Advanced Synthesis of Lamivudine Intermediates: Enhancing Safety and Scalability for Global Pharma Supply Chains
Advanced Synthesis of Lamivudine Intermediates: Enhancing Safety and Scalability for Global Pharma Supply Chains
The global demand for effective antiviral therapies, particularly for Hepatitis B and HIV, continues to drive the need for robust and scalable manufacturing processes for key active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in Patent CN101362752B, which discloses a novel synthetic method for producing the critical lamivudine intermediate, specifically (2R,5S)-5-(cytosin-1-yl)-1,3-oxathiolane-2-carboxylic acid-L-menthyl ester. This patent represents a significant technological leap by replacing traditional, hazardous chlorinating agents with a safer, solid-state alternative, thereby addressing long-standing safety and environmental concerns in fine chemical manufacturing. For R&D directors and procurement specialists, understanding this shift is crucial, as it directly impacts the reliability of the reliable pharmaceutical intermediates supplier network and the overall cost structure of antiviral drug production. The innovation lies not just in the chemical transformation but in the holistic improvement of the process safety profile, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of lamivudine intermediates has relied heavily on thionyl chloride (SOCl2) as the primary chlorinating agent to convert the hydroxyl group of the oxathiolane ring into a chloride. While chemically effective, this conventional approach presents severe operational and environmental drawbacks that modern manufacturing facilities strive to eliminate. Thionyl chloride is a volatile, corrosive liquid that poses significant health risks to operators and requires specialized, corrosion-resistant reactor equipment to handle safely. During the reaction, it generates large volumes of sulfur dioxide (SO2) gas, a toxic pollutant that necessitates complex and expensive scrubbing systems to prevent atmospheric release. Furthermore, the exothermic nature of the reaction with thionyl chloride can be difficult to control on a large scale, leading to potential safety incidents and variability in product quality. These factors collectively increase the capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturers, creating a bottleneck in the cost reduction in pharmaceutical intermediates manufacturing initiatives.
The Novel Approach
In a strategic move towards greener chemistry, Patent CN101362752B introduces the use of bis(trichloromethyl) carbonate, commonly known as Triphosgene, as a superior substitute for thionyl chloride. Unlike its liquid counterpart, Triphosgene is a crystalline solid at room temperature, which fundamentally changes the logistics and handling dynamics of the production process. Being a solid, it eliminates the risks associated with pumping and storing hazardous liquids, allowing for precise gravimetric dosing that enhances reaction reproducibility. Chemically, Triphosgene acts as a phosgene equivalent but decomposes to release carbon dioxide (CO2) as the primary gaseous byproduct, which is far less environmentally damaging than sulfur dioxide. This substitution effectively removes the need for aggressive acid gas scrubbing systems and reduces the corrosion burden on reactor vessels. The structural integrity and stability of this reagent are depicted below, highlighting its suitability for controlled chlorination reactions.
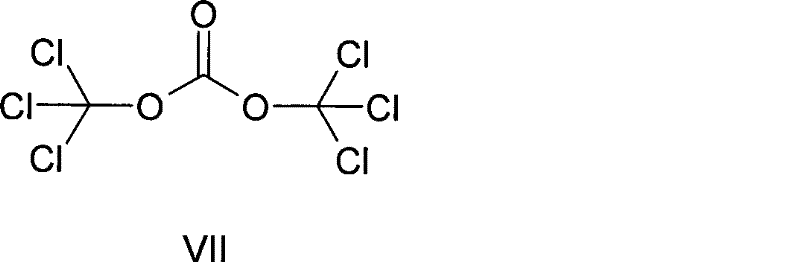
By integrating this reagent, the process achieves a level of operational safety and environmental compliance that aligns with modern ESG (Environmental, Social, and Governance) standards, making it a highly attractive route for high-purity pharmaceutical intermediates production.
Mechanistic Insights into Triphosgene-Mediated Chlorination and Condensation
The core of this innovative synthesis lies in the efficient chlorination of the (2R,5S)-5-hydroxyl-1,3-oxathiolane-2-carboxylic acid-L-menthyl ester substrate. In the presence of a catalytic amount of N,N-dimethylformamide (DMF), Triphosgene reacts with the hydroxyl group to form the corresponding chloro-intermediate (Compound IV). The mechanism involves the activation of the hydroxyl group by the chloroformate species generated in situ from Triphosgene, followed by nucleophilic displacement by chloride ions. This pathway is remarkably mild, typically proceeding at temperatures between 0°C and 50°C, which is critical for preserving the stereochemical integrity of the chiral centers at the 2 and 5 positions of the oxathiolane ring. Maintaining chirality is paramount, as the biological activity of lamivudine is strictly dependent on the (2R,5S) configuration; harsh conditions could lead to racemization, generating inactive or potentially toxic isomers that complicate downstream purification.
Following the chlorination, the process moves to the condensation step where the chloro-intermediate reacts with N,O-bis(trimethylsilyl)cytosine. This silylated cytosine derivative acts as a nucleophile, attacking the electrophilic carbon at the 5-position of the oxathiolane ring to displace the chloride and form the glycosidic bond. The use of silylated bases is a standard strategy to enhance solubility and reactivity in non-polar organic solvents like toluene or dichloromethane. The patent highlights that this condensation can be performed in a "one-pot" manner, where the chlorination mixture is directly added to the silylated cytosine solution without isolating the unstable chloro-intermediate. This telescoping of steps minimizes material loss, reduces solvent usage, and significantly shortens the overall cycle time. The hydrolysis of the silyl groups and the menthyl ester subsequently yields the final free acid or allows for further derivatization, ensuring a streamlined path to the target API precursor with minimal impurity generation.
