Optimizing Nifuratel Manufacturing: A Safer, Scalable Synthetic Route for Global Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency with stringent safety and environmental standards. Patent CN101037434A introduces a transformative production method for the antibacterial agent Nifuratel, addressing critical bottlenecks found in legacy manufacturing processes. This innovation shifts the synthetic foundation from hazardous methyl mercaptan derivatives to a safer thiourea-based initiation, fundamentally altering the risk profile of the supply chain. For global procurement teams and R&D directors, this represents a pivotal opportunity to secure a reliable nifuratel supplier capable of delivering high-purity pharmaceutical intermediates without the baggage of volatile reagent handling. The technical breakthrough lies not merely in yield optimization but in the structural simplification of the early-stage synthesis, which cascades into significant downstream benefits regarding waste management and operational safety. By adopting this methodology, manufacturers can align their production capabilities with modern green chemistry principles while maintaining the rigorous quality standards required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Nifuratel relied heavily on the utilization of methyl mercaptan or sodium methyl mercaptide to construct the essential sulfur-containing side chain. As illustrated in the conventional pathway below, these reagents present severe logistical and safety challenges, including extreme toxicity, foul odors, and complex containment requirements that drive up operational expenditures. Furthermore, traditional cyclization steps often necessitated the use of metallic sodium in methanol-sodium methylate systems, introducing a substantial risk of explosion and fire that complicates large-scale reactor operations. These legacy constraints not only inflate the cost reduction in pharmaceutical intermediates manufacturing but also create fragile supply chains vulnerable to regulatory shutdowns due to environmental non-compliance. The reliance on such hazardous materials limits the ability of producers to offer competitive lead times, as safety protocols inevitably slow down batch turnover and increase the complexity of waste disposal procedures.
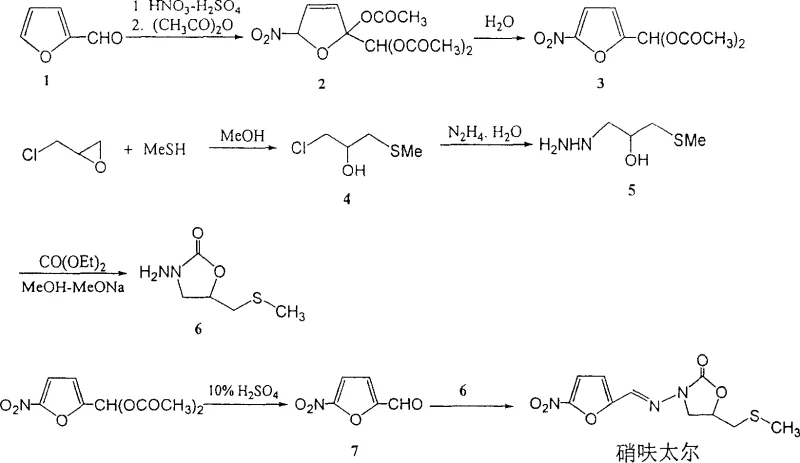
The Novel Approach
In stark contrast, the novel methodology outlined in the patent data replaces these dangerous precursors with thiourea and dimethyl sulfate, creating a stable S-methylisothiourea intermediate that is far easier to handle and store. This strategic substitution eliminates the need for gaseous or highly volatile sulfur sources, thereby drastically simplifying the engineering controls required for the production facility. The subsequent reaction with epichlorohydrin under phase-transfer catalysis conditions ensures high conversion rates without the generation of noxious byproducts, fostering a cleaner production environment. By removing metallic sodium from the equation and utilizing standard alkali metal hydroxides or hydrides, the process mitigates explosion hazards and allows for more flexible temperature control during the critical cyclization phases. This approach not only enhances supply chain reliability by reducing the risk of accident-related stoppages but also positions the manufacturer as a leader in sustainable chemical processing, appealing to increasingly eco-conscious global partners.
