Advanced Manufacturing of Tenofovir Alafenamide Intermediate via Novel Salt-Mediated Phosphorylation
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiviral intermediates, particularly for next-generation prodrugs like Tenofovir Alafenamide (TAF). Patent CN112390824B introduces a transformative preparation method for the key intermediate, 9-[(R)-2-[[(phenoxyphosphinyl)methoxy]propyl]]adenine (Compound I), which serves as the foundational scaffold for TAF fumarate. This novel approach addresses longstanding challenges in nucleotide analog synthesis by replacing traditional condensing agents with a streamlined inorganic base-mediated protocol. By utilizing Tenofovir as the starting material and converting it into an alkali metal monosalt prior to phosphorylation, the process achieves exceptional control over reaction stoichiometry and impurity generation. For R&D directors and procurement specialists, this represents a significant leap forward in process chemistry, offering a pathway that balances high chemical purity with operational simplicity. The method effectively mitigates the risks associated with residual coupling reagents and toxic solvents, positioning it as a preferred candidate for GMP-compliant commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Compound I has been plagued by inefficient coupling strategies that introduce significant downstream processing burdens. Traditional methods often rely on carbodiimide coupling agents like DCC (dicyclohexylcarbodiimide) to facilitate the reaction between Tenofovir and phenol derivatives. As illustrated in prior art reactions, the use of DCC generates substantial quantities of dicyclohexylurea byproducts, which are notoriously difficult to separate from the polar nucleotide product, leading to compromised yields and purity issues. Furthermore, alternative routes employing triphenyl phosphate with catalytic amounts of DMAP (4-dimethylaminopyridine) suffer from prolonged reaction times and the persistence of the catalyst in the final matrix, necessitating rigorous and costly purification steps. Other approaches involving Boc-protected precursors add unnecessary synthetic length, requiring additional deprotection steps that utilize hazardous ammonia solutions under pressure, thereby increasing safety risks and capital expenditure for specialized equipment.
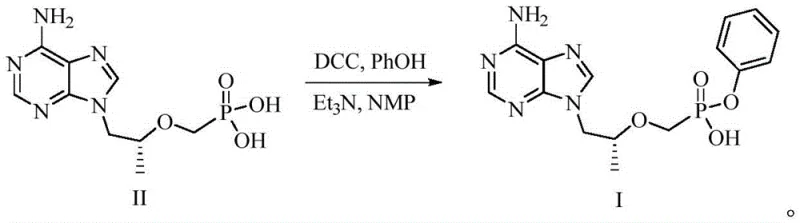
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN112390824B employs a direct and atom-economical strategy centered on the activation of the phosphonic acid moiety via salt formation. The process initiates by treating Tenofovir (Compound II) with a stoichiometric amount of an inorganic base, such as sodium carbonate or hydroxide, to generate a reactive alkali metal monosalt (Compound Ia). This activated species then undergoes a clean nucleophilic substitution with triphenyl phosphite in a high-boiling solvent system. This strategic shift eliminates the need for exogenous condensing agents entirely, thereby removing the source of urea-based impurities. The reaction conditions are mild yet effective, typically operating between 80°C and 140°C depending on the solvent choice, allowing for rapid conversion without the degradation often seen in harsher acidic or basic hydrolysis methods. The result is a streamlined workflow that significantly reduces the E-factor (environmental factor) of the synthesis.
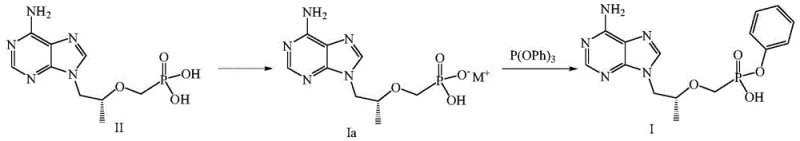
Mechanistic Insights into Inorganic Base-Mediated Phosphorylation
The core mechanistic advantage of this process lies in the enhanced nucleophilicity of the phosphonate monoanion generated in the first step. When Tenofovir is neutralized with an inorganic base like sodium hydroxide or potassium carbonate, the resulting monosalt exists in a state that is highly conducive to attacking the electrophilic phosphorus center of triphenyl phosphite. Unlike the neutral phosphonic acid, which requires strong activation to participate in P-O bond formation, the anionic species facilitates a smoother displacement of the phenoxy leaving group from the phosphite. This mechanism bypasses the high-energy transition states associated with carbodiimide activation, leading to a more favorable kinetic profile. The use of polar aprotic solvents such as N,N-dimethylacetamide (DMAc) or acetonitrile further stabilizes the ionic intermediates, ensuring that the reaction proceeds to completion with minimal side reactions such as diester formation or hydrolysis of the sensitive adenine base.
