Optimizing Tribenoside Synthesis: A High-Purity, Genotoxic-Free Route for Commercial Scale-Up
Optimizing Tribenoside Synthesis: A High-Purity, Genotoxic-Free Route for Commercial Scale-Up
The pharmaceutical landscape for venous active agents continues to evolve, driven by the demand for safer, higher-purity intermediates that streamline regulatory approval processes. Patent CN112028950B introduces a transformative preparation method for Tribenoside (CAS 10310-32-4), a critical active pharmaceutical ingredient widely utilized for its capillary protective and anti-inflammatory properties in treating hemorrhoidal disease. This technical insight analyzes the shift from hazardous traditional acid-catalyzed glycosylation to a refined trifluoroacetic acid (TFA) mediated process. By replacing corrosive mineral acids with organic acid catalysis in an ethanol system, this innovation addresses long-standing challenges regarding genotoxic impurity formation and reaction equilibrium. For R&D directors and procurement strategists, understanding this mechanistic pivot is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering material that meets increasingly rigorous global safety standards.
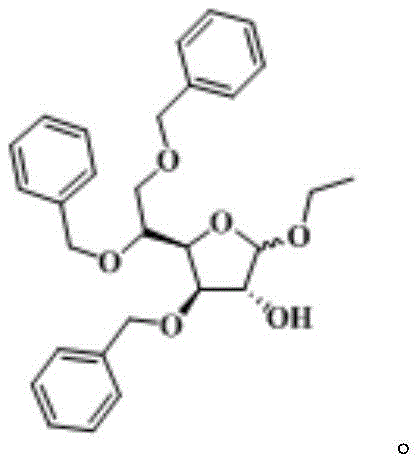
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tribenoside has relied heavily on strong mineral acids such as hydrochloric acid or sulfuric acid within an ethanol solvent system, as documented in legacy patents like US3157634 and GB1310382. While chemically feasible, these conventional routes suffer from severe drawbacks that compromise both operational safety and product quality. The primary concern lies in the side reactions inherent to using HCl or H2SO4 in ethanol; these conditions inevitably promote the formation of ethyl chloride and diethyl sulfate. As highlighted in recent toxicological assessments, these byproducts possess genotoxic warning structures, posing significant risks to patient safety and creating immense burdens for quality control laboratories tasked with detecting them at parts-per-million levels.

Furthermore, the presence of water generated during the glycosylation reaction in these older methods often drives the equilibrium backward, leading to hydrolysis of the starting material or the product itself. This results in suboptimal yields, typically necessitating multiple rounds of energy-intensive recrystallization and distillation to achieve acceptable purity. Such extensive downstream processing not only inflates production costs but also increases the environmental footprint through solvent waste generation. For supply chain managers, reliance on these outdated methodologies introduces volatility, as the difficulty in purging genotoxic impurities can lead to batch failures and unpredictable lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN112028950B represents a paradigm shift by employing a trifluoroacetic acid (TFA) and ethanol system, augmented by triethyl orthoformate. This approach elegantly circumvents the formation of alkyl halides and sulfate esters, effectively eliminating the risk of genotoxic contamination at the source rather than attempting to remove it post-reaction. The use of TFA provides sufficient acidity to catalyze the glycosidic bond formation under mild conditions, typically between 0°C and 5°C, preserving the stereochemical integrity of the sugar moiety. Moreover, the strategic inclusion of triethyl orthoformate serves a dual purpose: it acts as a reactant and a dehydrating agent, scavenging the water produced during etherification. This drives the reaction equilibrium decisively toward the product side, resulting in substantially improved conversion rates and simplified isolation procedures.
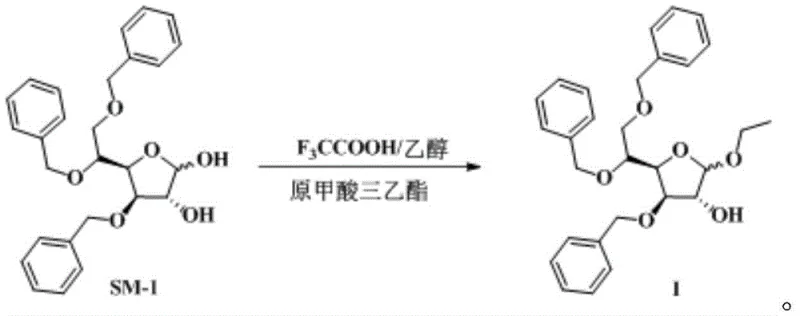
Mechanistic Insights into TFA-Catalyzed Glycosylation
To fully appreciate the technical superiority of this route, one must examine the underlying reaction mechanism. The transformation begins with the protonation of the anomeric hydroxyl group of the starting material, 3,5,6-tribenzyloxy-D-glucopyranose (SM-1), by trifluoroacetic acid. Unlike the aggressive protonation seen with mineral acids, TFA offers a more controlled acidic environment that minimizes degradation of the sensitive furanose ring system. The subsequent nucleophilic attack by ethanol leads to the formation of the ethyl glycoside bond. Crucially, this step releases a molecule of water, which, if left unchecked, would hydrolyze the newly formed glycosidic linkage back to the hemiacetal. Here, triethyl orthoformate reacts with the liberated water to form ethyl formate and ethanol, effectively removing water from the reaction matrix. This Le Chatelier-driven process ensures that the forward reaction is favored kinetically and thermodynamically.
