Advanced Manufacturing of Nifuratel via Boc-Protected Intermediates for Global Pharma Supply Chains
Advanced Manufacturing of Nifuratel via Boc-Protected Intermediates for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking more efficient, environmentally benign, and cost-effective pathways for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in patent CN112457304A, which details a novel preparation method for Nifuratel, a broad-spectrum antibiotic widely used for treating vulvar and vaginal infections, urinary tract infections, and various parasitic diseases. This new synthetic strategy represents a paradigm shift from traditional, hazardous protocols to a cleaner, Boc-protected route that ensures superior product quality and operational safety. By leveraging advanced heterocyclic chemistry, this method addresses long-standing challenges regarding odor control, waste management, and yield optimization, positioning it as a preferred choice for reliable pharmaceutical intermediates supplier networks globally.
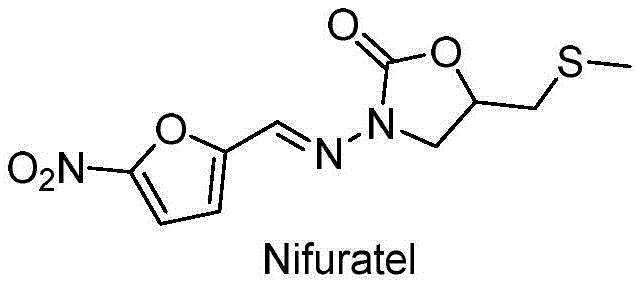
Nifuratel, chemically known as 5-[(methylthio)methyl]-3-[(5-nitrofurylidene)amino]-2-oxazolidinone, is a vital component in veterinary and human medicine. The structural complexity of this molecule, particularly the sensitive nitrofuran ring and the thioether linkage, demands precise synthetic control to avoid degradation and impurity formation. The patented process introduces a strategic sequence starting from epichlorohydrin and tert-butyl carbazate, effectively bypassing the handling of volatile and toxic reagents in the initial stages. This approach not only enhances the safety profile of the manufacturing facility but also streamlines the supply chain by utilizing stable, solid-state reagents that are easier to transport and store compared to gaseous or highly volatile liquid precursors used in legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Nifuratel has relied on routes that pose significant environmental and operational hazards, creating bottlenecks for procurement managers and supply chain heads. As illustrated in prior art such as Scheme 1, traditional methods often involve the direct reaction of epichlorohydrin with methyl mercaptan. Methyl mercaptan is a gas with an intensely foul odor and high toxicity, requiring specialized containment equipment and rigorous scrubbing systems to prevent atmospheric release. Furthermore, these conventional pathways frequently necessitate the use of excessive hydrazine hydrate, often in molar ratios exceeding 3:1, leading to substantial chemical waste and increased costs associated with wastewater treatment and disposal.
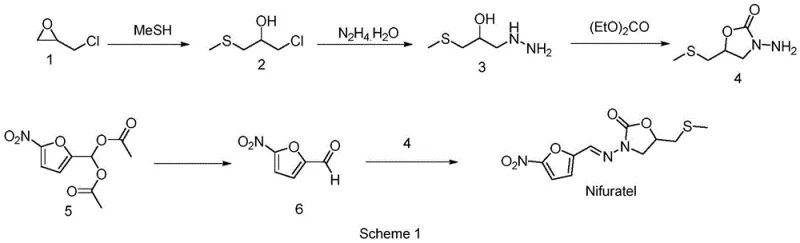
In addition to the issues with methyl mercaptan, older synthetic routes generate low-boiling sulfur-containing intermediates that are prone to volatilization and loss during processing. This volatility not only reduces the overall mass balance and yield of the final product but also contributes to unpleasant working conditions and potential regulatory compliance issues regarding volatile organic compound (VOC) emissions. The reliance on such unstable intermediates makes scale-up unpredictable and risky, as minor fluctuations in temperature or pressure can lead to significant deviations in product quality. Consequently, manufacturers adhering to these legacy processes face higher operational expenditures and greater difficulty in maintaining consistent supply continuity for their downstream pharmaceutical clients.
The Novel Approach
The innovative methodology disclosed in patent CN112457304A fundamentally reengineers the synthetic landscape by introducing a Boc-protection strategy that mitigates these risks at the source. Instead of reacting epichlorohydrin directly with toxic thiols, the new route begins with the reaction of epichlorohydrin and tert-butyl carbazate to form a stable hydrazine intermediate. This strategic pivot eliminates the need for handling hazardous methyl mercaptan in the early stages and avoids the generation of low-boiling sulfur compounds until the very end of the synthesis, where they are immediately incorporated into the final stable structure. The use of carbonyldiimidazole (CDI) for cyclization further exemplifies this modern approach, offering a mild and efficient mechanism to construct the oxazolidinone ring without the harsh conditions associated with diethyl carbonate cyclization.