From an impurity control perspective, this mechanism offers superior selectivity. Traditional methods often struggle with the formation of symmetrical pyrophosphate byproducts or incomplete esters due to poor stoichiometric control. By pre-forming the monosalt, the process ensures a precise 1:1 molar ratio of reactive species entering the phosphorylation step. The absence of organic amines like triethylamine or pyridine in large excess prevents the formation of quaternary ammonium salts that can co-crystallize with the product. Consequently, the crude product obtained after the reaction exhibits a remarkably clean profile, often exceeding 99% purity by HPLC before the final crystallization. This inherent purity reduces the reliance on preparative chromatography or multiple recrystallizations, which are major cost drivers in the manufacturing of high-purity pharmaceutical intermediates.
How to Synthesize Tenofovir Alafenamide Intermediate Efficiently
The execution of this synthesis requires careful attention to the isolation of the intermediate salt to ensure optimal reactivity in the second step. The process begins with the suspension of Tenofovir in a solvent like acetone, followed by the controlled addition of the aqueous base solution to form the monosalt precipitate. This solid is then isolated, dried, and immediately subjected to the phosphorylation conditions with triphenyl phosphite. The detailed operational parameters, including specific temperature ramps and solvent volumes, are critical for maximizing yield and minimizing the formation of the diphenyl ester impurity. For a comprehensive understanding of the standardized operating procedures required to replicate these results in a pilot or production setting, please refer to the technical guide below.
- Dissolve Tenofovir (Compound II) in a solvent such as acetone or methanol to form a slurry, then add an aqueous solution of an inorganic base (e.g., sodium carbonate or hydroxide) to generate the alkali metal monosalt (Compound Ia).
- Isolate the dried Compound Ia and react it with triphenyl phosphite in a suitable solvent like N,N-dimethylacetamide or toluene under heating conditions.
- Perform post-treatment by adding water and ethyl acetate, separating the aqueous phase, adjusting pH to 2-3 with hydrochloric acid, and crystallizing to obtain high-purity Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The primary economic driver is the elimination of expensive and hazardous reagents. By removing DCC and DMAP from the bill of materials, manufacturers avoid the high costs associated with these specialty chemicals and the complex waste disposal fees linked to their byproducts. Furthermore, the simplified workup procedure, which relies on standard aqueous extractions and acidification rather than complex chromatographic separations, drastically reduces solvent consumption and processing time. This efficiency gain allows for faster batch turnover and higher throughput in existing reactor trains, effectively increasing capacity without the need for new capital investment in infrastructure.
- Cost Reduction in Manufacturing: The substitution of costly condensing agents with inexpensive inorganic bases like sodium carbonate creates a profound impact on the cost of goods sold (COGS). Since the process avoids the generation of difficult-to-remove urea byproducts, the yield loss typically associated with aggressive purification steps is virtually eliminated. Additionally, the ability to recover and reuse solvents like acetone and toluene further enhances the economic viability of the process. The overall material utilization rate is significantly improved, ensuring that a higher percentage of the input mass is converted into valuable saleable product rather than waste.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triphenyl phosphite and common inorganic bases ensures a robust and resilient supply chain. Unlike routes dependent on proprietary protected intermediates or specialized catalysts that may face supply bottlenecks, the raw materials for this process are widely available from multiple global vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of the intermediate salt allows for potential storage or transport if the two steps need to be decoupled geographically, offering flexibility in manufacturing logistics.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, characterized by simple unit operations that are easily managed in large-scale reactors. The reduction in toxic organic waste, specifically the absence of pyridine and carbodiimide residues, aligns perfectly with increasingly stringent environmental regulations regarding pharmaceutical effluent. This green chemistry profile not only reduces the burden on wastewater treatment facilities but also enhances the corporate sustainability metrics of the manufacturing site. The straightforward crystallization process ensures consistent particle size and morphology, which are critical quality attributes for downstream formulation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for potential licensees or manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into an existing production portfolio.
Q: How does the inorganic base method improve impurity profiles compared to DCC coupling?
A: The inorganic base method eliminates the use of DCC, which generates difficult-to-remove urea byproducts. By forming a clean alkali metal monosalt intermediate, the subsequent phosphorylation with triphenyl phosphite proceeds with higher selectivity, resulting in significantly lower levels of organic impurities and easier purification.
Q: What are the scalability advantages of avoiding organic bases like pyridine or DMAP?
A: Avoiding large excesses of toxic organic bases like pyridine or expensive catalysts like DMAP simplifies the workup procedure drastically. This reduces the environmental burden of waste liquid treatment and lowers the risk of residual amine impurities in the final API, making the process more robust for multi-ton commercial production.
Q: Is the raw material availability stable for this synthetic route?
A: Yes, the route utilizes Tenofovir and triphenyl phosphite, both of which are commercially available commodity chemicals. Unlike routes requiring specialized Boc-protected precursors or complex phase-transfer systems, this method relies on standard inorganic bases and common solvents, ensuring a stable and continuous supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Alafenamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for antiviral therapeutics. Our team of expert chemists has thoroughly analyzed the technological merits of the salt-mediated phosphorylation route described in CN112390824B and is fully prepared to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and reproducible. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for API synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive value and security for your Tenofovir Alafenamide projects.