Mechanistic Insights into Thiourea-Based Cyclization
The core of this technological advancement resides in the efficient construction of the oxazolidone ring system through a refined nucleophilic substitution and cyclization sequence. The process begins with the formation of 2-(methylthiomethyl)-oxirane from thiourea, a reaction that proceeds with high selectivity under mild alkaline conditions facilitated by phase-transfer catalysts like tetra-n-butyl ammonium bromide. This intermediate then undergoes ring-opening with hydrazine hydrate to yield the key hydrazine derivative, which serves as the precursor for the final heterocyclic assembly. The elimination of harsh reducing agents and the use of diethyl carbonate as both a reagent and solvent in the cyclization step demonstrate a sophisticated understanding of atom economy and process safety. Such mechanistic precision ensures that impurity profiles remain tightly controlled, a critical factor for R&D directors evaluating the feasibility of integrating this intermediate into complex drug formulations. The robustness of this chemistry allows for consistent batch-to-batch reproducibility, which is essential for maintaining the stringent purity specifications demanded by regulatory bodies.
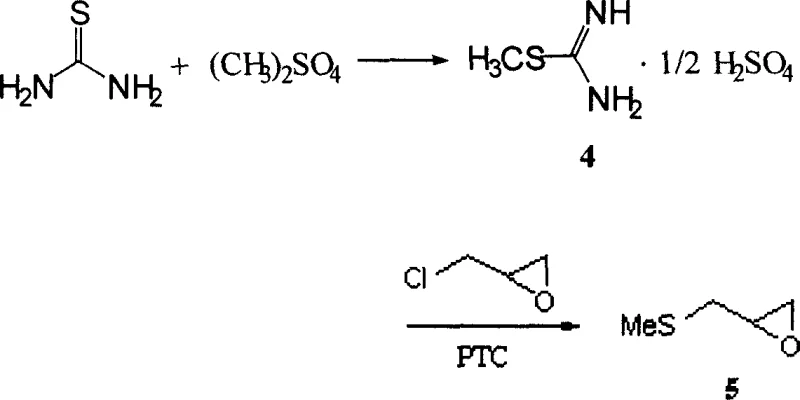
Furthermore, the impurity control mechanism is inherently built into the choice of reagents and the sequential purification strategies employed throughout the synthesis. By avoiding the use of metallic sodium, the formation of unwanted side products associated with radical reactions or over-reduction is effectively suppressed, leading to a cleaner crude product profile. The final condensation with 5-nitrofurfural is conducted under controlled acidic conditions that favor the formation of the desired hydrazone linkage while minimizing degradation of the sensitive nitro-furan moiety. Recrystallization steps using glacial acetic acid or acetonitrile-ethanol mixtures further refine the product, removing trace organic impurities and residual solvents to meet pharmacopeial standards. This level of chemical rigor ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed without compromising on the quality attributes that define therapeutic efficacy. The result is a high-purity OLED material equivalent in terms of chemical consistency, tailored specifically for the demanding requirements of the antibiotic sector.
How to Synthesize Nifuratel Efficiently
Implementing this advanced synthetic route requires a clear understanding of the sequential reaction conditions and the specific roles of each reagent in the transformation cascade. The process is designed to be modular, allowing for the isolation or telescoping of key intermediates depending on the specific capacity and equipment available at the manufacturing site. Detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry and thermal parameters needed to achieve optimal yields. Operators must pay close attention to the temperature ranges during the thiourea alkylation and the subsequent hydrazine condensation to prevent thermal runaway or decomposition. Adhering to these protocols ensures that the reducing lead time for high-purity pharmaceutical intermediates is realized without sacrificing safety or quality control measures.
- React thiourea with dimethyl sulfate under cooling to form S-methylisothiourea sulfate, then react with epichlorohydrin under phase-transfer catalysis.
- Condense the resulting epoxide intermediate with hydrazine hydrate to generate the hydrazine derivative without purification.
