Advanced Synthesis of Lamivudine Intermediates: A Scalable Commercial Solution for Global API Manufacturing
Advanced Synthesis of Lamivudine Intermediates: A Scalable Commercial Solution for Global API Manufacturing
The global demand for effective antiretroviral therapies continues to drive innovation in the synthesis of nucleoside analogues, particularly for the treatment of HIV and Hepatitis B. Patent CN104926806B discloses a highly optimized method for synthesizing a critical Lamivudine intermediate, addressing longstanding challenges in yield, purity, and operational feasibility. This technical breakthrough represents a significant leap forward for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials at scale. By refining the stereochemical control and simplifying the downstream processing, this route offers a compelling value proposition for the production of high-purity antiviral agents. The following analysis details the mechanistic advantages and commercial implications of this novel synthetic pathway.
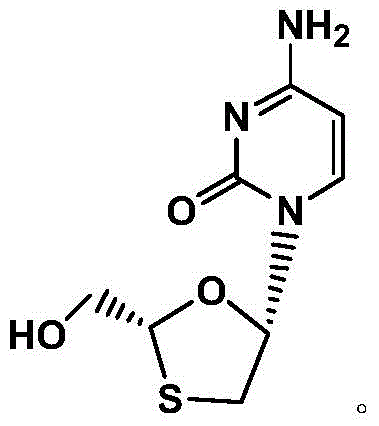
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes, such as those disclosed in earlier patents like CN101307048, have historically struggled with severe operational bottlenecks that hinder industrial scalability. A primary defect in these conventional processes is the tendency for severe emulsification to occur during the post-treatment washing stages, which complicates phase separation and leads to significant product loss. Furthermore, the concentration processes in older methods often result in the generation of large amounts of degradation impurities, compromising the overall purity profile of the intermediate. The separation of enantiomers in these traditional routes is notoriously difficult, often requiring multiple recrystallization steps that drive down yields and increase solvent consumption. These inefficiencies create substantial barriers to cost reduction in pharmaceutical intermediates manufacturing, making the final API prohibitively expensive for broad market access.
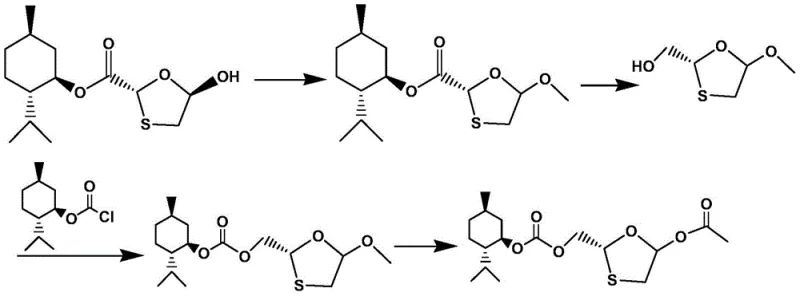
The Novel Approach
The methodology presented in CN104926806B introduces a series of strategic chemical modifications that effectively dismantle the barriers found in legacy processes. By implementing a specific methyl etherification followed by a controlled reduction, the new route establishes a robust foundation for the subsequent glycosylation steps. Crucially, the process replaces problematic solvents and workup conditions with a system designed to prevent emulsification, utilizing ethyl acetate or n-hexane to ensure clean phase separation. The introduction of a mixed solvent system for the final resolution step solves the issue of gel-like agglomeration, ensuring that the crystalline product has excellent fluidity for filtration. This holistic redesign of the synthetic pathway ensures that the commercial scale-up of complex nucleoside analogues is not only feasible but economically superior to previous iterations.
