Advanced Synthesis of Rosuvastatin Intermediates: A High-Yield Wittig Strategy for Commercial Scale-Up
Advanced Synthesis of Rosuvastatin Intermediates: A High-Yield Wittig Strategy for Commercial Scale-Up
The global demand for HMG-CoA reductase inhibitors, commonly known as statins, continues to drive innovation in pharmaceutical intermediate manufacturing. Specifically, the synthesis of Rosuvastatin, a potent lipid-lowering agent, requires robust and scalable chemical pathways to meet stringent quality and cost targets. Patent CN1307187C introduces a transformative approach to constructing the Rosuvastatin skeleton, shifting away from traditional phosphonate-based methodologies toward a more efficient phosphonium salt strategy. This technical insight report analyzes the mechanistic advantages and commercial implications of this novel route, offering a blueprint for reliable rosuvastatin intermediate supplier partnerships. By leveraging a key Wittig condensation between a pyrimidine-based phosphonium ylide and a chiral side-chain aldehyde, this method achieves superior stereocontrol and yield, addressing critical bottlenecks in cost reduction in statin manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin has relied heavily on the methodology disclosed in European Patent EP0521471A, which utilizes a Horner-Wadsworth-Emmons (HWE) reaction or similar phosphonate condensations. A significant drawback of this conventional route is the reliance on toxic and environmentally hazardous reagents, specifically trialkyl phosphites such as trimethyl phosphite, to generate the necessary phosphonate esters. These reagents pose severe safety risks during large-scale handling and necessitate complex waste treatment protocols to neutralize phosphorus-containing byproducts. Furthermore, literature and patent data indicate that the condensation step in these traditional processes often suffers from moderate efficiency, with reported yields hovering around 60-70%. This inefficiency translates directly into higher raw material consumption and increased downstream purification burdens, as separating the desired E-isomer from reaction impurities becomes more challenging at lower conversion rates. The cumulative effect of these factors is a manufacturing process that is not only costly but also difficult to scale sustainably under modern green chemistry standards.
The Novel Approach
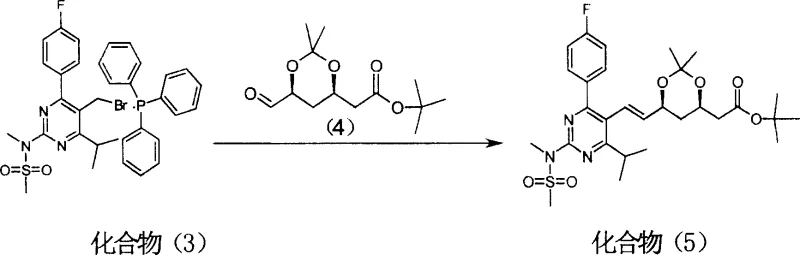
In stark contrast, the methodology outlined in Patent CN1307187C revolutionizes the construction of the statin backbone by inverting the synthetic logic: instead of attaching a phosphonate to the side chain, the pyrimidine core is converted into a reactive phosphonium salt. This strategic pivot allows for a classic Wittig olefination against a chiral aldehyde, a reaction known for its robustness and predictability. The process begins with the bromination of the pyrimidine methanol precursor, followed by quaternization with triphenylphosphine to form a stable phosphonium salt intermediate (Compound 3). This intermediate then undergoes condensation with the side-chain aldehyde (Compound 4) under basic conditions. The result is a dramatic improvement in process metrics; the key condensation step achieves a yield of 78.3%, significantly outperforming the legacy 60-70% benchmark. Moreover, the final product exhibits high purity (98.8% by HPLC) after simple recrystallization, demonstrating that this route inherently suppresses the formation of difficult-to-remove impurities.
Mechanistic Insights into Phosphonium-Mediated Wittig Olefination
The core of this synthetic breakthrough lies in the generation and reactivity of the pyrimidine-based phosphonium ylide. In the presence of a strong base such as sodium hydride (NaH), the phosphonium salt (Compound 3) undergoes deprotonation at the methylene position adjacent to the phosphorus atom. This generates a nucleophilic ylide species that attacks the carbonyl carbon of the chiral aldehyde (Compound 4). The mechanism proceeds through a four-membered oxaphosphetane intermediate, which subsequently collapses to release triphenylphosphine oxide and form the carbon-carbon double bond. Crucially, the reaction conditions—specifically the use of tetrahydrofuran (THF) as a solvent and controlled temperature profiles—favor the formation of the thermodynamically stable E-isomer (trans-alkene), which is the biologically active configuration required for Rosuvastatin. The steric bulk of the triphenylphosphine group and the specific substitution pattern on the pyrimidine ring work synergistically to direct the stereochemical outcome, minimizing the formation of the Z-isomer impurity.
