Advanced Manufacturing of High-Purity Orlistat Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Orlistat Intermediates for Global Pharmaceutical Supply Chains
The global demand for anti-obesity therapeutics continues to surge, placing immense pressure on the supply chains responsible for producing critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN111518131A introduces a groundbreaking preparation method for a high-purity Orlistat intermediate, addressing long-standing inefficiencies in yield and environmental impact that have plagued traditional synthesis routes. This technological advancement is particularly relevant for manufacturers seeking to optimize their production of gastrointestinal lipase inhibitors, where the structural integrity and purity of the chiral side chain are paramount. By refining reaction parameters and implementing a sophisticated purification regime involving alternating polar and non-polar solvent extractions, this method achieves exceptional recovery rates suitable for large-scale industrial application. 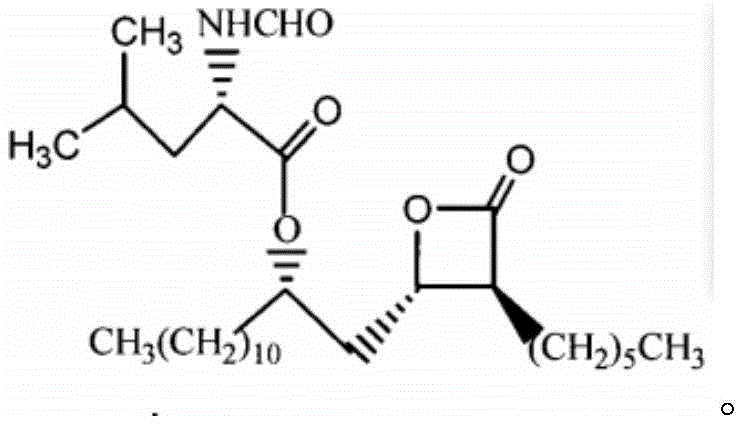
For R&D directors and process chemists, the significance of this patent lies in its ability to consistently deliver intermediates with purity levels exceeding 99.6%, a benchmark that drastically reduces the burden on downstream purification processes. The synthesis targets the specific chiral building block required for Orlistat, a molecule known for its competitive inhibition of dietary fat absorption. Traditional methods often struggle with the removal of trace impurities that can affect the stability of the final beta-lactone ring or the stereochemical purity of the product. This new approach mitigates those risks through a carefully controlled sequence of protection, reduction, and crystallization steps, ensuring that the final intermediate meets the stringent quality standards required by top-tier pharmaceutical regulators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Orlistat intermediates have been fraught with challenges related to solvent management and product isolation. In many legacy processes, the post-treatment phase involves complex workups where solvents are difficult to recover, leading to significant material loss and increased operational expenditures. Furthermore, the crude products generated from these older methods typically contain a heterogeneous mixture of polar and non-polar impurities that are notoriously difficult to separate using standard washing techniques. This impurity profile not only depresses the overall yield but also necessitates additional, costly purification steps such as column chromatography, which are impractical for multi-ton commercial production. The accumulation of waste solvents and the inability to efficiently recycle reagents have made these conventional pathways economically unsustainable in the current regulatory environment.
The Novel Approach
The methodology disclosed in CN111518131A represents a paradigm shift by integrating a highly efficient solvent recovery system directly into the reaction workflow. The process begins with a hydroxyl protection step followed by a specialized reduction, but the true innovation lies in the subsequent purification strategy. By employing a mixed solvent system of polar and non-polar components—such as dilute acetic acid paired with petroleum ether or heptane—the method effectively partitions impurities away from the desired product. 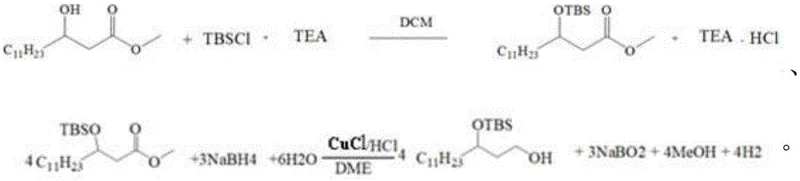
This novel approach allows for the distillation and recycling of non-polar solvents at normal pressure, significantly lowering the environmental footprint. The final recrystallization step, conducted at low temperatures using medium-polarity solvents like acetonitrile or ethyl acetate mixed with non-polar hydrocarbons, ensures the removal of residual by-products. This results in a light yellow liquid intermediate (Compound VI) that possesses both high chemical purity and excellent stereochemical integrity, ready for the final coupling reaction to form Orlistat without further extensive processing.
