Advanced Microwave-Assisted Synthesis of Caspofungin Acetate for Commercial Scale-Up
Advanced Microwave-Assisted Synthesis of Caspofungin Acetate for Commercial Scale-Up
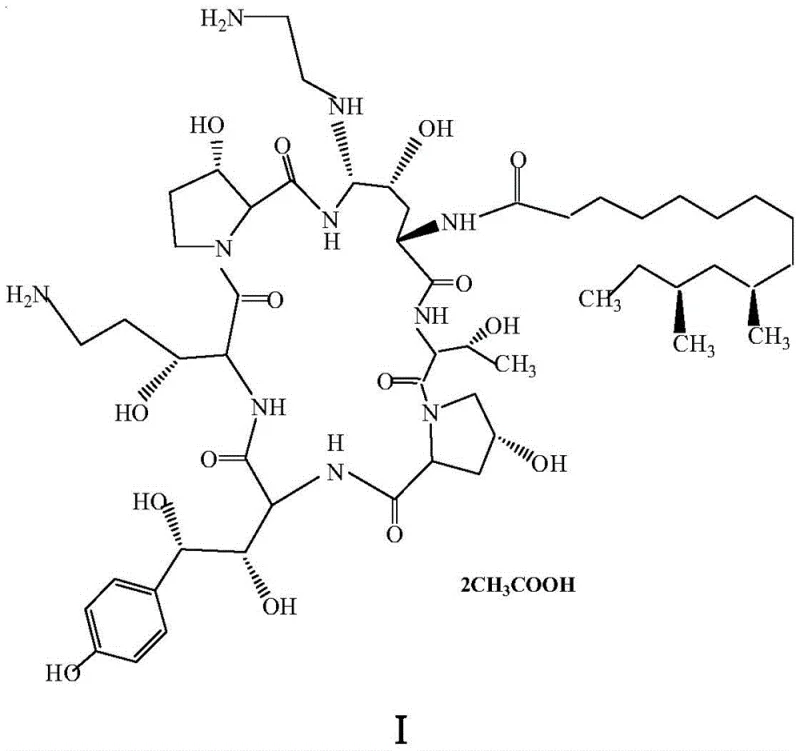
The pharmaceutical landscape for antifungal agents has been significantly transformed by the introduction of echinocandins, with Caspofungin Acetate standing out as a critical therapeutic for invasive aspergillosis and candidemia. The preparation method detailed in patent CN107778360B represents a paradigm shift in the synthetic route for this complex molecule, addressing long-standing inefficiencies in yield and reaction time that have plagued previous manufacturing protocols. By leveraging advanced microwave irradiation technology and optimized stereoselective agents, this novel process offers a robust pathway for the commercial scale-up of complex peptide mimetics. The core innovation lies in the strategic replacement of the hemiaminal hydroxyl group in Pneumocandin B0 with a specifically chosen thioether moiety, followed by a streamlined reduction and substitution sequence. This approach not only enhances the overall throughput but also ensures the production of high-purity echinocandin intermediates that meet stringent regulatory standards for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Caspofungin Acetate has been hindered by tedious multi-step sequences that result in abysmal overall yields and significant purification challenges. The original process developed by Merck, protected under US5378804, suffered from an overall yield of merely 0.7%, primarily due to the inefficiency of the initial derivatization steps and the reliance on harsh conditions that degraded the sensitive peptide backbone. Subsequent improvements, such as those described by Leonard et al., managed to raise the yield to approximately 45%, yet the process remained burdened by prolonged reaction times, often requiring up to 48 hours for the critical substitution steps using conventional thiophenols. Furthermore, many prior art methods relied on the use of expensive and toxic transition metal catalysts, such as cobalt chloride, for the reduction of nitrile intermediates, necessitating complex downstream processing to remove heavy metal residues. These factors collectively contributed to high production costs and extended lead times, creating a bottleneck for cost reduction in antifungal API manufacturing.
The Novel Approach
The methodology disclosed in CN107778360B fundamentally reengineers the synthetic pathway by introducing microwave-assisted organic synthesis (MAOS) coupled with superior stereoselective agents. The first critical innovation is the utilization of beta-naphthylthiophenol, or its derivatives like 6-chloro-2-naphthylthiophenol, as a stereostructure selector. Unlike simple phenylthiophenols used in the past, these naphthyl-based selectors exhibit enhanced reactivity and steric properties that facilitate a much faster and cleaner substitution of the hemiaminal hydroxyl group. When combined with microwave irradiation at controlled temperatures (10-20°C), the reaction time for this step is slashed from days to merely 30-35 minutes, achieving yields exceeding 93% and purities greater than 98%. This dramatic improvement in the first step sets a high-quality foundation for the subsequent transformations, effectively minimizing the accumulation of impurities that are difficult to remove in later stages.
