Advanced Synthesis of Tribenoside: Enhancing Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with scalable manufacturing capabilities. Patent CN109776624B, published in early 2022, introduces a transformative preparation method for Tribenoside, a critical capillary vessel protectant widely used in the treatment of hemorrhoids and vascular disorders. This technical disclosure addresses long-standing inefficiencies in the production of ethyl-3,5,6-tribenzyloxy-D-furanoside by replacing cumbersome chromatographic separations with a streamlined crystallization protocol. The innovation lies in a precise two-step refining strategy that converts a crude oily intermediate into a high-purity solid, subsequently enabling a clean etherification reaction. For R&D directors and supply chain managers, this represents a pivotal shift towards more sustainable and cost-effective pharmaceutical intermediates manufacturing, eliminating the bottlenecks associated with traditional purification techniques.

Tribenoside, chemically known as ethyl-3,5,6-tribenzyloxy-D-furanoside, serves as a vital active pharmaceutical ingredient with potent anti-inflammatory and wound-healing properties. Historically, the synthesis of this molecule has been plagued by low yields and complex purification requirements. The structural complexity, featuring multiple benzyl protecting groups and a specific furanoside configuration, demands rigorous control over stereochemistry and impurity profiles. The new method detailed in the patent leverages a specific acid-catalyzed deprotection mechanism followed by sequential solvent engineering to achieve unprecedented purity levels. This approach not only enhances the quality of the final high-purity pharmaceutical intermediate but also aligns with modern green chemistry principles by reducing solvent waste and processing time.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those described in US Patent 3157634 and GB Patent 1310382, have historically relied on starting materials with suboptimal purity, leading to a cascade of byproduct formation throughout the synthesis. As illustrated in the legacy reaction schemes, these conventional pathways often necessitate repeated recrystallizations, distillation, and, most critically, low-pressure column chromatography to isolate the target compound. Column chromatography, while effective on a laboratory scale, is notoriously difficult to scale up for commercial production due to high solvent consumption, low throughput, and significant batch-to-batch variability. Furthermore, the reliance on impure starting materials in these older routes results in a complex impurity spectrum that is challenging to separate, ultimately capping the overall yield and compromising the economic viability of large-scale commercial scale-up of complex pharmaceutical intermediates.
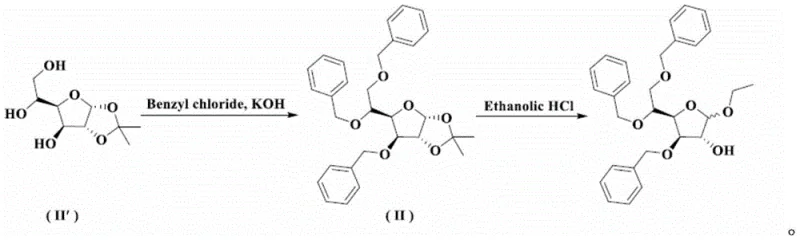
The Novel Approach
In stark contrast, the methodology disclosed in CN109776624B introduces a sophisticated yet operationally simple strategy that bypasses the need for chromatographic separation entirely. The core innovation involves converting the initial deprotection product, which typically exists as an intractable oil, into a crystalline solid through a carefully optimized solvent system. By utilizing a mixed solvent system of ether and hydrocarbons for the first refinement, followed by an alcohol and hydrocarbon mixture for the second, the process effectively precipitates the desired intermediate while leaving impurities in the solution. This physical separation technique is far more amenable to industrial filtration and drying processes than chromatography. The result is a highly pure intermediate that feeds directly into the final etherification step, ensuring that the final Tribenoside product meets stringent quality specifications with minimal downstream processing.
Mechanistic Insights into Acid-Catalyzed Deprotection and Etherification
The chemical foundation of this improved synthesis rests on a controlled acid-catalyzed deprotection of the isopropylidene group from the starting glucopyranose derivative. The patent specifies the use of a sulfuric acid-acetic acid system, where the ratio of reactants is critically tuned to facilitate the cleavage of the acetal protecting group without inducing excessive degradation of the sensitive glycosidic backbone. The reaction proceeds through a protonation of the acetal oxygen, followed by the elimination of acetone and the formation of a reactive oxocarbenium ion, which is subsequently trapped by water to form the hemiacetal. Maintaining the reaction temperature between 65°C and 100°C is crucial; lower temperatures result in incomplete conversion, while higher temperatures risk hydrolyzing the benzyl ethers or promoting caramelization. This precise control over the acidic environment ensures that the crude product retains the necessary structural integrity for the subsequent purification steps.
Following deprotection, the mechanism shifts to physical chemistry, specifically solubility differentials, to achieve purification. The crude oily product contains various isomers and degradation byproducts that possess different solubility profiles in polar versus non-polar media. The first refinement step exploits the differential solubility in an isopropyl ether-cyclohexane system to induce nucleation and crystal growth of the target intermediate. The second refinement, using an isopropanol-n-heptane system with activated carbon, further adsorbs colored impurities and trace organic contaminants. Finally, the purified intermediate undergoes Fischer glycosidation with ethanol in the presence of hydrochloric acid. This etherification proceeds via an SN1-like mechanism where the anomeric hydroxyl group is protonated and displaced by ethanol, forming the stable ethyl glycoside. The mild conditions (-10°C to 25°C) prevent anomerization and ensure the preservation of the desired stereochemical configuration.
