Scalable Green Synthesis of 1,3,4-Thiadiazole Intermediates Using Potassium Hydrogen Sulfate Catalysis
Introduction to Advanced 1,3,4-Thiadiazole Synthesis
The 1,3,4-thiadiazole scaffold represents a cornerstone structure in modern medicinal chemistry, serving as a critical pharmacophore in a vast array of bioactive molecules. As illustrated in the structural diversity of approved drugs, this heterocyclic system is integral to the function of diuretics like Acetazolamide, antibacterial agents such as Cefazedone, and potent kinase inhibitors like BAS0338872. 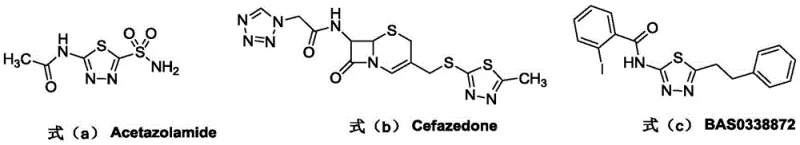 . Recognizing the immense therapeutic potential of this motif, recent intellectual property developments, specifically patent CN111995620A, have introduced a transformative preparation method that addresses long-standing synthetic challenges. This innovation leverages potassium hydrogen sulfate as a dual-function catalyst to facilitate the condensation and cyclization of thiohydrazides and amides. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology offers a pathway to high-purity compounds while drastically mitigating the environmental and safety liabilities associated with conventional manufacturing protocols.
. Recognizing the immense therapeutic potential of this motif, recent intellectual property developments, specifically patent CN111995620A, have introduced a transformative preparation method that addresses long-standing synthetic challenges. This innovation leverages potassium hydrogen sulfate as a dual-function catalyst to facilitate the condensation and cyclization of thiohydrazides and amides. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology offers a pathway to high-purity compounds while drastically mitigating the environmental and safety liabilities associated with conventional manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3,4-thiadiazole ring has relied heavily on harsh chemical environments that pose significant operational hazards and cost inefficiencies. Traditional routes often necessitate the use of aggressive dehydrating agents such as phosphorus oxychloride (POCl3), titanium tetrachloride (TiCl4), or sulfuric acid, which are not only highly corrosive to reactor vessels but also generate substantial quantities of acidic waste streams requiring neutralization. Alternatively, oxidative cyclization methods employing reagents like iodobenzene diacetate or bromine introduce toxic heavy metal residues and strong oxidants that complicate downstream purification. These legacy processes frequently suffer from poor atom economy and require rigorous safety protocols to handle volatile or corrosive reagents, ultimately inflating the cost of goods sold (COGS) and creating bottlenecks in the supply chain for high-purity 1,3,4-thiadiazole derivatives.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the methodology disclosed in CN111995620A utilizes potassium hydrogen sulfate (KHSO4) as a mild, inexpensive, and easily handled solid acid catalyst. This innovative approach enables the direct conversion of thiohydrazides and amides into the target thiadiazole core under solvent-minimized conditions, effectively bypassing the need for corrosive liquid acids or toxic oxidants. 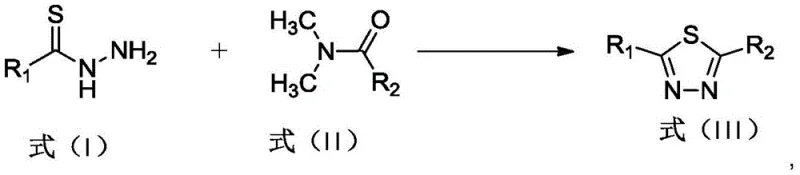 . The reaction proceeds efficiently at moderate temperatures between 90°C and 130°C, achieving yields as high as 85% with exceptional selectivity. By eliminating the reliance on dangerous reagents like Lawesson's reagent or POCl3, this novel route not only enhances operator safety but also streamlines the post-reaction workup to a simple aqueous extraction, representing a paradigm shift towards greener and more cost-effective API intermediate manufacturing.
. The reaction proceeds efficiently at moderate temperatures between 90°C and 130°C, achieving yields as high as 85% with exceptional selectivity. By eliminating the reliance on dangerous reagents like Lawesson's reagent or POCl3, this novel route not only enhances operator safety but also streamlines the post-reaction workup to a simple aqueous extraction, representing a paradigm shift towards greener and more cost-effective API intermediate manufacturing.
Mechanistic Insights into KHSO4-Catalyzed Cyclization
The efficacy of this synthesis lies in the unique dual functionality of potassium hydrogen sulfate, which acts simultaneously as a Brønsted acid catalyst and a dehydrating agent to drive the equilibrium forward. Mechanistically, the bisulfate anion facilitates the protonation of the amide carbonyl oxygen, thereby increasing the electrophilicity of the carbonyl carbon and promoting nucleophilic attack by the amino group of the thiohydrazide. This initial condensation forms a key acyl-thiohydrazide intermediate, which subsequently undergoes intramolecular cyclization. The acidic environment provided by the KHSO4 is crucial for the final dehydration step, assisting in the elimination of water to aromatize the 1,3,4-thiadiazole ring without inducing the degradation often seen with stronger mineral acids. This controlled acidity ensures that sensitive functional groups on the aromatic rings, such as methoxy or halogen substituents, remain intact throughout the transformation.
