Scalable Manufacturing of 5-Azaindole: A Cost-Effective Route for Kinase Inhibitor Intermediates
Scalable Manufacturing of 5-Azaindole: A Cost-Effective Route for Kinase Inhibitor Intermediates
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed 5-azaindole at the forefront of critical heterocyclic building blocks. As disclosed in patent CN103421004A, a transformative preparation method has emerged that fundamentally alters the economic and operational landscape of producing this high-value scaffold. This innovative protocol leverages 3-methylpyridine as a readily accessible feedstock, navigating through a sophisticated five-step sequence that includes oxidation, nitration, deoxygenation, enamine formation, and finally, a metal-catalyzed reductive cyclization. Unlike legacy processes that struggle with thermal instability or hazardous reagents, this approach prioritizes operational safety and industrial feasibility without compromising on chemical integrity. For R&D directors and procurement strategists alike, understanding this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of modern drug discovery pipelines.
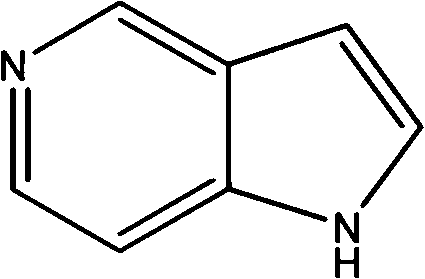
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-azaindole has been plagued by significant technical hurdles that hinder large-scale commercialization. Prior art, such as the method described in U.S. Pat 4625033, relies on the reaction of 3-methyl-4-nitropyridine N-oxide with DMF dimethyl acetal followed by hydrogenation. However, this legacy route suffers from the generation of methanol as a byproduct, which possesses a low boiling point that inadvertently lowers the reaction temperature, often resulting in incomplete conversion and difficult-to-remove impurities. Furthermore, alternative literature methods, such as those reported in Synthesis (1996), necessitate the use of n-butyllithium at cryogenic temperatures of -40°C. Such extreme conditions not only escalate energy consumption drastically but also introduce severe safety risks associated with handling pyrophoric organolithium reagents on a multi-kilogram scale. These factors collectively render traditional methodologies economically unviable and operationally fragile for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN103421004A presents a robust solution that circumvents these historical bottlenecks through intelligent process design. By initiating the synthesis with the oxidation of 3-methylpyridine to its N-oxide derivative, the process activates the pyridine ring for highly regioselective nitration at the 4-position. Subsequent deoxygenation using phosphorus trichloride restores the aromatic system while retaining the crucial nitro group, setting the stage for the key carbon-carbon bond formation. The utilization of DMF dimethyl acetal under heating conditions facilitates the formation of the vinyl intermediate without the generation of volatile methanol that plagues older routes. Finally, the reductive cyclization employs Raney nickel and hydrazine hydrate, a combination that operates effectively at moderate temperatures, thereby eliminating the need for high-pressure hydrogenation equipment or cryogenic cooling. This streamlined workflow ensures that high-purity 5-azaindole can be produced with consistent quality and superior throughput.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Reductive Closure
The core of this synthetic strategy lies in the precise manipulation of electronic effects within the pyridine ring to achieve the desired substitution pattern. The initial oxidation step converts the electron-rich pyridine nitrogen into an N-oxide, which dramatically alters the electron density distribution across the ring system. This activation is critical because it directs the subsequent electrophilic aromatic substitution (nitration) specifically to the 4-position, adjacent to the methyl group, minimizing the formation of unwanted regioisomers that would complicate downstream purification. Following nitration, the treatment with phosphorus trichloride serves a dual purpose: it acts as a deoxygenating agent to revert the N-oxide back to the pyridine form, and it simultaneously activates the nitro group for subsequent transformations. This careful orchestration of functional group interconversions ensures that the molecular architecture is built up logically, preserving the integrity of the heterocyclic core throughout the synthesis.
The final cyclization step represents the most chemically intricate phase of the entire process, where the linear precursor is converted into the fused bicyclic 5-azaindole system. The reaction of 3-methyl-4-nitropyridine with DMF dimethyl acetal generates an electron-rich enamine side chain at the 3-position. Upon exposure to Raney nickel and hydrazine hydrate, the nitro group undergoes reduction to an amine, which then spontaneously attacks the adjacent electrophilic carbon of the enamine moiety. This intramolecular nucleophilic attack triggers the closure of the five-membered pyrrole ring, aromatizing the system to yield the final product. The choice of Raney nickel is particularly strategic, as it provides the necessary catalytic surface for nitro reduction while tolerating the sensitive functionalities present in the intermediate. This mechanism effectively suppresses the formation of over-reduced byproducts, such as saturated indolines, ensuring that the final commercial scale-up of complex pharmaceutical intermediates proceeds with high selectivity and minimal waste generation.