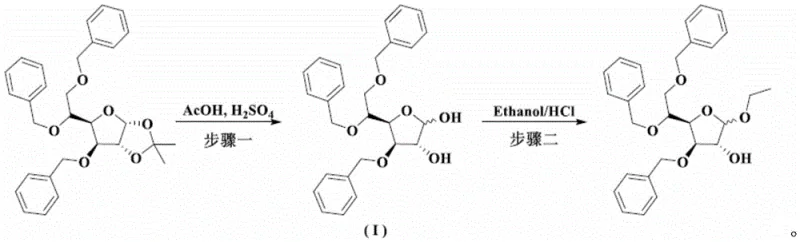
How to Synthesize Ethyl-3,5,6-tribenzyloxy-D-glucopyranoside Efficiently
The synthesis of this critical intermediate requires strict adherence to the solvent ratios and temperature profiles outlined in the patent to ensure reproducibility and high purity. The process begins with the preparation of the acid catalyst mixture, followed by the controlled addition of the starting material to manage exothermicity. Operators must monitor the reaction progress closely to determine the optimal endpoint for deprotection before quenching the reaction. The subsequent crystallization steps are equally critical; the rate of cooling and the volume of anti-solvent added dictate the crystal habit and purity of the solid. Detailed standard operating procedures regarding the specific mass ratios of sulfuric acid to acetic acid, as well as the volume ratios of the refining solvents, are essential for successful implementation. For a comprehensive guide on the exact operational parameters and safety considerations, please refer to the standardized synthesis steps provided below.
- Perform acidic deprotection of 3,5,6-tribenzyloxy-1,2-oxo-isopropylidene-alpha-D-glucopyranose using a sulfuric acid-acetic acid system at 65-100°C.
- Execute a two-stage purification: first refine the crude oil in an ether-hydrocarbon solvent, then further purify the solid in an alcohol-hydrocarbon solvent.
- Conduct the final etherification reaction of the pure intermediate with ethanol under acidic conditions (HCl-Ethanol) at -10 to 25°C to obtain the target Tribenoside.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chromatography-based purification to crystallization-based refinement offers profound economic and logistical benefits. The elimination of silica gel columns and the associated large volumes of elution solvents drastically reduces the direct material costs and waste disposal fees associated with production. Moreover, crystallization is a unit operation that is inherently easier to scale from pilot plant to full commercial production compared to chromatography, which often requires specialized equipment and longer cycle times. This process intensification leads to a significant reduction in the overall production period, allowing manufacturers to respond more agilely to market demand fluctuations. The ability to produce high-purity pharmaceutical intermediates with a simplified workflow translates directly into improved margin potential and supply security for downstream API manufacturers.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the removal of column chromatography, a notoriously expensive and solvent-intensive process. By replacing this with a two-step crystallization protocol, the consumption of high-grade organic solvents is minimized, and the need for costly stationary phases is eliminated. Additionally, the higher yield of the final etherification step, driven by the use of a purer intermediate, means that less raw material is wasted per kilogram of finished product. This cumulative effect results in substantial cost savings across the entire value chain, making the final Tribenoside product more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the new synthetic route. The reagents required, such as sulfuric acid, acetic acid, and common hydrocarbon solvents, are commodity chemicals with stable global supply lines, reducing the risk of raw material shortages. Furthermore, the shortened production cycle time means that inventory turnover is faster, allowing suppliers to maintain leaner stock levels while still meeting delivery commitments. This agility is crucial for mitigating risks associated with logistics disruptions or sudden spikes in demand for vascular health medications, ensuring a continuous flow of materials to pharmaceutical formulators.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the new method offers a cleaner profile that aligns with increasingly stringent global emissions standards. The reduction in solvent waste lowers the burden on wastewater treatment facilities and decreases the facility's overall carbon footprint. The crystallization process generates solid waste that is often easier to handle and dispose of than the liquid waste streams generated by chromatography. This environmental advantage not only simplifies regulatory compliance but also enhances the corporate sustainability profile of the manufacturer, a key factor for multinational corporations prioritizing green supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Tribenoside synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this technology outperforms legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production. The answers highlight the specific advantages in purity, yield, and operational simplicity that define this technological advancement.
Q: Why is the new purification method superior to column chromatography for Tribenoside?
A: The new method replaces time-consuming and low-throughput column chromatography with a robust two-step crystallization process. This significantly improves operational efficiency, reduces solvent consumption, and achieves higher purity levels suitable for industrial scaling.
Q: What specific solvent systems are utilized for the dual-stage refinement?
A: The process utilizes an isopropyl ether-cyclohexane system for the primary refinement to solidify the crude oil, followed by an isopropanol-n-heptane system for the secondary refinement to remove trace impurities and achieve >99% purity.
Q: How does this synthesis route impact the impurity profile of the final API intermediate?
A: By strictly controlling the acid-catalyzed deprotection and employing sequential crystallization, the method drastically reduces known impurities such as benzyl alcohol and benzaldehyde to trace levels (below 0.02%), ensuring a cleaner impurity spectrum for downstream drug formulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tribenoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of effective vascular therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Tribenoside and related pharmaceutical intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to implement the advanced crystallization protocols described in recent patents allows us to offer a superior product profile that meets the exacting demands of global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can drive value for your organization. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability. Let us be your trusted partner in bringing high-quality vascular health solutions to the market.
