Advanced Alkaline Synthesis of Tribenoside: Eliminating Genotoxic Risks for Commercial Scale-up
Advanced Alkaline Synthesis of Tribenoside: Eliminating Genotoxic Risks for Commercial Scale-up
The pharmaceutical industry constantly seeks robust synthetic routes that balance high efficiency with stringent safety profiles, particularly for active pharmaceutical ingredients (APIs) and their critical intermediates. Patent CN112028947B introduces a groundbreaking synthetic method for Tribenoside, a vital capillary vessel protectant used extensively in hemorrhoid treatments and wound healing applications. This innovation shifts the paradigm from traditional harsh acidic conditions to a mild, alkaline catalytic system, effectively resolving long-standing issues regarding genotoxic impurities and low yields. By leveraging a unique combination of triethyl orthoformate and alkaline reagents in an ethanol-DMSO system, this technology offers a pathway to high-purity products suitable for global regulatory standards. For R&D directors and procurement specialists, understanding this transition is key to securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality.
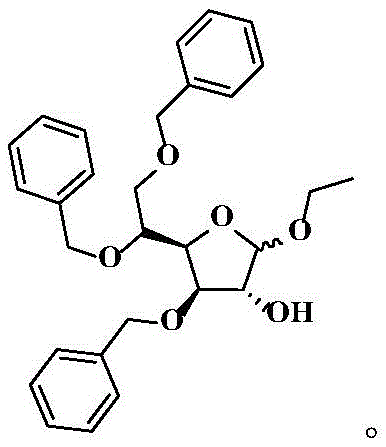
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tribenoside has relied heavily on acidic catalysis systems, typically utilizing hydrochloric acid or sulfuric acid in ethanol solutions, as depicted in earlier patents like US3157634 and GB1310382. These conventional routes suffer from severe drawbacks, primarily the formation of hazardous genotoxic impurities such as ethyl chloride and diethyl sulfate during the reaction process. The presence of these mutagenic byproducts necessitates complex and costly purification steps, including repeated recrystallization and distillation, which drastically reduce the overall process efficiency. Furthermore, the acidic environment often leads to lower reaction yields, typically hovering around 70 percent, and final product purities that struggle to exceed 94 percent. The generation of water as a byproduct in these glycosylation reactions further inhibits forward progress, creating an equilibrium bottleneck that limits industrial scalability. Consequently, manufacturers face significant challenges in meeting modern safety regulations while maintaining cost-effective production schedules.
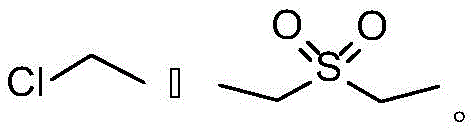
The Novel Approach
In stark contrast, the method disclosed in CN112028947B utilizes a sophisticated alkaline system that fundamentally eliminates the risk of forming genotoxic warning structures. By replacing corrosive acids with mild bases like potassium hydroxide (KOH) and incorporating triethyl orthoformate, the reaction proceeds under much gentler conditions that preserve the integrity of the molecular structure. This novel approach not only prevents the formation of ethyl chloride and diethyl sulfate but also actively drives the reaction forward by scavenging the water produced during glycosylation. The result is a dramatic improvement in both yield and purity, with experimental data demonstrating yields exceeding 94 percent and purities reaching 99.73 percent. This shift represents a critical advancement for cost reduction in pharmaceutical intermediate manufacturing, as it simplifies downstream processing and reduces the burden of impurity control. The streamlined workflow ensures that the final Tribenoside product meets the rigorous specifications required for clinical applications without the need for excessive purification cycles.
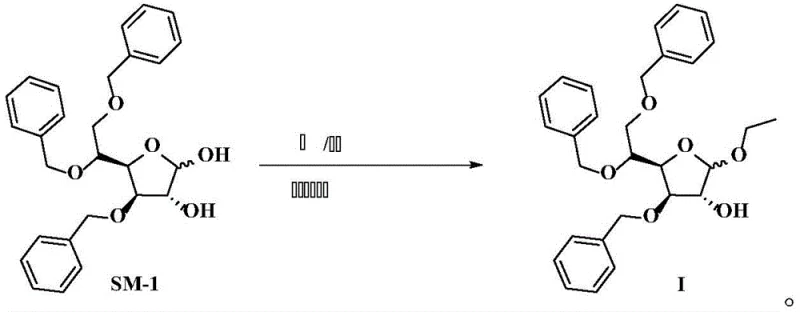
Mechanistic Insights into Alkaline Catalytic Glycosylation
The core of this technological breakthrough lies in the synergistic interaction between the alkaline catalyst and the dehydrating agent within the reaction matrix. In this mechanism, the base, preferably KOH, activates the hydroxyl groups on the 3,5,6-tribenzyloxy-D-glucopyranose (SM-1) substrate, facilitating nucleophilic attack on the ethanol source. Unlike acidic mechanisms that protonate the leaving group and risk side reactions, the alkaline environment promotes a cleaner substitution pathway that minimizes degradation of the sensitive furanoside ring. The addition of triethyl orthoformate is particularly ingenious; it reacts with the water generated during the etherification to form ethanol and ethyl formate, effectively removing water from the equilibrium. This dehydration effect pushes the reaction kinetics strongly towards the product side, overcoming the thermodynamic limitations that plague traditional aqueous or wet acidic systems. Such precise control over the reaction environment allows for the consistent production of high-quality intermediates essential for complex API synthesis.