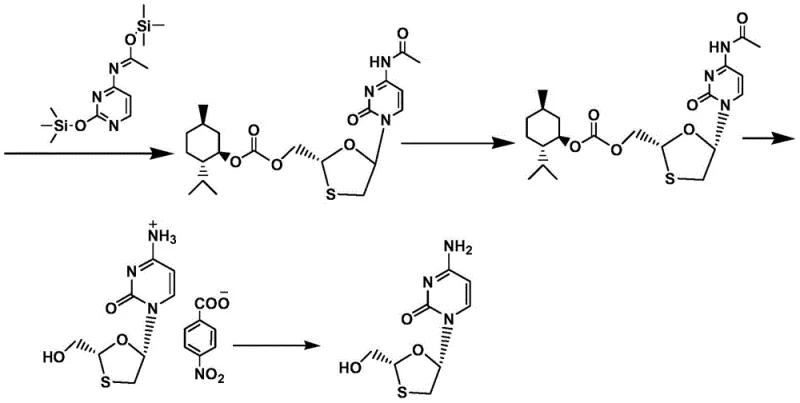
Mechanistic Insights into Stereoselective Glycosylation and Resolution
The core of this synthetic advancement lies in the precise control of stereochemistry during the glycosylation and resolution phases. The process begins with the reduction of the ketone intermediate using sodium borohydride in ethanol, a choice that offers high conversion rates and short reaction times while allowing for the recovery of the chiral auxiliary, menthol. Following esterification and acetylation, the key glycosylation step employs trimethyl iodosilane (TMSI) as a catalyst to couple the sugar moiety with the silane-protected N4-acetamido cytosine. This catalytic system is renowned for its ability to promote the formation of the desired beta-anomer with high stereoselectivity, minimizing the formation of inactive alpha-isomers that would otherwise act as difficult-to-remove impurities.
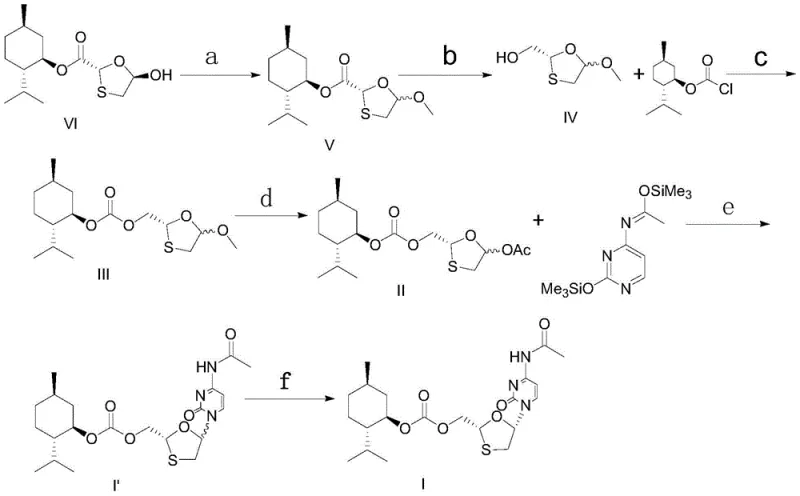
Furthermore, the purification strategy employs a sophisticated recrystallization resolution technique that leverages the differential solubility of diastereoisomers in mixed solvent systems. By dissolving the meso-compound in an alcohol solvent and dripping an alkane solvent under reflux, the process induces crystallization under thermodynamically controlled conditions. This specific solvent combination prevents the formation of amorphous or gel-like solids, which are common failure modes in nucleoside crystallization. The result is a highly crystalline product with a diastereoisomer content of less than 1 percent achieved in a single resolution step. This level of purity control is essential for meeting the stringent regulatory requirements for high-purity pharmaceutical intermediates intended for human therapeutic use.
How to Synthesize Lamivudine Intermediate Efficiently
The synthesis of this critical antiviral intermediate requires strict adherence to the optimized reaction conditions outlined in the patent to ensure maximum yield and stereochemical integrity. The process involves a sequential cascade of etherification, reduction, esterification, acetylation, and finally, stereoselective glycosylation followed by resolution. Each step has been fine-tuned to minimize side reactions and facilitate easy workup, making the entire sequence amenable to large-scale production. For laboratory or pilot plant execution, it is imperative to maintain precise temperature controls, particularly during the acetylation and glycosylation steps, to prevent thermal degradation. The detailed standardized synthesis steps for this process are provided in the guide below.
