Advanced Synthetic Route for High-Purity Atorvastatin Calcium Intermediates via Organocatalysis
Introduction to Advanced Statin Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and safe methodologies for producing key active pharmaceutical ingredient (API) precursors. Patent CN103145584A introduces a significant technological breakthrough in the synthesis of (3R,5R)-3,5-dihydroxy-6-cyanohexanoate, a pivotal chiral building block for the blockbuster drug Atorvastatin Calcium. This compound serves as the core structural motif for HMG-CoA reductase inhibitors, which dominate the cardiovascular therapeutic market. The disclosed methodology represents a paradigm shift from traditional enzymatic or heavy metal-catalyzed processes towards a more chemically elegant organocatalytic approach. By leveraging inexpensive starting materials derived from citric acid and employing sophisticated asymmetric induction strategies, this route addresses critical pain points in modern drug manufacturing, including toxicity, cost, and stereocontrol. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital statin side chain has been plagued by significant safety and operational challenges. Prior art, such as World Patent WO 2003053950 and US Patent 5155251A, often relies on the use of highly toxic cyanide salts like potassium cyanide (KCN) or sodium cyanide (NaCN) for ring-opening reactions of epoxides. These reagents pose severe occupational health risks, requiring specialized containment equipment and rigorous labor protection protocols that drastically inflate operational expenditures. Furthermore, enzymatic routes utilizing deoxyribose phosphate aldolase (DERA), while selective, suffer from the difficulty in sourcing stable, high-activity enzymes at a commercial scale. The reliance on biological catalysts often introduces variability in batch-to-batch consistency and limits the reaction concentration due to solubility issues in aqueous media. Additionally, many conventional routes involve harsh reaction conditions, extreme temperatures, or complex protection-deprotection sequences that lower overall throughput and generate substantial chemical waste, hindering cost reduction in API manufacturing.
The Novel Approach
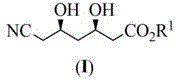
In stark contrast, the methodology outlined in CN103145584A offers a streamlined, chemically robust alternative that circumvents these legacy issues. The process initiates with the benzylation of diethyl 3-hydroxyglutarate using benzyl trichloroacetimide, a reagent that allows for mild acidic catalysis rather than harsh basic conditions. A key innovation lies in the formation of a 3-benzyloxycyclopentanoic anhydride intermediate, which serves as a versatile platform for stereochemical induction. Instead of toxic cyanide ring openings, the nitrile functionality is introduced later via a condensation reaction with cyanoacetate, a much safer and commercially abundant reagent. The route employs a Krapcho deesterification strategy to selectively remove ester groups without affecting the sensitive nitrile moiety. This logical disconnection of the carbon skeleton allows for milder reaction parameters, typically ranging from -20°C to 50°C, which are far more manageable in standard stainless steel reactors compared to cryogenic or high-pressure systems. This strategic redesign facilitates the commercial scale-up of complex pharmaceutical intermediates by simplifying unit operations and enhancing process safety.
Mechanistic Insights into Quinine Sulfonamide-Catalyzed Asymmetric Alcoholysis
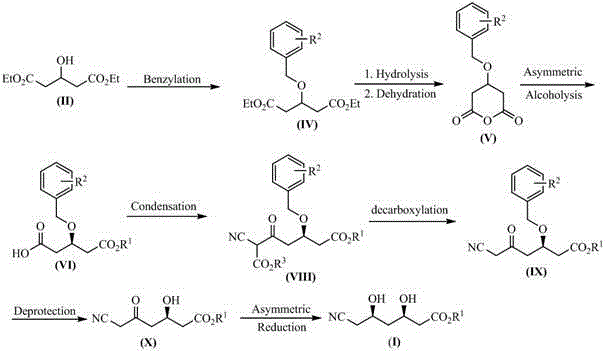
The cornerstone of this synthesis is the establishment of the first chiral center through an asymmetric alcoholysis reaction. The process utilizes a quinine sulfonamide catalyst (A), specifically derivatives where R4 can be a 3,5-bis(trifluoromethyl)phenyl group, to desymmetrize the meso-like anhydride intermediate. Mechanistically, the sulfonamide moiety acts as a bifunctional organocatalyst, simultaneously activating the nucleophilic alcohol and the electrophilic anhydride through hydrogen bonding networks. This precise spatial arrangement ensures that the nucleophilic attack occurs exclusively on one face of the anhydride ring, yielding the (R)-3-benzyloxy-5-alkoxy-5-oxopentanoic acid with high enantiomeric excess. The patent highlights that catalyst loading can be kept low (0.01 to 0.5 equivalents) while maintaining quantitative recovery, which is a critical factor for process economics. Following this, the second stereocenter is installed via an asymmetric reduction of the beta-keto ester using borohydrides in the presence of chiral control agents like methoxydiethoxyborane. This dual-catalytic strategy ensures that the final product possesses the requisite (3R,5R) configuration with diastereoselectivity exceeding 90%, eliminating the need for costly and yield-eroding chiral chromatography purification steps.