Mechanistic Insights into Microwave-Assisted Stereoselective Substitution
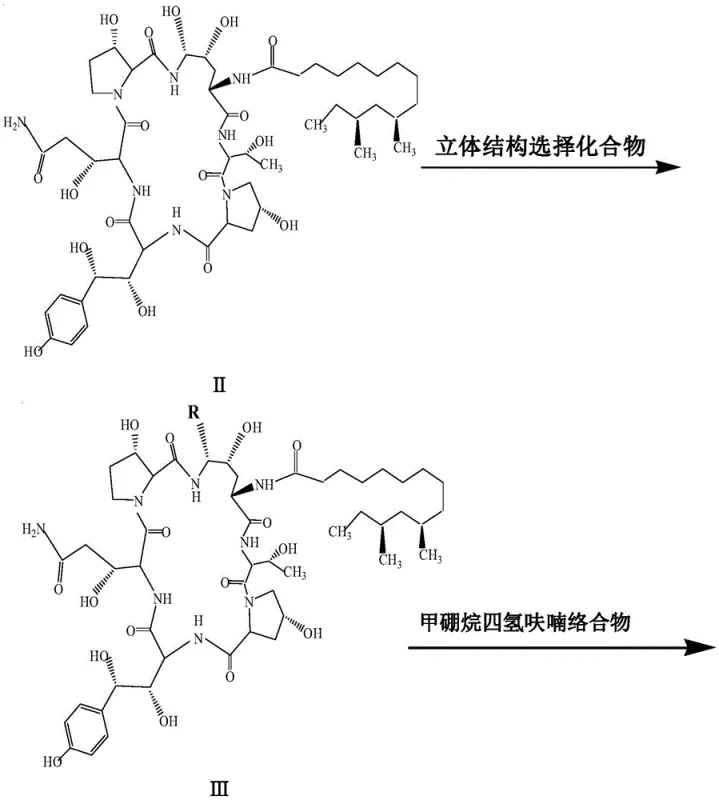
The mechanistic superiority of this process stems from the synergistic effect of the specific thioether selector and the unique heating profile provided by microwave irradiation. In the initial step, the reaction between Pneumocandin B0 and the naphthylthiophenol is catalyzed by trifluoromethanesulfonic acid in the presence of phenylboronic acid. The microwave energy facilitates rapid dipolar polarization of the solvent and reactants, leading to instantaneous heating that overcomes the activation energy barrier much more efficiently than conductive heating. This results in a highly stereoselective formation of the thioether intermediate (Compound III), where the bulky naphthyl group effectively directs the substitution to the desired configuration while suppressing the formation of isomers. The subsequent reduction of the primary amide to the primary amine (Compound IV) is achieved using a borane-tetrahydrofuran complex following silylation protection. This choice of reducing agent is pivotal; it avoids the use of cobalt salts entirely, thereby eliminating the risk of metal contamination and simplifying the purification protocol to a straightforward liquid-phase separation.
In the final transformation, the thioether group in Compound IV is displaced by ethylenediamine to install the critical side chain required for antifungal activity. Conventional methods for this nucleophilic substitution often require prolonged heating (40+ hours) to drive the reaction to completion, which risks degrading the sensitive cyclic hexapeptide core. However, the application of microwave irradiation in this step allows the reaction to proceed at moderate temperatures (30-40°C) within 1 to 2.5 hours. The kinetic energy provided by the microwaves enhances the nucleophilicity of the ethylenediamine, ensuring a clean displacement of the thioether leaving group. This efficiency is crucial for maintaining the integrity of the molecule and achieving the high final purity of 99.3% reported in the examples. The entire sequence demonstrates a profound understanding of physical organic chemistry applied to process optimization.