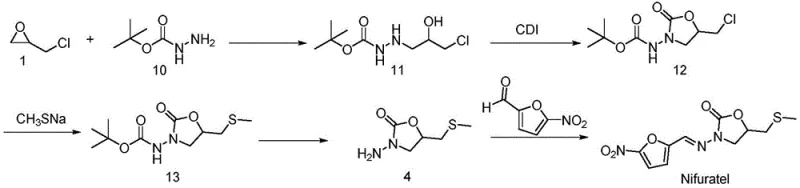
This novel approach delivers tangible benefits in terms of yield and purity, which are critical metrics for R&D directors evaluating new suppliers. Experimental data from the patent indicates that key intermediates can be obtained with yields exceeding 90%, such as the 96.8% yield for the initial hydrazine adduct and 94.8% for the cyclized oxazolidinone. The final condensation step with 5-nitrofurfural proceeds efficiently to provide Nifuratel with a purity of 99.8%, demonstrating the robustness of the purification profile. By shortening the route and stabilizing reactive intermediates through Boc-protection, this method offers a streamlined pathway that is not only environmentally friendly but also economically superior, aligning perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into CDI-Mediated Cyclization and Substitution
The core of this synthetic advancement lies in the precise mechanistic execution of the ring-closing and substitution steps, which dictate the final impurity profile of the API. In the second step of the process, the linear hydrazine intermediate undergoes cyclization mediated by CDI (carbonyldiimidazole). Unlike traditional phosgene or chloroformate reagents which can leave toxic residues, CDI activates the hydroxyl group to form an imidazolyl carbonate intermediate, which is then intramolecularly attacked by the adjacent nitrogen to close the oxazolidinone ring. This reaction is typically conducted in tetrahydrofuran at temperatures between 60-65°C, conditions that are mild enough to preserve the integrity of the acid-sensitive Boc group while driving the reaction to completion. The result is a clean conversion with minimal side products, facilitating easier downstream processing.
Following cyclization, the introduction of the methylthio group is achieved through a nucleophilic substitution reaction using sodium methanethiolate. This step replaces the chlorine atom on the side chain with the sulfur moiety. Crucially, because the sulfur is introduced as a salt (sodium methanethiolate) rather than a volatile gas, the reaction can be performed in a closed system with high atom economy. The subsequent deprotection of the Boc group using trifluoroacetic acid or hydrochloric acid reveals the free hydrazine necessary for the final condensation. This sequence ensures that the reactive hydrazine functionality is masked during the harsher substitution conditions, preventing polymerization or decomposition, thereby ensuring a high-purity intermediate ready for the final coupling with 5-nitrofurfural.
How to Synthesize Nifuratel Efficiently
The synthesis of Nifuratel via this patented route involves a logical sequence of five distinct operational steps that transform simple commodity chemicals into a high-value pharmaceutical intermediate. The process begins with the alkylation of tert-butyl carbazate, followed by ring closure, sulfur incorporation, deprotection, and final condensation. Each step has been optimized for solvent choice and stoichiometry to maximize yield and minimize waste. For R&D teams looking to implement this technology, the detailed standardized synthesis steps are outlined below to ensure reproducibility and compliance with the patent specifications.
- React epichlorohydrin with tert-butyl carbazate in THF at 25°C to form the protected hydrazine intermediate.
- Cyclize the intermediate using CDI at 60-65°C to construct the oxazolidinone ring structure.
- Perform nucleophilic substitution with sodium methanethiolate to introduce the methylthio group, followed by acid deprotection and condensation with 5-nitrofurfural.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the raw material portfolio. By replacing hazardous gases like methyl mercaptan and toxic dimethyl sulfate with stable solids like tert-butyl carbazate and sodium methanethiolate, the supply chain becomes significantly more resilient. Solid reagents are easier to source, store, and handle, reducing the logistical complexities and insurance costs associated with transporting dangerous goods. This stability translates directly into enhanced supply chain reliability, ensuring that production schedules are not disrupted by regulatory hurdles or transportation delays common with volatile chemicals.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the elimination of expensive waste treatment protocols. Traditional methods require extensive scrubbing systems to neutralize foul-smelling sulfur gases and treat hydrazine-laden wastewater, which incurs substantial operational expenditures. By avoiding these pollutants, the new route significantly lowers the cost of environmental compliance. Furthermore, the high yields observed in each step—often exceeding 90%—mean that less raw material is required to produce the same amount of final product, directly improving the cost of goods sold (COGS) and allowing for more competitive pricing structures in the global market.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a consistent supply flow. Unlike specialized reagents that may have limited suppliers, epichlorohydrin and tert-butyl carbazate are commodity chemicals produced at scale by multiple manufacturers worldwide. This diversification of the supply base mitigates the risk of single-source dependency. Additionally, the robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic alloys for corrosion resistance against strong acids or gases, means that production can be easily transferred between different manufacturing sites without significant capital investment, further securing supply continuity.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns with the increasing global demand for green chemistry in the pharmaceutical sector. The absence of heavy metal catalysts and the reduction of volatile organic emissions make the process inherently cleaner and easier to permit in regions with strict environmental regulations. The scalability is evidenced by the straightforward workup procedures, such as simple extraction and crystallization, which are easily adaptable from kilogram to tonne scales. This ease of scale-up reduces the time-to-market for new batches and ensures that the manufacturing process remains compliant with evolving international standards for pharmaceutical production.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the implementation and benefits of this specific Nifuratel synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent, providing a clear picture of what partners can expect regarding performance and operational requirements.
Q: How does the new Nifuratel synthesis route improve environmental safety compared to traditional methods?
A: The novel process eliminates the use of foul-smelling and toxic methyl mercaptan gas in the early stages and avoids the generation of low-boiling volatile sulfur-containing intermediates, significantly reducing environmental pollution and operator exposure risks.
Q: What are the key advantages of using tert-butyl carbazate in this manufacturing process?
A: Using tert-butyl carbazate allows for a controlled, step-wise construction of the molecule without requiring excessive amounts of hydrazine hydrate, leading to higher purity profiles and simplified downstream purification processes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method features short reaction steps, uses cheap and readily available raw materials, and achieves high yields (over 90% in key steps), making it highly robust and cost-effective for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nifuratel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN112457304A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of Nifuratel meets the highest international pharmacopoeia standards.
We invite pharmaceutical companies and agrochemical manufacturers to leverage our technical proficiency to optimize their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener, more efficient route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering high-quality Nifuratel intermediates.
