Advanced Catalytic Strategy for 5-Azaindole Production Enhancing Commercial Scalability and Purity
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed significant demand on the supply chain for high-quality heterocyclic building blocks, specifically 5-azaindole. As detailed in patent CN1730481A, a groundbreaking preparation method has been developed that addresses the longstanding inefficiencies associated with traditional synthetic routes. This innovative protocol leverages a sophisticated three-step sequence involving condensation, selective reduction, and cyclization to deliver a product with exceptional purity and stability. For R&D directors and procurement specialists, understanding the nuances of this technology is critical, as it represents a shift towards more sustainable and economically viable manufacturing practices. The method utilizes 3-methyl-4-nitropyridine N-oxide as a starting material, reacting it with N,N-dialkylformamide dialkyl acetal under controlled thermal conditions to establish the carbon framework necessary for the final heterocyclic structure. By optimizing reaction parameters such as temperature and solvent choice, the process achieves yields that were previously difficult to attain consistently on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-azaindole has been plagued by thermodynamic and kinetic barriers that hindered large-scale production efficiency. Prior art, such as the method disclosed in U.S. Patent No. 4,625,033, relied on reaction conditions that inadvertently generated low-boiling byproducts like methanol during the process. The evolution of methanol vapor within the reaction mixture effectively lowered the internal reaction temperature, preventing the system from reaching the energy threshold required for complete conversion. Consequently, manufacturers faced the persistent challenge of incomplete reactions, necessitating complex purification steps to remove unreacted starting materials and intermediates. Furthermore, these legacy processes often demanded excessive quantities of Raney nickel catalyst to drive the hydrogenation, which not only inflated raw material costs but also created significant safety hazards associated with handling large volumes of pyrophoric materials. The inability to fully control the reaction environment resulted in variable product quality, making it difficult for supply chain managers to guarantee consistent batch-to-batch specifications for sensitive pharmaceutical applications.
The Novel Approach
In stark contrast to these outdated techniques, the methodology outlined in CN1730481A introduces a robust strategy that eliminates the formation of volatile interfering byproducts. By employing aprotic polar solvents such as N,N-dimethylformamide or N,N-diethylformamide, the reaction environment remains stable at elevated temperatures ranging from 100°C to 150°C, ensuring that the condensation step proceeds to near completion. This strategic choice of solvent not only facilitates the formation of the 3-dialkylamine vinyl-4-nitropyridine N-oxide intermediate with high efficiency but also allows for the recovery and recycling of the solvent, thereby enhancing the overall green chemistry profile of the synthesis. The subsequent steps utilize a dual-catalyst system where palladium on carbon is first employed for the mild reduction of the nitro group, followed by Raney nickel for the final deoxygenation and cyclization. This sequential catalytic approach minimizes side reactions and ensures that the delicate azaindole core is constructed with precision. 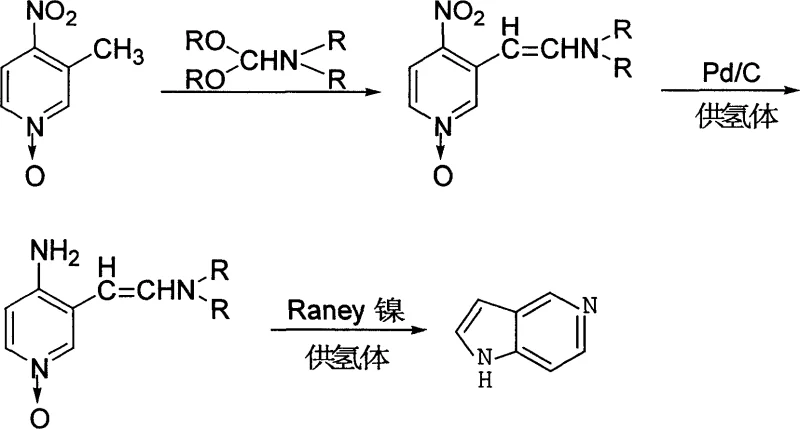
Mechanistic Insights into Reductive Cyclization and Catalyst Selectivity
The success of this synthetic route lies in the precise orchestration of electronic effects and catalytic selectivity during the transformation of the pyridine N-oxide derivative. In the initial condensation phase, the electron-withdrawing nature of the N-oxide moiety activates the methyl group at the 3-position, facilitating the attack by the electrophilic species generated from the formamide acetal. This Vilsmeier-Haack type interaction creates an extended conjugated system that is primed for subsequent reduction. The use of a palladium-containing catalyst in the second step is particularly ingenious, as it allows for the chemoselective reduction of the nitro group to an amine without prematurely reducing the N-oxide or the vinyl double bond under mild conditions between 0°C and room temperature. This selectivity is crucial because premature reduction of the N-oxide could lead to polymerization or the formation of undesired regioisomers, complicating the purification landscape. The hydrogen donor, whether hydrazine hydrate or ammonium formate, serves as a safe and effective source of nascent hydrogen, avoiding the need for high-pressure hydrogen gas equipment which often poses logistical challenges in multi-purpose plants.
