Advanced Orlistat Intermediate Synthesis Pathway Enables Cost-Efficient Commercial Scale-Up for Global Pharma Supply Chains
The patent CN108484536B introduces a groundbreaking 7-step linear synthesis route for the key intermediate of orlistat, a globally recognized anti-obesity drug. This innovative methodology utilizes n-dodecanal as the starting material to achieve high product yield through mild reaction conditions and simplified operational procedures. By reducing synthetic steps compared to conventional approaches, this process delivers significant cost reduction in API manufacturing while ensuring high-purity output essential for pharmaceutical applications. As a reliable API intermediate supplier, this advancement addresses critical pain points for R&D directors seeking purity control, procurement managers focused on cost efficiency, and supply chain heads requiring consistent delivery timelines.
Overcoming Traditional Synthesis Limitations for Orlistat Intermediate
The Limitations of Conventional Methods
Traditional synthesis routes for orlistat intermediates documented in prior literature suffer from multiple critical drawbacks including extended step counts exceeding ten steps, complex reaction conditions requiring specialized equipment, and the use of expensive or toxic reagents. These factors collectively contribute to low overall yields due to cumulative losses at each transformation stage and increased impurity profiles that complicate purification processes. The operational complexity often leads to inconsistent batch quality and extended lead times as multiple protection/deprotection strategies require additional validation steps. Furthermore, reliance on chiral resolution techniques introduces variability that impacts scalability and reproducibility across different manufacturing sites, creating significant challenges for global supply chain management in pharmaceutical production.
The Novel Approach
The patented methodology (CN108484536B) presents a streamlined alternative through its elegant 7-step linear sequence that eliminates unnecessary synthetic detours while maintaining high stereochemical control from n-dodecanal to compound (9). Starting with Evans aldol chemistry under titanium tetrachloride mediation at mild temperatures (-78°C), the process establishes critical chiral centers with high diastereoselectivity without requiring cryogenic equipment beyond standard laboratory capabilities. Subsequent nucleophilic substitution using monomethyl malonate potassium salt under magnesium chloride catalysis proceeds with excellent regiocontrol at ambient temperatures, minimizing unwanted side reactions that could compromise purity profiles. The integrated design ensures consistent high purity throughout the synthesis without requiring extensive purification steps between transformations.
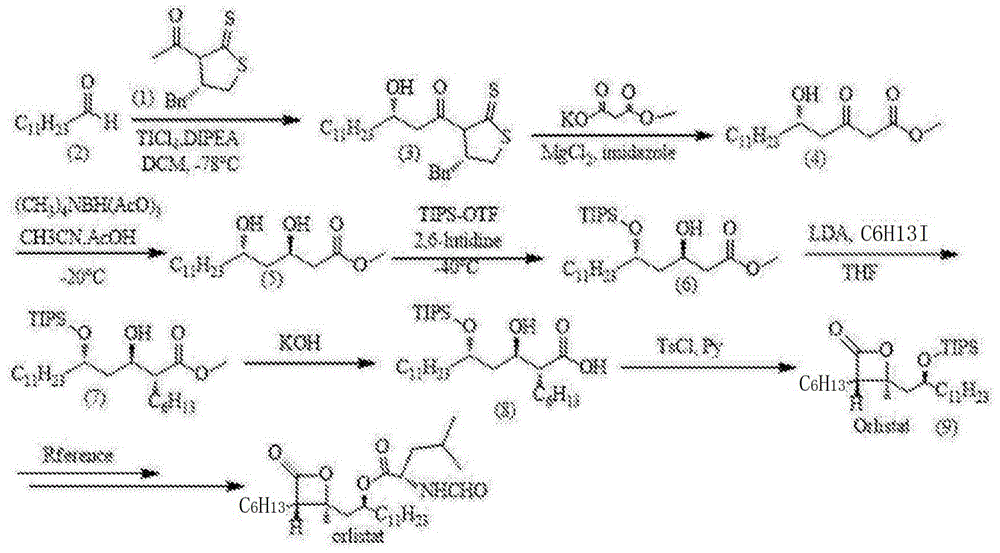
Precision Chemistry for High-Purity Orlistat Intermediate
The synthetic pathway demonstrates exceptional control over stereochemistry through carefully optimized Evans aldol methodology where titanium tetrachloride-mediated reaction with N-dodecanal establishes the critical chiral center with high diastereoselectivity as evidenced by the patent's experimental section showing consistent product formation across multiple batches. The subsequent nucleophilic substitution using monomethyl malonate potassium salt under magnesium chloride catalysis proceeds with excellent regiocontrol at room temperature conditions that prevent epimerization while minimizing unwanted side products that could compromise purity specifications required by regulatory authorities. Trans-reduction with tetramethyltriacetoxyborohydride ammonium achieves selective formation of the desired diol configuration without racemization through controlled low-temperature processing at -20°C as specified in the patent examples.
