Advanced Synthesis of Pyridine Derivatives for Scalable Herbicide Intermediate Production
Advanced Synthesis of Pyridine Derivatives for Scalable Herbicide Intermediate Production
The global agrochemical industry continuously seeks robust manufacturing pathways that balance high yield with operational safety, particularly for complex heterocyclic scaffolds. Patent CN1199948C introduces a transformative methodology for synthesizing novel pyridine derivatives, specifically designed as critical intermediates for fluoroalkylpyridine sulfonylurea herbicides. This intellectual property details the preparation of compounds falling under general formula (1), which serve as precursors to the highly active herbicidal agents defined in formula (2). The significance of this patent lies not merely in the new chemical entities themselves, but in the fundamental shift away from hazardous cryogenic chemistry toward a more manageable copper-mediated halogenation process. For R&D directors and procurement strategists, this represents a pivotal opportunity to re-evaluate supply chains for key agrochemical building blocks. By leveraging this technology, manufacturers can achieve reliable production of high-purity agrochemical intermediates while mitigating the risks associated with traditional organometallic reagents.
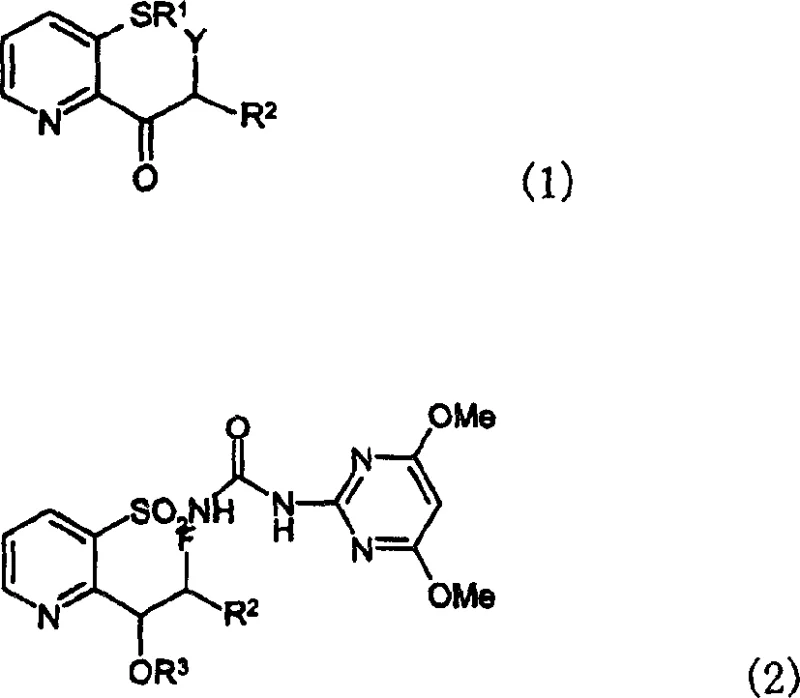
The structural versatility of these pyridine derivatives allows for extensive customization of the final herbicidal product. As illustrated in the patent documentation, the R1 group can vary significantly, encompassing hydrogen, cyano, alkyl, cycloalkyl, or various substituted benzyl groups, providing a broad chemical space for optimization. Furthermore, the ability to introduce specific halogen atoms at the alpha-position of the ketone side chain via the Y substituent is crucial for subsequent functionalization. This flexibility ensures that the synthetic route remains applicable across a wide portfolio of sulfonylurea herbicides, making it a valuable asset for any reliable agrochemical intermediate supplier looking to diversify their catalog with high-margin specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO 92/14728 and related documents, rely heavily on the use of n-butyllithium (n-BuLi) to introduce carbon-carbon bonds at the 2-position of the pyridine ring. This conventional approach imposes severe operational constraints, necessitating reaction temperatures below -70°C and strictly anhydrous conditions to prevent reagent decomposition and safety incidents. The handling of large quantities of pyrophoric organolithium reagents requires specialized cryogenic equipment and rigorous safety protocols, which significantly inflate capital expenditure (CAPEX) and operating expenses (OPEX). Moreover, the sensitivity of these reactions often leads to batch-to-batch variability, complicating quality control efforts and potentially delaying time-to-market for new herbicide formulations. The inherent instability of the intermediates generated under such extreme conditions also poses challenges for storage and transport, limiting the geographical reach of suppliers who rely on these legacy processes.