How to Synthesize (2R,5S)-5-(Cytosin-1-yl)-1,3-oxathiolane-2-carboxylic Acid-L-menthyl Ester Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and purity. The patent outlines a robust protocol where the molar ratio of the hydroxy-ester to Triphosgene is optimized between 1:0.35 and 1:1.0, ensuring complete conversion while minimizing excess reagent waste. The reaction is typically initiated at low temperatures (0-10°C) to manage the initial exotherm, followed by a gradual warm-up to 30-35°C to drive the reaction to completion over 1 to 6 hours. For the subsequent coupling, the temperature is maintained between 30°C and 80°C, often utilizing the reflux of the solvent to provide the necessary thermal energy for the nucleophilic substitution. Detailed standardized synthesis steps see the guide below.
- Perform chlorination by reacting (2R,5S)-5-hydroxyl-1,3-oxathiolane-2-carboxylic acid-L-menthyl ester with bis(trichloromethyl) carbonate and DMF in an organic solvent like dichloromethane at 0-50°C.
- Prepare N,O-bis(trimethylsilyl)cytosine by refluxing cytosine with hexamethyldisilazane and a catalyst, then condense it with the chlorinated intermediate.
- Execute workup by pouring the reaction mixture into a hexane/water/triethylamine solution, filtering the solid product, washing, and vacuum drying to obtain the pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the Triphosgene-based route described in Patent CN101362752B offers tangible strategic advantages beyond mere chemical elegance. The transition from a hazardous liquid gas-generator to a stable solid reagent fundamentally de-risks the supply chain. Thionyl chloride often faces regulatory scrutiny and transportation restrictions due to its classification as a toxic inhalation hazard, which can lead to supply disruptions and increased logistics costs. In contrast, Triphosgene is easier to ship, store, and handle, ensuring a more consistent and reliable flow of raw materials. This stability translates directly into reducing lead time for high-purity pharmaceutical intermediates, as manufacturers face fewer regulatory hurdles and safety inspections related to raw material storage. Furthermore, the elimination of sulfur dioxide emissions simplifies environmental permitting and reduces the ongoing costs associated with waste gas treatment facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of unit operations and the reduction of auxiliary costs. By eliminating the need for specialized corrosion-resistant alloys in reactors and extensive gas scrubbing systems, the capital intensity of the production facility is lowered. Additionally, the ability to perform the synthesis in a "one-pot" fashion significantly reduces solvent consumption and labor hours, as there is no need to isolate and dry the intermediate chloro-compound. The qualitative reduction in waste disposal costs, particularly regarding hazardous sulfur-containing waste, further contributes to substantial cost savings, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of Triphosgene, which is a commercially available commodity chemical with a stable global supply base. Unlike thionyl chloride, which may be subject to strict quotas or seasonal production shutdowns due to environmental crackdowns in certain regions, Triphosgene offers a more secure sourcing option. The solid nature of the reagent also allows for longer shelf-life and safer inventory management, enabling manufacturers to maintain strategic stockpiles without the degradation or safety risks associated with liquid chlorinating agents. This reliability ensures that production schedules for critical antiviral medications remain uninterrupted, safeguarding patient access to essential medicines.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is facilitated by the inherent safety of the reagents. The absence of toxic gas evolution (SO2) means that scaling up does not require a proportional increase in expensive emission control infrastructure. The process generates benign byproducts like carbon dioxide and urea derivatives, which are easier to treat and dispose of in accordance with increasingly stringent environmental regulations. This "green" profile not only future-proofs the manufacturing site against tightening laws but also enhances the brand reputation of the manufacturer as a responsible producer of pharmaceutical intermediates, appealing to top-tier multinational clients who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield, purity, and operational feasibility. Understanding these details is essential for technical teams evaluating the transfer of this technology to their own production lines.
Q: Why is bis(trichloromethyl) carbonate preferred over thionyl chloride for this synthesis?
A: Bis(trichloromethyl) carbonate (Triphosgene) is a solid at room temperature, making it significantly safer to transport, store, and meter compared to the toxic and corrosive liquid thionyl chloride. Furthermore, its byproduct is carbon dioxide rather than sulfur dioxide gas, drastically reducing environmental pollution and equipment corrosion risks.
Q: What are the purity specifications achievable with this novel method?
A: According to the experimental data in Patent CN101362752B, this method consistently yields the target intermediate with HPLC purity exceeding 99% and chiral purity above 99%, meeting the stringent requirements for antiviral API production.
Q: Is a one-pot synthesis feasible for this process to reduce costs?
A: Yes, the patent explicitly describes a 'one-pot' methodology where the chlorination and condensation steps are integrated. This approach simplifies the operational workflow, reduces solvent consumption, and lowers overall production costs without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lamivudine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, safe, and efficient synthetic routes like the one described in Patent CN101362752B to meet the evolving demands of the global pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of lamivudine intermediate meets the highest standards for chirality and chemical purity required for antiviral API synthesis. Our infrastructure is designed to handle complex chemistries safely, leveraging our expertise in hazardous reagent management to provide a secure supply partner for your most critical projects.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security for your antiviral drug portfolio.