Impurity profile management is systematically addressed through strategic reaction design that minimizes side reactions at each stage by leveraging selective protecting groups like triisopropylsilyltriflate which prevent unwanted reactions at sensitive functional sites during alkylation steps. The choice of mild reducing agents avoids over-reduction pathways that could introduce ketone impurities while maintaining functional group tolerance throughout the sequence as demonstrated in the patent's detailed experimental procedures showing clean reaction progress by TLC monitoring. The absence of transition metal catalysts eliminates concerns about heavy metal contamination entirely which is critical for pharmaceutical intermediates requiring strict ICH Q3D compliance standards.
Supply Chain and Cost Advantages of the Optimized Process
This innovative synthesis route directly addresses three major pain points in pharmaceutical manufacturing supply chains by reducing synthetic complexity while enhancing operational efficiency across procurement, production, and logistics functions. The elimination of multiple synthetic steps removes associated purification requirements and intermediate storage needs that typically create bottlenecks in production workflows while improving raw material availability through common laboratory reagents instead of specialized catalysts or exotic materials. These fundamental improvements translate into tangible benefits without requiring substantial capital investment in new equipment or facilities as all transformations utilize standard manufacturing infrastructure available at most fine chemical production sites globally.
- Cost Reduction in API Manufacturing: The elimination of multiple synthetic steps directly reduces raw material consumption through fewer transformation requirements while avoiding expensive chiral resolution procedures saves significant processing costs associated with specialized equipment validation and operator training. Simplified purification requirements decrease solvent usage and waste disposal expenses by eliminating multiple chromatographic separations required in conventional routes as evidenced by the patent's streamlined workup procedures described in examples one through seven. The use of standard reagents instead of specialized catalysts reduces material costs substantially compared to conventional routes while maintaining consistent quality standards required for pharmaceutical applications without additional quality control testing layers.
- Reducing Lead Time for High-Purity Intermediates: The shortened synthetic sequence reduces manufacturing cycle time by eliminating non-value-added steps that typically require additional validation periods between transformations as described in the patent's background section regarding conventional methods' limitations. Simplified process flow enables faster batch turnover through existing manufacturing equipment without requiring additional cleaning validation cycles between different unit operations which is particularly valuable during peak demand periods when procurement teams face urgent supply requests from global manufacturing sites.
- Commercial Scale-Up of Complex Intermediates: The mild reaction conditions including standard temperature ranges (-78°C to room temperature) and atmospheric pressure operations make this process inherently scalable from laboratory to commercial production volumes without major engineering modifications as demonstrated by the patent's detailed experimental procedures using standard glassware that translates directly to pilot plant equipment. The robust nature of each transformation ensures consistent product quality across different batch sizes due to well-defined critical process parameters documented throughout the patent description which minimizes scale-up risks typically associated with complex multi-step syntheses requiring specialized reactor configurations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN108484536B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