From an impurity control perspective, this mechanism offers distinct advantages over phosphonate routes. In HWE reactions, the formation of phosphate byproducts can sometimes lead to emulsions or difficult extractions. In this Wittig variant, the byproduct is triphenylphosphine oxide, a solid that can often be removed effectively during the aqueous workup or subsequent crystallization steps described in the patent (using ethanol/hexane systems). The high selectivity of the ylide for the aldehyde functionality ensures that other sensitive groups on the chiral side chain, such as the protected hydroxyl groups, remain intact. This chemoselectivity is vital for maintaining the integrity of the chiral centers at the 3R and 5S positions, ensuring that the final API meets the rigorous enantiomeric purity specifications demanded by regulatory agencies for cardiovascular drugs.
How to Synthesize Rosuvastatin Intermediate Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the yield of the novel phosphonium intermediate and the subsequent coupling. The process is divided into distinct operational units: activation of the core, salt formation, coupling, and final deprotection. Each step utilizes common industrial solvents and reagents, facilitating easy technology transfer. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundation for process engineers aiming to replicate this high-efficiency route.
- Bromination of the pyrimidine methanol precursor (Compound 1) using phosphorus tribromide to form the bromomethyl intermediate (Compound 2).
- Formation of the novel triphenylphosphonium salt (Compound 3) by reacting the bromide with triphenylphosphine in toluene.
- Wittig condensation of the phosphonium salt with the chiral side-chain aldehyde (Compound 4) using sodium hydride to form the alkene skeleton (Compound 5).
- Final deprotection, hydrolysis, and salt formation with calcium acetate to yield Rosuvastatin Calcium (Compound 6).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN1307187C pathway represents a strategic opportunity to optimize the cost structure and reliability of the Rosuvastatin supply chain. The shift from toxic phosphites to commodity triphenylphosphine eliminates a major safety hazard, thereby reducing insurance premiums and safety infrastructure costs associated with handling hazardous materials. Furthermore, the substantial increase in yield at the key coupling step means that less starting material is required to produce the same amount of final API, directly lowering the variable cost of goods sold (COGS). The use of robust crystallization techniques for purification, rather than complex chromatography, further drives down processing costs and cycle times.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic trimethyl phosphite removes a significant cost center related to both raw material procurement and hazardous waste disposal. By replacing it with triphenylphosphine, a widely available commodity chemical, the process leverages economies of scale. Additionally, the improved yield of 78.3% in the condensation step, compared to the industry standard of roughly 60-70%, implies a drastic reduction in raw material waste. This efficiency gain compounds through the synthesis tree, meaning fewer kilograms of precious chiral side-chain aldehyde are consumed per kilogram of final product, resulting in substantial overall cost savings without compromising quality.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel pathway, such as phosphorus tribromide, triphenylphosphine, and sodium hydride, are staple chemicals in the fine chemical industry with stable, multi-vendor supply bases. This diversification reduces the risk of supply disruptions that can occur with specialized or niche reagents. Moreover, the isolation of the phosphonium salt (Compound 3) as a stable solid intermediate allows manufacturers to build strategic inventory buffers. This decouples the upstream synthesis of the pyrimidine core from the downstream coupling reaction, providing greater flexibility in production scheduling and ensuring consistent delivery timelines for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like liquid-liquid extraction and crystallization from solvents like toluene, ethyl acetate, and ethanol. These solvents are easily recoverable and recyclable, aligning with modern sustainability goals. The avoidance of heavy metal catalysts or highly toxic phosphorus reagents simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities. This "green" profile not only ensures compliance with increasingly strict environmental regulations but also enhances the brand value of the final pharmaceutical product by appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in Patent CN1307187C, offering clarity on why this method is preferred for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the primary advantage of the phosphonium salt method over conventional phosphonate methods?
A: The phosphonium salt method avoids the use of highly toxic trimethyl phosphite required in conventional routes. Additionally, it significantly improves the condensation yield from approximately 60-70% to over 78%, reducing raw material waste and purification costs.
Q: Is the intermediate Compound (3) stable for storage and transport?
A: Yes, Compound (3), the triphenylphosphonium salt, is isolated as a stable white solid with a high yield of 81%. Its stability facilitates easier handling and inventory management compared to unstable ylide precursors.
Q: How does this process impact environmental compliance in statin manufacturing?
A: By eliminating toxic phosphite esters and utilizing standard solvents like toluene and ethanol with efficient crystallization steps, the process reduces hazardous waste generation. This simplifies wastewater treatment and aligns with stricter environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
The technical superiority of the phosphonium salt route for Rosuvastatin synthesis underscores the importance of partnering with a CDMO that possesses deep expertise in organic synthesis and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this capability, combining advanced R&D facilities with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch of intermediate meets the exacting standards required for global regulatory filings. We understand that in the competitive landscape of generic and branded statins, consistency and purity are non-negotiable.
We invite procurement leaders and technical directors to engage with our team to explore how this optimized synthesis route can be tailored to your specific volume requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield methodology. Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for securing your supply of high-purity Rosuvastatin intermediates.