The Novel Approach
In stark contrast, the methodology presented in CN1199948C utilizes copper(II) salts, such as copper(II) chloride or copper(II) bromide, to effect alpha-halogenation under remarkably mild conditions. This novel approach eliminates the need for cryogenic cooling, allowing reactions to proceed efficiently at temperatures ranging from -20°C to 150°C, with a preferred window of 10°C to 80°C. By replacing hazardous organolithiums with stable, inexpensive inorganic salts, the process dramatically simplifies the engineering requirements for commercial-scale reactors. This shift facilitates cost reduction in herbicide intermediate manufacturing by reducing energy consumption for cooling and minimizing the need for specialized containment systems. Additionally, the tolerance for a wider range of solvents, including common chlorinated hydrocarbons and aromatic solvents, enhances the adaptability of the process to existing manufacturing infrastructure, thereby accelerating the commercial scale-up of complex agrochemical intermediates.
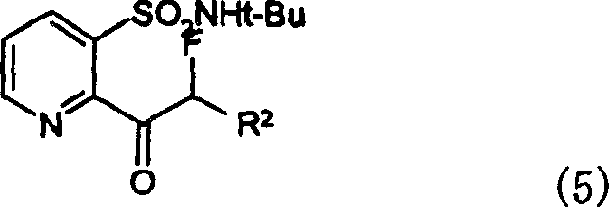
Mechanistic Insights into Copper-Mediated Alpha-Halogenation
The core innovation of this patent resides in the mechanistic pathway for converting the starting pyridone ketone (Formula 6) into the halogenated intermediate (Formula 1c). The reaction involves the interaction of the enolizable ketone with a copper(II) species, which acts as both an oxidant and a source of the halogen atom. Unlike radical halogenation methods that often suffer from poor regioselectivity and over-halogenation, the copper-mediated process offers superior control over the substitution pattern. The mechanism likely proceeds through the formation of a copper-enolate complex, followed by a single-electron transfer or reductive elimination step that installs the halogen atom at the alpha-position with high fidelity. This precision is vital for maintaining the integrity of the sensitive pyridine ring and the thioether substituents, which might otherwise be degraded under harsher oxidative conditions. The ability to selectively halogenate the side chain without affecting the heterocyclic core ensures that the downstream conversion to the sulfonamide (Formula 5) proceeds with minimal impurity formation.
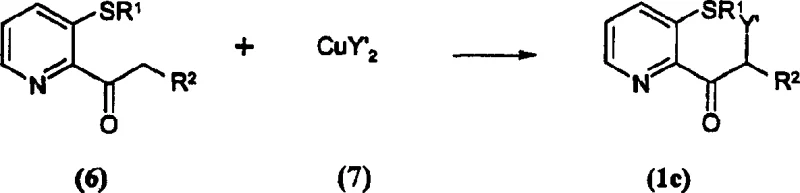
Furthermore, the patent details a subsequent nucleophilic substitution step where the alpha-halo ketone (Formula 1c) is converted to the alpha-fluoro ketone (Formula 1b) using alkali metal fluorides. This transformation is facilitated by the use of phase transfer catalysts or polar solvents like polyethylene glycol, which enhance the solubility and reactivity of the fluoride ion. The mechanistic understanding of this substitution is critical for impurity control, as incomplete conversion or competing elimination reactions could lead to difficult-to-remove byproducts. By optimizing the stoichiometry of the fluoride source and the reaction temperature, manufacturers can drive the equilibrium towards the desired fluoro-product, ensuring high-purity agrochemical intermediates that meet stringent regulatory specifications. This two-step sequence—halogenation followed by fluorination—provides a modular and robust platform for introducing diverse functional groups, thereby expanding the chemical diversity available for herbicide discovery and development.
How to Synthesize 1-(3-Substituted-Pyridin-2-yl)-2-Halopropan-1-one Efficiently
The practical implementation of this synthetic route involves straightforward unit operations that are familiar to most fine chemical manufacturing facilities. The process begins with the dissolution of the starting ketone in a suitable organic solvent, followed by the controlled addition of the copper salt. Reaction progress is typically monitored via HPLC or TLC until the starting material is fully consumed. Upon completion, the reaction mixture is quenched with water, and the product is extracted into the organic phase. Standard purification techniques, such as silica gel chromatography or crystallization, are then employed to isolate the target halogenated intermediate. For the subsequent fluorination step, the isolated halide is reacted with a fluoride salt in a polar medium, followed by aqueous workup to remove inorganic salts. Detailed standardized synthesis steps are provided below to guide process engineers in adapting this technology to their specific production environments.