How to Synthesize Caspofungin Acetate Efficiently
The synthesis of Caspofungin Acetate via this novel route involves a carefully orchestrated sequence of reactions that prioritize speed, selectivity, and safety. The process begins with the suspension of Pneumocandin B0 and phenylboronic acid in acetonitrile, followed by the addition of the stereoselective thiol agent. Upon the dropwise addition of trifluoromethanesulfonic acid at low temperatures, the mixture is subjected to microwave irradiation to generate the thioether intermediate. Following isolation and purification, the intermediate undergoes silylation and reduction using borane-THF to yield the amine precursor. Finally, the amine is reacted with ethylenediamine under microwave conditions, and the resulting free base is converted to the acetate salt. For a detailed breakdown of the specific reagents, stoichiometry, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- React Pneumocandin B0 with beta-naphthylthiophenol and phenylboronic acid under microwave irradiation to form the thioether intermediate.
- Reduce the primary amide of the intermediate to a primary amine using a borane-tetrahydrofuran complex after silylation protection.
- Substitute the thioether group with ethylenediamine under microwave conditions and convert to the acetate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-assisted synthesis route offers tangible benefits that extend far beyond the laboratory bench. The most immediate impact is seen in the drastic reduction of cycle times; by compressing reaction steps that previously took nearly two days into mere hours, manufacturers can significantly increase batch turnover rates without expanding facility footprint. This acceleration directly translates to improved supply continuity and the ability to respond more agilely to market demand fluctuations for this critical antifungal agent. Furthermore, the elimination of expensive transition metal catalysts like cobalt chloride removes a significant cost driver and mitigates the environmental liabilities associated with heavy metal waste disposal. The simplified purification requirements, driven by the high selectivity of the microwave reactions, reduce the consumption of chromatographic media and solvents, further driving down the variable costs per kilogram of API produced.
- Cost Reduction in Manufacturing: The implementation of this process eliminates the need for costly cobalt-based reduction systems and reduces the reliance on extensive chromatographic purification steps. By achieving yields of over 90% in the final substitution step and maintaining high purity throughout the sequence, the process minimizes raw material waste and maximizes the output from each batch of expensive Pneumocandin B0 starting material. The removal of heavy metal catalysts also negates the need for specialized scavenging resins or additional washing steps, streamlining the operational expenditure.
- Enhanced Supply Chain Reliability: The robustness of the microwave-assisted steps ensures consistent batch-to-batch quality, reducing the risk of failed batches that can disrupt supply schedules. The use of readily available reagents such as beta-naphthylthiophenol and ethylenediamine, combined with standard solvents like acetonitrile and methanol, ensures that the supply chain is not vulnerable to the shortages of exotic or highly regulated specialty chemicals. This stability allows for more accurate forecasting and inventory management for downstream formulation partners.
- Scalability and Environmental Compliance: Modern microwave reactors are increasingly available in scales suitable for pilot and commercial production, allowing this efficient chemistry to be translated directly to large-scale manufacturing. The process generates less hazardous waste due to the absence of heavy metals and the reduced use of solvents associated with shorter reaction times and fewer purification cycles. This aligns with green chemistry principles and simplifies compliance with increasingly stringent environmental regulations regarding solvent emissions and waste treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and process descriptions found in the source patent documentation, providing a clear overview of the operational parameters and expected outcomes for potential manufacturing partners.
Q: How does microwave irradiation improve the synthesis of Caspofungin Acetate?
A: Microwave irradiation significantly accelerates reaction kinetics, reducing the reaction time for the stereoselective substitution step from 48 hours in conventional methods to approximately 30-35 minutes, while simultaneously improving yield and purity.
Q: What is the advantage of using beta-naphthylthiophenol as a selector?
A: Beta-naphthylthiophenol acts as a superior stereostructure selector compared to traditional thiophenols, facilitating easier reaction rates and higher stereoselectivity, which results in a thioether intermediate with purity exceeding 98%.
Q: Does this process eliminate the need for expensive metal catalysts?
A: Yes, unlike prior art methods that utilize cobalt chloride for reduction, this novel process employs a borane-tetrahydrofuran complex, eliminating the need for expensive transition metals and simplifying the purification workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caspofungin Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for life-saving antifungal medications. Our team of expert chemists has extensively evaluated the microwave-assisted pathway described in CN107778360B and possesses the technical capability to implement this advanced methodology at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity profiles observed in the laboratory can be consistently replicated in our manufacturing facilities. Our state-of-the-art infrastructure includes rigorous QC labs equipped to verify stringent purity specifications, guaranteeing that every batch of Caspofungin Acetate meets the highest global pharmacopeial standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that details how switching to this microwave-enabled process can optimize your specific production economics. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your volume requirements. Let us help you secure a reliable, cost-effective, and high-quality supply of this essential antifungal API.
