspective, the mildness of the reaction conditions is paramount. By avoiding strong, non-selective bases and high thermal stress, the formation of byproducts such as self-condensation polymers or hydrolysis products is minimized. This inherent selectivity ensures that the crude reaction mixture is cleaner, reducing the burden on downstream purification processes like column chromatography. For manufacturers of high-purity electronic chemical or pharmaceutical intermediates, this level of control over the impurity profile is essential for meeting stringent regulatory specifications and ensuring batch-to-batch consistency.
How to Synthesize Enol Silyl Ethers Efficiently
Implementing this advanced synthetic strategy requires precise adherence to the optimized parameters outlined in the patent data to ensure maximum yield and reproducibility. The process is designed to be operationally simple, requiring standard laboratory glassware and common solvents, which facilitates easy translation from bench scale to pilot plant operations. The key to success lies in the correct stoichiometric balance between the silyl acylate substrate and the nitroalkane nucleophile, as well as the careful selection of the base and catalyst loading. Operators should note that while the reaction is robust, maintaining the specified stirring speed and vacuum conditions during workup is crucial for isolating the product in high purity. The detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the guide below.
- Charge a reaction vessel with silyl acylate substrate, nitroalkane nucleophile, cesium carbonate base, and tetrabutylammonium chloride catalyst in dichloromethane solvent.
- Stir the mixture at room temperature (approx. 25°C) with a rotation speed of 300-500 r/min until reaction completion, typically around 1 hour.
- Remove solvent via vacuum concentration at 35°C and purify the residue by column chromatography using petroleum ether and ethyl acetate to isolate the target enol silyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates directly into tangible strategic advantages regarding cost structure and operational resilience. The shift from cryogenic, multi-step protocols to a single-step, room-temperature process fundamentally alters the cost equation for producing these valuable intermediates. By eliminating the need for energy-intensive cooling systems and reducing the consumption of expensive, specialized reagents, the overall manufacturing cost is significantly reduced. Furthermore, the use of commodity chemicals like nitroalkanes and tetrabutylammonium chloride ensures a stable and secure supply chain, mitigating the risks associated with sourcing niche or hazardous materials. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction conditions. Conventional methods often incur high utility costs due to the maintenance of sub-zero temperatures over extended periods, alongside the expense of handling and disposing of pyrophoric bases. In contrast, this room-temperature protocol utilizes inexpensive, shelf-stable reagents and standard solvents like dichloromethane. The elimination of cryogenic infrastructure not only lowers capital expenditure but also reduces ongoing operational expenses related to energy consumption and equipment maintenance. Additionally, the high atom economy of the reaction means less raw material is wasted, further driving down the cost per kilogram of the final active ingredient or intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on complex, multi-step syntheses that have multiple points of failure. This streamlined one-pot synthesis minimizes unit operations, thereby reducing the probability of batch failures and delays. The raw materials, specifically nitroalkanes and silyl acylates, are widely available from bulk chemical suppliers, ensuring that production is not bottlenecked by the scarcity of exotic reagents. This accessibility allows for more flexible inventory management and faster response times to fluctuations in market demand. For a reliable pharmaceutical intermediates supplier, this robustness is a key differentiator that ensures consistent product availability for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to multi-ton production is notoriously difficult when harsh conditions are involved. The mild nature of this phase-transfer catalyzed reaction makes it inherently safer and easier to scale. The absence of extreme temperatures and hazardous reagents simplifies the engineering controls required for large reactors, facilitating a smoother transition to commercial manufacturing. Moreover, the improved atom economy and reduced waste generation align with increasingly strict environmental regulations and sustainability goals. By minimizing the generation of saline waste and avoiding the use of toxic heavy metals or pyrophoric substances, this method supports greener manufacturing practices and reduces the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. The answers highlight the versatility of the substrate scope and the practical advantages of the operational parameters.
Q: What are the primary advantages of this new synthesis method over traditional enol silyl ether production?
A: Unlike conventional methods that require harsh conditions such as cryogenic temperatures (-70°C) and strong, non-nucleophilic bases like LDA, this novel approach operates efficiently at room temperature. It utilizes cheap and readily available nitroalkanes as nucleophiles and employs a phase transfer catalyst, resulting in higher atom economy and simplified operational procedures suitable for industrial scaling.
Q: What is the expected yield and purity profile for the synthesized enol silyl ethers?
A: Experimental data from the patent indicates high reaction efficiency, with yields reaching up to 94% for specific substrates like benzyl 2-(tert-butyldimethylsilyloxy)-2-oxoacetate derivatives. The mild reaction conditions minimize side reactions, facilitating the production of high-purity intermediates essential for downstream pharmaceutical applications.
Q: Is this process scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly amenable to commercial scale-up. The use of common solvents like dichloromethane, the absence of extreme temperature requirements, and the use of stable, commercially available reagents like tetrabutylammonium chloride make it robust for large-scale production environments without needing specialized cryogenic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enol Silyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the phase-transfer catalyzed synthesis of enol silyl ethers for the next generation of pharmaceutical and fine chemical products. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. We are committed to delivering high-purity enol silyl ethers that meet the most stringent purity specifications required by the global pharmaceutical industry. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that our clients receive materials of the highest quality and consistency.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you are looking to optimize an existing supply chain or develop a new synthetic route for a complex intermediate, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and comprehensive route feasibility assessments to determine how this efficient synthesis method can enhance your production capabilities and reduce your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