From an impurity control perspective, this mechanism is robust. By avoiding chloride and sulfate ions entirely, the pathway precludes the nucleophilic substitution reactions that generate ethyl chloride and diethyl sulfate. The result is a crude reaction mixture that is inherently cleaner, reducing the load on purification columns and crystallization vessels. For process chemists, this means a more predictable impurity profile where the focus can remain on controlling diastereomeric ratios (alpha vs. beta anomers) rather than chasing trace genotoxins. The ability to operate at near-ambient temperatures (0-10°C) further suppresses thermal degradation pathways, ensuring that the final API intermediate retains the high optical purity required for biological efficacy.
How to Synthesize Tribenoside Efficiently
The practical execution of this synthesis is designed for scalability and reproducibility in a GMP environment. The process initiates by dissolving the protected glucose derivative in absolute ethanol, followed by the careful addition of the acid catalyst under cooling. The subsequent addition of the dehydrating agent locks in the yield potential. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup parameters are critical for maintaining the high purity specifications observed in the patent examples.
- Dissolve 3,5,6-tribenzyloxy-D-glucopyranose (SM-1) in absolute ethanol and cool the mixture to a controlled temperature range of 0°C to 5°C.
- Add trifluoroacetic acid (or its ethanol solution) dropwise while maintaining strict temperature control, followed by stirring for approximately 1 hour to activate the glycosyl donor.
- Introduce triethyl orthoformate as a dehydrating agent to drive the equilibrium forward, maintain reaction temperature for 2.5 hours, and proceed to aqueous workup and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TFA-mediated synthesis offers tangible strategic benefits beyond mere chemical elegance. The elimination of genotoxic impurities translates directly into reduced analytical testing costs and lower risk of regulatory rejection during drug master file (DMF) reviews. By sourcing Tribenoside manufactured via this cleaner route, pharmaceutical companies can significantly streamline their vendor qualification audits and reduce the complexity of their incoming quality control protocols.
- Cost Reduction in Manufacturing: The enhanced reaction yield, reported to reach levels as high as 93.8% in optimized examples, fundamentally improves material efficiency. Higher yields mean less raw material is required per kilogram of finished product, directly lowering the cost of goods sold (COGS). Furthermore, the simplification of the workup procedure—avoiding multiple recrystallizations—reduces solvent consumption, energy usage for heating and cooling, and labor hours, contributing to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Traditional methods prone to batch-to-batch variability due to difficult purification steps create supply bottlenecks. The robustness of the new method, characterized by mild conditions and high selectivity, ensures consistent production output. This reliability allows for more accurate forecasting and inventory planning, reducing the lead time for high-purity pharmaceutical intermediates and safeguarding against production stoppages caused by failed quality tests.
- Scalability and Environmental Compliance: The avoidance of hazardous reagents like thionyl chloride or concentrated sulfuric acid improves the safety profile of the manufacturing plant, lowering insurance premiums and compliance overheads. Additionally, the reduced solvent waste and energy demand align with green chemistry principles, making the commercial scale-up of complex pharmaceutical intermediates more sustainable and compliant with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents the current state-of-the-art.
Q: Why is the TFA-Ethanol system superior to traditional HCl-Ethanol methods for Tribenoside?
A: Traditional HCl-Ethanol systems generate ethyl chloride and diethyl sulfate, which are genotoxic impurities requiring stringent and costly removal. The TFA-Ethanol route described in CN112028950B completely avoids these hazardous byproducts, ensuring a safer impurity profile.
Q: What role does triethyl orthoformate play in this synthesis?
A: Triethyl orthoformate acts as a water scavenger. Since glycosylation produces water which can reverse the reaction or hydrolyze the product, removing water shifts the chemical equilibrium towards the desired Tribenoside, significantly boosting yield.
Q: What purity levels can be achieved with this novel method?
A: Experimental data indicates that this method consistently achieves purity levels exceeding 99%, with specific examples demonstrating up to 99.65% purity without the need for complex recrystallization steps often required in older protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tribenoside Supplier
As the global demand for high-quality venous active agents grows, partnering with a manufacturer who understands the nuances of advanced synthetic chemistry is paramount. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this efficient Tribenoside synthesis to the market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Tribenoside we supply is free from genotoxic impurities and ready for immediate formulation.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By switching to our optimized supply chain, you can secure a stable source of this critical intermediate while reducing total landed costs. Contact our technical procurement team today to request specific COA data and route feasibility assessments for your next project.