- Perform methyl etherification on the starting oxathiolane compound followed by sodium borohydride reduction to obtain the key alcohol intermediate.
- Execute esterification with chlorocarbonate and subsequent acetylation using acetic anhydride under controlled acidic conditions to prepare the glycosyl donor.
- Conduct glycosylation with silane-protected cytosine using trimethyl iodosilane catalyst, followed by recrystallization resolution using a mixed alcohol-alkane solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis method offers transformative benefits that directly impact the bottom line and operational reliability. The elimination of emulsification issues during workup translates to significantly reduced processing times and lower labor costs, as operators no longer need to manage stubborn emulsions or perform excessive washing cycles. Additionally, the ability to recover and reuse menthol, a relatively expensive chiral starting material, represents a direct material cost saving that accumulates over large production batches. The robustness of the crystallization process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by off-spec material. These factors combine to create a supply chain that is both more resilient and cost-effective for downstream API manufacturers.
- Cost Reduction in Manufacturing: The process achieves cost optimization through several mechanisms, primarily the efficient recovery of the chiral auxiliary menthol and the use of inexpensive reagents like sodium borohydride and acetic anhydride. By avoiding the need for complex chromatographic separations or multiple recrystallizations to remove degradation products, the overall consumption of solvents and energy is drastically lowered. The high conversion rates in the reduction and glycosylation steps further maximize the throughput of the reactor vessels, improving capital efficiency. Consequently, this route offers a clear pathway for cost reduction in antiviral drug manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The simplified post-treatment procedures, specifically the avoidance of emulsification and the use of non-hazardous solvent systems for extraction, reduce the dependency on specialized waste treatment capabilities. This simplicity allows for a broader range of qualified contract manufacturing organizations (CMOs) to produce the intermediate, thereby diversifying the supply base and mitigating single-source risks. The high purity of the crude product prior to final resolution also means that the final polishing step is more predictable and less prone to failure. This reliability is crucial for maintaining continuous supply lines for essential medicines like Lamivudine, ensuring that reducing lead time for high-purity intermediates becomes a reality.
- Scalability and Environmental Compliance: The transition from single-solvent to mixed-solvent crystallization not only improves product quality but also aligns with green chemistry principles by optimizing solvent usage and recovery. The process avoids the generation of heavy metal waste or persistent organic pollutants, simplifying the environmental compliance burden for manufacturing sites. The physical properties of the final crystalline solid, characterized by good fluidity and lack of agglomeration, facilitate automated handling and packaging, which is a key requirement for multi-ton scale production. This scalability ensures that the technology can seamlessly transition from pilot trials to full commercial production to meet global demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this new standard for intermediate production. The responses highlight the specific advantages in terms of purity, yield, and operational safety that distinguish this method from prior art.
Q: How does this new synthesis method improve upon previous industrial routes for Lamivudine?
A: The new method eliminates critical bottlenecks such as emulsification during washing and degradation during concentration. It introduces a mixed-solvent crystallization system that prevents gel-like agglomeration, significantly improving filtration efficiency and final product purity compared to older single-solvent methods.
Q: What specific catalyst is used for the glycosylation step to ensure stereoselectivity?
A: The process utilizes trimethyl iodosilane (TMSI) as a catalyst for the glycosylation reaction. This reagent provides excellent stereoselectivity and high conversion rates, allowing for the efficient formation of the desired beta-anomer configuration essential for biological activity.
Q: Why is the recovery of menthol significant in this manufacturing process?
A: Menthol is used as a chiral auxiliary in the esterification step. The optimized reduction and workup procedures allow for the recovery and reutilization of menthol through crystallization. This circular usage drastically reduces raw material costs and minimizes waste generation, enhancing the overall economic viability of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lamivudine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral therapies depends on the availability of high-quality, cost-effective intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the stereochemical integrity and impurity profiles of every batch, guaranteeing that the Lamivudine intermediate produced via this advanced route performs flawlessly in downstream API synthesis. We are committed to supporting the global pharmaceutical industry with reliable solutions that enhance both efficacy and accessibility.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest degree of technical confidence and commercial viability.