Mechanistic Insights into CuCl-Catalyzed Reduction and Silyl Protection
The core of this synthesis relies on two critical chemical transformations: the silyl protection of the hydroxyl group and the subsequent reduction of the ester functionality. The initial step involves reacting R-3-hydroxytetradecanoic acid methyl ester (Compound I) with tert-butyldimethylsilyl chloride (TBSCl) in the presence of triethylamine (TEA). This protection is essential to prevent unwanted side reactions at the hydroxyl site during the subsequent reduction phase. The use of dichloromethane (DCM) as the solvent facilitates a homogeneous reaction environment, and the precise control of reaction time (15.5 to 16.5 hours) ensures complete conversion while minimizing the formation of over-silylated by-products. 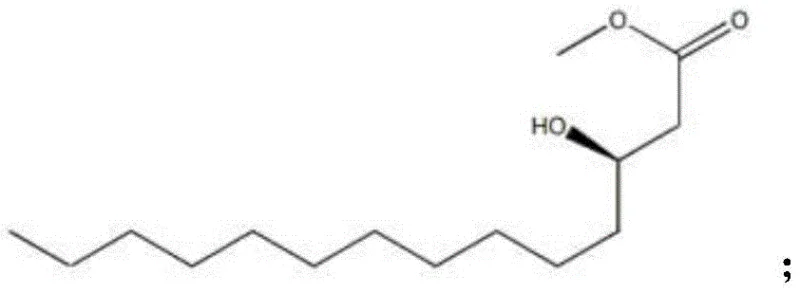
Following protection, the intermediate (Compound IV) undergoes a reduction reaction utilizing cuprous chloride (CuCl) and sodium borohydride (NaBH4) in 1,2-dimethoxyethane (DME). This specific catalytic system is chosen for its ability to selectively reduce the ester group while maintaining the stability of the silyl ether protecting group. The reaction is conducted under a nitrogen atmosphere to prevent oxidation of the copper catalyst and the sensitive intermediate species. The mechanistic pathway likely involves the formation of a copper hydride species which transfers hydride to the carbonyl carbon. The subsequent workup, involving quenching with dilute hydrochloric acid and extraction, is designed to neutralize boron by-products and recover the expensive amine base, further enhancing the economic viability of the process.
How to Synthesize High-Purity Orlistat Intermediate Efficiently
The synthesis of Compound VI is a streamlined process that balances reaction kinetics with practical isolation techniques to maximize throughput. The procedure starts with the protection of the starting alcohol, followed by a rigorous extraction protocol that recovers the amine base for reuse. The reduction step is then performed under strictly anhydrous conditions to ensure high conversion. Finally, the product is isolated through a series of distillations and a low-temperature recrystallization that acts as the final polishing step. For detailed operational parameters, including specific temperature ramps, vacuum levels, and solvent ratios, please refer to the standardized synthesis guide below which outlines the critical control points for GMP manufacturing.
- Perform hydroxyl protection on R-3-hydroxytetradecanoic acid methyl ester using TBSCl and TEA in DCM, followed by solvent recovery and extraction.
- Conduct a reduction reaction on the protected intermediate (Compound IV) using CuCl and NaBH4 in DME under nitrogen atmosphere.
- Execute a multi-stage purification involving extraction with polar/non-polar solvent mixtures, distillation, and low-temperature recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical performance. The primary advantage lies in the drastic simplification of the solvent recovery loop. Unlike traditional methods where solvents are often lost to aqueous waste streams, this process enables the distillation and reuse of non-polar solvents like petroleum ether and heptane. This closed-loop capability translates directly into substantial cost savings on raw materials and significantly reduces the volume of hazardous waste requiring disposal, thereby lowering compliance costs associated with environmental regulations.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps and the ability to recycle key reagents like triethylamine and solvents create a leaner cost structure. By achieving yields approaching 98% and purities near 99.8%, the process minimizes the loss of expensive chiral starting materials. This efficiency means that less feedstock is required per kilogram of finished intermediate, directly improving the gross margin profile for the final API. Furthermore, the use of commodity chemicals like cuprous chloride and sodium borohydride avoids the reliance on exotic, high-cost catalysts that can introduce supply volatility.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted API production schedules. The method's tolerance for standard industrial equipment and its reliance on widely available solvents reduce the risk of supply disruptions caused by niche reagent shortages. Additionally, the simplified workup procedure shortens the overall cycle time per batch, allowing manufacturing facilities to increase throughput and respond more agilely to fluctuations in market demand for weight-loss therapeutics.
- Scalability and Environmental Compliance: Designed with industrial scale-up in mind, the process avoids exothermic runaways and utilizes mild reaction conditions that are easily managed in large reactors. The effective removal of impurities through solvent extraction rather than energy-intensive distillation of the product itself preserves the thermal stability of the intermediate. This approach aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product), making it an attractive option for companies aiming to meet corporate sustainability goals and stricter environmental discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this technology into their supply chain.
Q: How does this new method improve upon conventional Orlistat intermediate synthesis?
A: The patented method significantly enhances yield (up to 97.78%) and purity (up to 99.77%) by optimizing solvent systems for better recovery and implementing a rigorous recrystallization protocol that removes both polar and non-polar impurities effectively.
Q: What are the key reagents used in the reduction step of this process?
A: The reduction step utilizes Cuprous Chloride (CuCl) and Sodium Borohydride (NaBH4) in 1,2-dimethoxyethane (DME) under a nitrogen atmosphere, ensuring controlled reaction conditions and high conversion rates.
Q: Is the solvent system in this process environmentally sustainable?
A: Yes, the process is designed for industrial sustainability by allowing the recycling of solvents like DCM, petroleum ether, and heptane through distillation, thereby reducing waste generation and overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Orlistat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Orlistat depends on a secure and high-quality supply of intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global clinical and commercial programs. We are committed to delivering materials that adhere to stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards before shipment.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience and drive down your total cost of ownership.