How to Synthesize (3R,5R)-3,5-dihydroxy-6-cyanohexanoate Efficiently
Executing this synthesis requires careful attention to reaction stoichiometry and solvent selection to maximize yield and optical purity. The protocol begins with the protection of the hydroxyl group in diethyl 3-hydroxyglutarate, followed by cyclization to the anhydride using acetyl chloride or thionyl chloride. The critical asymmetric alcoholysis step is best performed in ethers like methyl tert-butyl ether (MTBE) at 0°C to optimize enantioselectivity. Subsequent condensation with methyl cyanoacetate utilizes thionyl chloride as an activator, forming the beta-keto nitrile scaffold. The final stages involve Krapcho decarboxylation using lithium or sodium chloride in polar aprotic solvents like DMF, followed by catalytic hydrogenolysis for debenzylation. Each step has been optimized in the patent examples to demonstrate reproducibility, with isolated yields frequently exceeding 75% per step. For detailed operational parameters, temperature profiles, and workup procedures, please refer to the standardized guide below.
- Benzylation of diethyl 3-hydroxyglutarate using benzyl trichloroacetimide followed by hydrolysis and dehydration to form 3-benzyloxycyclopentanoic anhydride.
- Asymmetric alcoholysis of the anhydride using a quinine sulfonamide catalyst to establish the (R)-stereocenter, followed by condensation with cyanoacetate.
- Krapcho deesterification, debenzylation, and final asymmetric reduction using borohydrides with chiral control agents to yield the target diol.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this patented route offers distinct advantages that translate directly into bottom-line value and risk mitigation. The shift away from regulated, high-toxicity cyanide salts to benign cyanoacetates simplifies regulatory compliance and reduces the burden on environmental health and safety (EHS) departments. This change alone can lead to substantial cost savings by eliminating the need for specialized hazardous waste disposal contracts and reducing insurance premiums associated with handling acute toxins. Furthermore, the raw materials, such as citric acid derivatives and common organic solvents like dichloromethane and ethyl acetate, are commodity chemicals with stable global supply chains, ensuring reducing lead time for high-purity pharmaceutical intermediates. The ability to recover and recycle the expensive quinine-based catalyst adds another layer of economic efficiency, minimizing the consumption of precious chiral pool resources. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a greener manufacturing footprint that aligns with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive enzymatic catalysts and the use of organocatalysts that can be quantitatively recovered significantly lowers the cost of goods sold (COGS). Additionally, the avoidance of cryogenic conditions reduces utility costs, while the high atom economy of the condensation and deesterification steps minimizes raw material waste. The process utilizes cheap alkali metal halides for decarboxylation instead of noble metals, further driving down input costs without compromising quality.
- Enhanced Supply Chain Reliability: By relying on broadly available chemical reagents rather than niche biocatalysts or tightly controlled poisons, manufacturers can diversify their supplier base and mitigate disruption risks. The robustness of the chemical steps means that production schedules are less susceptible to the variability often seen in fermentation-based processes. This stability ensures a consistent flow of materials to downstream API synthesis sites, supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: The synthetic pathway is designed with industrial scalability in mind, utilizing unit operations like extraction, distillation, and crystallization that are standard in multipurpose chemical plants. The reduced toxicity profile simplifies effluent treatment, allowing facilities to meet stringent environmental discharge standards more easily. This compliance readiness accelerates technology transfer from pilot plant to full commercial production, enabling rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific production needs. It clarifies the safety profile, the scalability potential, and the quality attributes of the final intermediate.
Q: How does this method improve safety compared to traditional cyanide routes?
A: Unlike conventional methods utilizing highly toxic potassium cyanide or sodium cyanide for ring-opening reactions, this patent employs cyanoacetate condensation and avoids direct handling of free cyanide salts, significantly reducing occupational hazards and waste treatment complexity.
Q: What is the stereochemical purity achievable with this organocatalytic route?
A: The process utilizes quinine sulfonamide catalysts for asymmetric alcoholysis and chiral borane control agents for reduction, achieving diastereoselectivity higher than 90% and ensuring the critical (3R,5R) configuration required for biological activity.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (ranging from -20°C to 50°C), utilize cheap and readily available raw materials like citric acid derivatives, and employ recyclable solvents, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3R,5R)-3,5-dihydroxy-6-cyanohexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of cardiovascular therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of (3R,5R)-3,5-dihydroxy-6-cyanohexanoate meets the exacting standards required for GMP API synthesis. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this advanced organocatalytic route, delivering products with consistent stereochemical integrity.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the competitive statin market.