How to Synthesize 5-Azaindole Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and temperature controls defined in the patent to maximize yield and safety. The process begins with the controlled oxidation of 3-picoline, followed by a carefully managed nitration where temperature must be kept below 30°C during addition to prevent runaway exotherms. The subsequent deoxygenation and enamine formation steps utilize common organic solvents like THF and DMF, which are easily recovered and recycled in a GMP environment. The final reductive cyclization is performed in methanol with hydrazine hydrate, a procedure that avoids the need for specialized high-pressure reactors. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Oxidize 3-methylpyridine using hydrogen peroxide or mCPBA to form 3-methylpyridine N-oxide.
- Perform nitration on the N-oxide using mixed acid to obtain 3-methyl-4-nitropyridine N-oxide.
- React the nitro-N-oxide with phosphorus trichloride in THF to remove the oxide group.
- Condense the resulting nitropyridine with DMF dimethyl acetal to form the vinyl intermediate.
- Execute reductive cyclization using Raney nickel and hydrazine hydrate to close the ring and yield 5-azaindole.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience. By shifting the starting material to 3-methylpyridine, a bulk commodity chemical produced on a massive global scale, manufacturers can decouple their supply chain from the volatility of specialized, low-volume reagents. The elimination of cryogenic steps removes the dependency on expensive cooling infrastructure and reduces energy overheads significantly. Moreover, the avoidance of n-butyllithium not only enhances site safety but also simplifies regulatory compliance and waste disposal protocols. These operational efficiencies translate into a more predictable manufacturing timeline, allowing procurement teams to secure reducing lead time for high-purity pharmaceutical intermediates and maintain buffer stocks without incurring prohibitive holding costs.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. Traditional methods relying on organolithium chemistry require stringent inert atmosphere conditions and specialized containment, which drive up capital expenditure and operational costs. In contrast, the new process utilizes hydrogen peroxide and phosphorus trichloride, which are inexpensive and widely available. Additionally, the improved reaction completeness reduces the burden on downstream purification, lowering solvent consumption and increasing overall material throughput. This holistic optimization results in substantial cost savings per kilogram of finished product, making it an attractive option for generic API production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often jeopardized by the reliance on single-source suppliers for exotic reagents. This synthesis mitigates that risk by utilizing a feedstock portfolio that is diversified and robust. 3-Methylpyridine is produced by multiple chemical giants worldwide, ensuring that raw material shortages are unlikely to disrupt production schedules. Furthermore, the mild reaction conditions reduce the likelihood of batch failures due to equipment malfunction or temperature excursions. This reliability allows supply chain managers to forecast demand more accurately and commit to longer-term contracts with downstream pharmaceutical clients, fostering stronger business relationships and market stability.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden inefficiencies, but this route is inherently designed for enlargement. The absence of high-pressure hydrogenation steps simplifies reactor design requirements, allowing for the use of standard glass-lined or stainless steel vessels. From an environmental standpoint, the process generates fewer hazardous byproducts compared to lithiation routes, which produce significant amounts of lithium salts. The use of hydrazine hydrate, while requiring careful handling, allows for a cleaner reduction profile with fewer heavy metal contaminants in the final product. This alignment with green chemistry principles facilitates easier environmental permitting and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-azaindole using this advanced methodology. These insights are derived directly from the comparative analysis of patent data and practical manufacturing considerations. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this intermediate into their broader drug substance supply chains.
Q: Why is the new 5-azaindole synthesis method safer than previous routes?
A: Previous methods relied on n-butyllithium at cryogenic temperatures (-40°C), posing significant thermal runaway and handling risks. The novel route operates under mild conditions using stable reagents like hydrogen peroxide and Raney nickel, eliminating the need for hazardous pyrophoric reagents and extreme cooling infrastructure.
Q: How does this process improve supply chain reliability for API manufacturers?
A: The process utilizes 3-methylpyridine (3-picoline) as a starting material, which is a commodity chemical with robust global availability. By avoiding specialized, low-volume reagents and complex cryogenic steps, the manufacturing lead time is significantly reduced, ensuring consistent supply continuity for downstream kinase inhibitor production.
Q: What are the purity advantages of this catalytic cyclization route?
A: The stepwise approach allows for intermediate purification, particularly after the nitration and deoxygenation steps. Furthermore, the final reductive cyclization using Raney nickel is highly selective, minimizing the formation of over-reduced byproducts often seen in high-pressure hydrogenation methods, thereby facilitating easier downstream crystallization and higher final assay.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Azaindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN103421004A process are fully realized in our manufacturing suites. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch of 5-azaindole against the highest industry standards. Our dedication to process optimization means we continuously refine reaction parameters to maximize yield and minimize impurity profiles, delivering a product that seamlessly integrates into your synthesis of kinase inhibitors.
We invite you to collaborate with us to unlock the full potential of this efficient synthetic route for your specific applications. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and project timelines. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence rather than speculation. Let us be your strategic partner in navigating the complexities of fine chemical manufacturing, ensuring that your supply of critical intermediates remains uninterrupted, cost-effective, and of the highest quality.