Furthermore, the choice of KHSO4 significantly impacts the impurity profile of the final product, a critical consideration for regulatory compliance in pharmaceutical applications. Unlike oxidative methods that can generate sulfone byproducts or over-oxidized species, this acid-catalyzed thermal cyclization is highly chemoselective. The moderate reaction temperature of 100°C prevents thermal decomposition of the heterocyclic core, while the solid nature of the catalyst minimizes localized hot spots that could lead to charring or polymerization. Consequently, the crude reaction mixture contains fewer side products, reducing the burden on purification units and ensuring that the resulting high-purity 1,3,4-thiadiazole intermediates meet stringent quality specifications required for downstream drug synthesis. 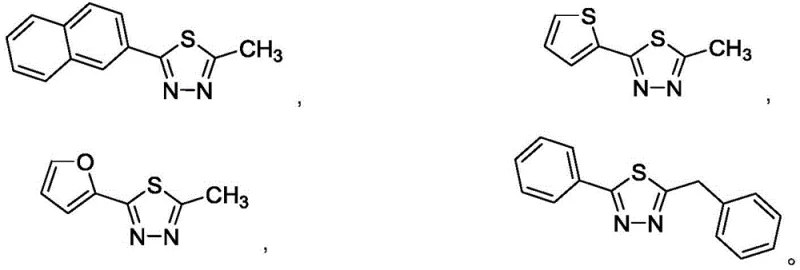 .
.
How to Synthesize 1,3,4-Thiadiazole Efficiently
To implement this robust synthetic strategy in a laboratory or pilot plant setting, operators follow a streamlined protocol that emphasizes safety and reproducibility. The process begins with the precise weighing of the thiohydrazide and amide starting materials, which are then mixed with the potassium hydrogen sulfate catalyst in a molar ratio optimized at 1:5 to ensure complete conversion without excess waste. The mixture is heated to the optimal temperature window, typically maintained at 100°C for 2 to 6 hours, allowing the cyclization to proceed to completion as monitored by TLC or HPLC.
- Combine thiohydrazide and amide substrates with potassium hydrogen sulfate (KHSO4) in a molar ratio of approximately 1: 5.
- Heat the reaction mixture to a temperature range of 90-130°C, optimally 100°C, and stir for 2 to 6 hours to ensure complete cyclization.
- Cool the mixture, add water, and extract the product with ethyl acetate, followed by washing and drying to isolate the high-purity thiadiazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this KHSO4-catalyzed process translates into tangible economic and logistical benefits that extend beyond mere chemical yield. By replacing expensive and hazardous reagents with a commodity chemical like potassium hydrogen sulfate, manufacturers can achieve significant cost reduction in API manufacturing through lower raw material expenditures and reduced waste disposal fees. The elimination of corrosive liquids extends the lifespan of reactor equipment, lowering capital expenditure (CAPEX) on maintenance and replacement, while the simplified aqueous workup reduces solvent consumption and processing time. These efficiencies collectively enhance the overall margin profile of the intermediate, making it a more attractive option for long-term supply contracts.
- Cost Reduction in Manufacturing: The substitution of specialized, high-cost dehydrating agents with bulk-grade potassium hydrogen sulfate drastically lowers the direct material costs associated with production. Furthermore, the avoidance of toxic heavy metals and strong oxidants eliminates the need for expensive scavenging resins or complex wastewater treatment protocols required to meet environmental discharge standards. This leaner chemical footprint allows for substantial cost savings that can be passed down the supply chain, offering competitive pricing for high-volume pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for supply chain continuity, and this method relies on reagents that are globally available and not subject to the geopolitical restrictions often placed on specialized catalysts. Potassium hydrogen sulfate is a stable, non-hazardous solid that is easy to store and transport, reducing the risk of supply disruptions due to regulatory changes or logistics issues. Additionally, the robustness of the reaction conditions means that production schedules are less likely to be delayed by safety incidents or equipment failures caused by corrosive chemicals, ensuring a consistent and reliable flow of materials to downstream customers.
- Scalability and Environmental Compliance: The transition from batch to commercial scale-up of complex heterocycles is often hindered by heat transfer issues and safety concerns with exothermic reagents; however, this thermal cyclization process is inherently safer and easier to control on a large scale. The absence of volatile solvents and corrosive gases simplifies the engineering controls required for containment, facilitating faster regulatory approval for new manufacturing lines. Moreover, the green chemistry principles embedded in this route align with increasingly strict global environmental regulations, future-proofing the supply chain against tightening emissions and waste disposal mandates.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and claims within the patent literature. These answers provide clarity on reaction parameters, substrate scope, and purification strategies to assist technical teams in evaluating the feasibility of this method for their specific projects.
Q: What are the primary advantages of using KHSO4 over traditional dehydrating agents like POCl3?
A: Potassium hydrogen sulfate (KHSO4) acts as a mild, solid acid catalyst that eliminates the need for highly corrosive liquids like phosphorus oxychloride (POCl3) or hazardous reagents like Lawesson's reagent. This significantly reduces equipment corrosion risks, simplifies waste treatment, and improves operator safety during industrial scale-up.
Q: What is the typical yield and purity profile for this synthesis method?
A: According to patent CN111995620A, this method achieves yields up to 85% with high purity. The mild reaction conditions minimize side reactions and decomposition, resulting in a cleaner crude product that requires less intensive purification compared to traditional oxidative cyclization methods.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process is highly suitable for commercial scale-up. It utilizes cheap, readily available raw materials, operates at moderate temperatures (90-130°C), and features a simple aqueous workup. These factors collectively lower production costs and enhance supply chain stability for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4-Thiadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of drug development and commercialization. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and compliant. We are committed to delivering high-purity 1,3,4-thiadiazole derivatives that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against customer requirements.
We invite global partners to engage with our technical procurement team to discuss how this advanced KHSO4-catalyzed technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective chemical solutions available in the market.