Following the formation of the amino intermediate, the final cyclization step relies on the unique properties of Raney nickel to effect both deoxygenation and ring closure simultaneously. At temperatures between 40°C and 80°C, the Raney nickel catalyst promotes the cleavage of the nitrogen-oxygen bond while facilitating the nucleophilic attack of the newly formed amine onto the vinyl carbon. This intramolecular cyclization restores aromaticity to the pyrrole ring, driving the reaction forward thermodynamically. The mechanism ensures that the nitrogen atom from the original nitro group is seamlessly integrated into the five-membered ring, preserving the atomic economy of the process. Moreover, the mild conditions of this final step prevent the degradation of the sensitive azaindole scaffold, which can be prone to decomposition under harsh acidic or basic conditions found in other synthetic methodologies. The result is a crystalline product with superior color and purity, ready for direct use in downstream coupling reactions without extensive recrystallization.
How to Synthesize 5-Azaindole Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the formamide acetal and the precise control of temperature gradients during the exothermic reduction phases. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors with minimal modification to the core protocol. Operators must ensure that the palladium catalyst is thoroughly filtered and recovered after the nitro reduction to prevent contamination of the final Raney nickel step, which could alter the reaction kinetics. Detailed standard operating procedures regarding the addition rates of hydrogen donors are essential to maintain safety and maximize yield. For a comprehensive breakdown of the specific operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Condense 3-methyl-4-nitropyridine N-oxide with N,N-dialkylformamide dialkyl acetal in an aprotic polar solvent at 100-150°C to form the vinyl intermediate.
- Reduce the nitro group to an amino group using a palladium-containing catalyst and a hydrogen donor at temperatures between 0°C and room temperature.
- Perform deoxygenation and cyclization using Raney nickel and a hydrogen donor at 40-80°C to finalize the 5-azaindole structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The ability to recycle aprotic polar solvents significantly reduces the volume of hazardous waste generated, aligning with increasingly strict environmental regulations and lowering disposal costs. Furthermore, the recovery and reuse of expensive heterogeneous catalysts like palladium on carbon and Raney nickel represent a major opportunity for cost optimization, as these materials constitute a significant portion of the variable manufacturing expenses. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or high-pressure infrastructure, thereby reducing the utility burden on production facilities. These factors combined create a manufacturing profile that is not only economically attractive but also robust enough to withstand fluctuations in raw material availability.
- Cost Reduction in Manufacturing: The elimination of high-pressure hydrogenation equipment and the ability to operate at atmospheric pressure significantly lowers capital expenditure requirements for new production lines. By utilizing transfer hydrogenation with safe donors like ammonium formate, the process avoids the logistical complexities and safety costs associated with storing and handling bulk hydrogen gas. Additionally, the high yield of the initial condensation step means that less starting material is wasted, directly improving the material cost per kilogram of the final API intermediate. The qualitative reduction in solvent consumption through recycling loops further drives down the operational expenditure, making the final product more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as 3-methyl-4-nitropyridine N-oxide ensures that the supply chain is not vulnerable to shortages of exotic or highly specialized precursors. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor variations in ambient temperature or humidity, providing procurement managers with greater predictability in lead times. Moreover, the ease of catalyst recovery ensures that the production process is not halted by delays in catalyst replenishment, as the same batch of catalyst can be utilized for multiple cycles without significant loss of activity. This continuity is vital for maintaining the steady flow of intermediates required for just-in-time manufacturing strategies employed by major pharmaceutical companies.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with heat management and mixing requirements that are easily met in standard stainless steel reactors. The absence of toxic heavy metal residues in the final product simplifies the regulatory filing process, as extensive heavy metal clearance testing is minimized due to the efficient filtration steps. Waste streams are primarily composed of recoverable organic solvents and water, which can be treated using standard effluent treatment protocols, reducing the environmental footprint of the facility. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this 5-azaindole synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs. The responses cover aspects of yield optimization, catalyst management, and product quality assurance to ensure a comprehensive understanding of the technology.
Q: How does this method improve upon previous synthesis routes for 5-azaindole?
A: Unlike conventional methods that suffer from incomplete reactions due to low-boiling byproducts like methanol, this novel approach utilizes aprotic polar solvents and specific catalytic systems to ensure high conversion rates and easier product isolation.
Q: What are the key advantages regarding catalyst recovery in this process?
A: The process employs heterogeneous catalysts such as palladium on carbon and Raney nickel, which can be easily separated via filtration and recycled for subsequent batches, significantly reducing material waste and operational costs.
Q: Is the purity of the final 5-azaindole suitable for pharmaceutical applications?
A: Yes, the method yields 5-azaindole with excellent color and high purity, meeting the stringent quality specifications required for downstream synthesis of kinase inhibitors and other active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Azaindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-azaindole meets the exacting standards required for pharmaceutical synthesis. Our commitment to technical excellence means that we can adapt this patented route to fit your specific capacity requirements while maintaining the highest levels of quality and consistency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this more efficient process. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project timeline. Let us partner with you to secure a reliable and cost-effective supply of this vital kinase inhibitor intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