- Cyclize the hydrazine derivative with diethyl carbonate using a safe alkali base, followed by condensation with 5-nitrofurfural.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this thiourea-based methodology offers profound advantages that extend well beyond the laboratory bench, directly impacting the bottom line and operational resilience of the supply chain. The elimination of hazardous gases like methyl mercaptan removes the need for specialized scrubbing systems and expensive personal protective equipment, resulting in substantial cost savings in facility maintenance and regulatory compliance. Additionally, the replacement of metallic sodium with common alkali bases reduces raw material volatility and storage costs, stabilizing the input pricing structure against market fluctuations. These efficiencies translate into a more competitive pricing model for the final API, allowing procurement managers to negotiate better terms while ensuring a steady flow of materials. The simplified waste stream also means lower disposal fees and a reduced environmental footprint, aligning with corporate sustainability goals that are increasingly becoming a prerequisite for vendor selection in the global market.
- Cost Reduction in Manufacturing: The removal of expensive and dangerous reagents such as metallic sodium and methyl mercaptan significantly lowers the direct material costs associated with production. By utilizing readily available commodities like thiourea and diethyl carbonate, the process insulates the supply chain from the price volatility often seen with specialty hazardous chemicals. Furthermore, the ability to recycle solvents like ethanol and benzene within the closed-loop system minimizes waste generation, leading to drastic reductions in effluent treatment expenses. This economic efficiency allows manufacturers to offer more attractive pricing structures without compromising on the rigorous quality standards required for pharmaceutical grade materials.
- Enhanced Supply Chain Reliability: The inherent safety of the new process reduces the likelihood of unplanned shutdowns caused by safety incidents or regulatory inspections related to hazardous material handling. Sourcing thiourea and standard alkalis is far more reliable than securing controlled substances like methyl mercaptan, ensuring continuous production even during periods of market tightness. This stability is crucial for maintaining consistent delivery schedules and building long-term trust with downstream partners who depend on just-in-time inventory models. The robust nature of the chemistry also allows for faster batch turnover, effectively reducing lead times and enhancing the overall responsiveness of the supply network to sudden demand spikes.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reactor configurations that do not require exotic materials of construction to withstand corrosive or explosive conditions. This ease of scale-up facilitates the transition from pilot plant quantities to multi-ton commercial production with minimal engineering hurdles. Moreover, the significant reduction in toxic emissions and hazardous waste aligns with strict international environmental regulations, mitigating the risk of fines or operational restrictions. This compliance advantage future-proofs the manufacturing asset, ensuring long-term viability in an increasingly regulated global chemical landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Nifuratel synthesis method. These insights are derived directly from the patent specifications and practical considerations for industrial application, providing clarity for stakeholders evaluating this technology. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification. The answers reflect a commitment to transparency and technical excellence, ensuring that all partners have a clear view of the capabilities and limitations of this advanced manufacturing route.
Q: Why is the thiourea route preferred over methyl mercaptan for Nifuratel production?
A: The thiourea route eliminates the use of methyl mercaptan, a highly toxic and foul-smelling gas, significantly improving workplace safety and reducing environmental compliance costs associated with hazardous waste treatment.
Q: How does the new method improve industrial scalability?
A: By replacing metallic sodium with standard alkali hydroxides or hydrides, the process removes explosion risks and allows for simpler reactor designs, facilitating easier scale-up from pilot to commercial tonnage.
Q: What are the purity specifications achievable with this method?
A: The method utilizes recrystallization techniques using glacial acetic acid or acetonitrile/ethanol mixtures, ensuring high-purity standards suitable for pharmaceutical applications while minimizing solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nifuratel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the thiourea-based Nifuratel method are translated into tangible supply chain assets. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for safety and efficacy. Our commitment to continuous improvement drives us to integrate such advanced methodologies, offering our clients a competitive edge through superior product quality and operational reliability.
We invite you to collaborate with us to explore how this optimized production method can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies. Partner with us to secure a sustainable and cost-effective supply of high-quality Nifuratel intermediates that drive your business forward.