- React the starting pyridone compound (Formula 6) with 2 to 3 equivalents of a copper(II) salt (CuCl2 or CuBr2) in a suitable solvent like toluene or dichloromethane at temperatures between 10°C and 80°C.
- Isolate the resulting alpha-halogenated ketone intermediate (Formula 1c) through standard aqueous workup and purification techniques such as silica gel chromatography.
- Perform a nucleophilic substitution reaction on the halogenated intermediate using an alkali metal fluoride (e.g., KF) in a polar solvent like polyethylene glycol to yield the final fluoro-compound (Formula 1b).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-mediated synthesis offers profound advantages for procurement managers and supply chain leaders tasked with optimizing costs and ensuring continuity. The elimination of cryogenic conditions and pyrophoric reagents directly translates to reduced operational complexity and lower insurance premiums associated with hazardous chemical handling. Furthermore, the use of commodity chemicals like copper chloride and bromide, which are abundantly available globally, mitigates the risk of supply disruptions often associated with specialized organometallic reagents. This stability in raw material sourcing is crucial for maintaining consistent production schedules and meeting the just-in-time delivery expectations of major agrochemical companies. By integrating this technology, suppliers can offer more competitive pricing structures while maintaining healthy margins, driven by the inherent efficiency and safety of the process.
- Cost Reduction in Manufacturing: The transition from n-butyllithium to copper salts removes the necessity for expensive cryogenic cooling systems and specialized inert atmosphere handling equipment. This significant reduction in utility costs and capital investment allows for a leaner manufacturing model. Additionally, the higher yields and cleaner reaction profiles observed in the examples reduce the burden on downstream purification processes, further lowering the cost of goods sold (COGS). The overall economic efficiency makes this route highly attractive for large-volume production campaigns.
- Enhanced Supply Chain Reliability: Relying on stable, shelf-stable inorganic salts rather than moisture-sensitive organolithiums enhances the resilience of the supply chain. Raw materials can be sourced from multiple vendors without compromising quality, reducing dependency on single-source suppliers. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment maintenance or environmental fluctuations, ensuring a steady flow of intermediates to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions facilitate easier scale-up from pilot plant to commercial tonnage without the exponential increase in safety risks seen with exothermic organolithium reactions. The process generates less hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations. This compliance advantage reduces the administrative burden of waste disposal and permits, streamlining the path to commercial authorization for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN1199948C, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific product portfolios. The answers reflect the balance between chemical innovation and practical manufacturing realities.
Q: Why is the copper-mediated method preferred over traditional n-BuLi routes?
A: Traditional methods require n-butyllithium at cryogenic temperatures below -70°C under extreme anhydrous conditions, posing significant safety hazards and high operational costs. The novel copper-mediated approach operates at mild temperatures (-20°C to 150°C) using stable inorganic salts, drastically improving process safety and scalability.
Q: What solvents are compatible with this halogenation process?
A: The process demonstrates high flexibility regarding solvent choice. Preferred solvents include chlorinated hydrocarbons like dichloromethane and chloroform, aromatic hydrocarbons such as toluene and xylene, and various esters or ethers, allowing manufacturers to optimize based on existing infrastructure.
Q: How does this process impact the purity of the final herbicide intermediate?
A: By avoiding the highly reactive and sensitive organolithium reagents, the new method minimizes side reactions such as amide dehydrogenation. This results in a cleaner reaction profile and higher purity of the core pyridone intermediate (Formula 5), which is critical for downstream herbicide efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like those described in CN1199948C for securing a competitive edge in the agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of pyridine derivatives meets the exacting standards required for herbicide synthesis. Our infrastructure is designed to handle complex heterocyclic chemistry safely and efficiently, making us an ideal partner for long-term supply agreements.
We invite you to engage with our technical procurement team to discuss how this novel copper-mediated route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this safer, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more sustainable and cost-effective manufacturing future.