Furthermore, the choice of solvent system plays a pivotal role in stabilizing the transition states and ensuring complete solubility of the reactants. The patent specifies a mixed solvent system of absolute ethanol and dimethyl sulfoxide (DMSO), which provides the necessary polarity to dissolve the ionic base and the organic substrate simultaneously. This homogeneity ensures that the reaction proceeds uniformly throughout the bulk solution, preventing localized hot spots or concentration gradients that could lead to byproduct formation. The absence of strong acids also means that the equipment corrosion rates are significantly lowered, extending the lifespan of reactor vessels and reducing maintenance downtime. From a quality control perspective, the elimination of acid-catalyzed side reactions results in a much cleaner impurity profile, simplifying the analytical validation required for regulatory filings. This mechanistic clarity provides R&D teams with the confidence to scale the process from laboratory benchtop to multi-ton commercial production.
How to Synthesize Tribenoside Efficiently
Implementing this advanced synthesis requires careful attention to reagent ratios and temperature control to maximize the benefits of the alkaline system. The process begins by dissolving the starting material, SM-1, in a mixture of anhydrous ethanol and DMSO, followed by the sequential addition of the base and triethyl orthoformate. Maintaining a reflux temperature ensures sufficient energy for the reaction to proceed to completion while the dehydrating agent continuously removes water. Detailed standard operating procedures regarding specific molar ratios, such as the preferred 1:2 ratio of SM-1 to base, are critical for reproducibility. For a comprehensive guide on the exact operational parameters and workup procedures, please refer to the standardized synthesis steps outlined below.
- Dissolve 3,5,6-tribenzyloxy-D-glucopyranose (SM-1) in a mixed solution of absolute ethanol and an organic solvent such as DMSO at room temperature.
- Add an alkaline reagent (preferably KOH) and triethyl orthoformate to the reaction mixture under stirring.
- Heat the mixture to reflux until reaction completion, then perform aqueous workup including extraction, washing, and drying to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this alkaline synthesis route offers profound strategic advantages beyond mere technical metrics. The elimination of genotoxic impurities removes a major regulatory hurdle, significantly reducing the time and cost associated with safety testing and batch rejection. By avoiding the use of hazardous acids and the subsequent generation of toxic byproducts, the process aligns perfectly with green chemistry principles, lowering waste disposal costs and environmental compliance burdens. The high yield and purity achieved directly translate to better raw material utilization, meaning less starting material is wasted per kilogram of final product. This efficiency creates a more resilient supply chain capable of meeting fluctuating market demands without the volatility associated with low-yield processes. Ultimately, this technology supports a sustainable and cost-effective sourcing strategy for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The shift to an alkaline system eliminates the need for expensive corrosion-resistant equipment and complex neutralization steps required by acidic processes. By preventing the formation of difficult-to-remove genotoxic impurities, the downstream purification workload is drastically simplified, leading to substantial savings in solvent usage and labor hours. The high conversion rate ensures that the cost of goods sold (COGS) is minimized through superior atom economy and reduced waste generation. Additionally, the mild reaction conditions lower energy consumption compared to high-pressure or extreme temperature alternatives. These factors combined create a highly competitive cost structure for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The use of commercially available and stable reagents like KOH and triethyl orthoformate mitigates the risk of raw material shortages that can plague specialized catalyst supply chains. Because the process is less sensitive to moisture and operational variances than acidic methods, it offers greater flexibility in manufacturing scheduling and location. This reliability allows supply chain planners to forecast inventory needs with higher accuracy and reduce safety stock levels. Consequently, partners can rely on a steady flow of high-purity intermediates to support their own production timelines.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of exothermic hazards associated with strong acid additions. The benign nature of the reagents simplifies effluent treatment, as the wastewater does not require extensive neutralization or heavy metal removal protocols. This environmental compatibility facilitates faster permitting and regulatory approval for new manufacturing facilities. The process design inherently supports continuous improvement and optimization, allowing for seamless capacity expansion as market demand grows. Such scalability ensures that the supply of Tribenoside can keep pace with the global demand for hemorrhoid treatments and vascular health medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Tribenoside synthesis. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the consensus on safety, efficiency, and regulatory alignment provided by the inventors.
Q: Why is the alkaline synthesis method superior to traditional acidic methods for Tribenoside?
A: Traditional acidic methods using HCl or H2SO4 generate genotoxic impurities like ethyl chloride and diethyl sulfate. The alkaline method described in CN112028947B completely avoids these hazardous byproducts, ensuring higher safety and regulatory compliance.
Q: What role does triethyl orthoformate play in this synthesis?
A: Triethyl orthoformate acts as a dehydrating agent. Since glycosylation produces water which can reverse the reaction, removing water drives the equilibrium towards the product, significantly improving yield and purity.
Q: What purity levels can be achieved with this new process?
A: The patented alkaline process consistently achieves purity levels exceeding 99%, with yields reaching up to 94.2%, which is a substantial improvement over the ~94% purity and 70% yield of older acidic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tribenoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the highest standards of product quality and safety. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this alkaline catalysis technology are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Tribenoside meets the exacting requirements of the global pharmaceutical market. Our commitment to innovation allows us to offer a reliable pharmaceutical intermediate supplier partnership that prioritizes both technical excellence and supply security.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient process. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to drive efficiency and quality in your supply chain together.
