Scalable Synthesis of 4-(4-Aminophenyl)morpholine-3-one for Rivaroxaban Production
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical active pharmaceutical ingredient (API) intermediates. A recent technological breakthrough detailed in patent CN115260117A introduces a highly efficient synthesis method for 4-(4-aminophenyl)morpholine-3-one, a pivotal building block in the manufacturing of Rivaroxaban, a widely prescribed oral anticoagulant. As the global demand for cardiovascular therapeutics continues to surge, the ability to produce key intermediates with high atom economy and minimal environmental footprint has become a strategic imperative for supply chain resilience. This novel approach leverages diethylene glycol as a foundational feedstock, bypassing the hazardous reagents and complex purification steps that have historically plagued conventional manufacturing protocols. By integrating oxidative cyclization, acid-catalyzed condensation, and catalytic reduction into a streamlined workflow, this technology offers a compelling solution for reliable pharmaceutical intermediate supplier networks aiming to optimize both cost structures and production safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(4-aminophenyl)morpholine-3-one has been fraught with significant technical and safety challenges that hinder large-scale commercial viability. One prominent prior art route, described in US 7157456, attempts to construct the morpholinone core by reacting ethanolamine with chloroacetyl chloride under alkaline conditions, followed by condensation with para-bromoaniline. However, empirical testing has revealed that this pathway frequently fails to yield the target product in the second step, often necessitating the use of excessive ketone catalysts which introduce problematic heavy metal residues into the final matrix. Another traditional approach utilizes p-bromonitrobenzene as a starting material, yet this method similarly suffers from poor ring-closure efficiency, leading to low overall yields and substantial waste generation.
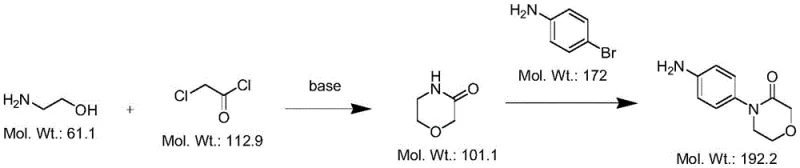
Furthermore, a more recent method employing phthalimide as a raw material presents severe safety hazards during the oxidation phase. This process relies on sodium chlorite, a reagent known for its unstable thermal profile. During reaction monitoring, it was observed that temperatures exceeding 30°C trigger the rapid evolution of toxic yellow-green smoke, indicating a dangerous exothermic event that complicates temperature control and poses significant risks to plant personnel and equipment integrity. These cumulative drawbacks—ranging from failed cyclizations to hazardous oxidation steps—underscore the urgent need for a safer, more reliable alternative in the production of this critical anticoagulant intermediate.
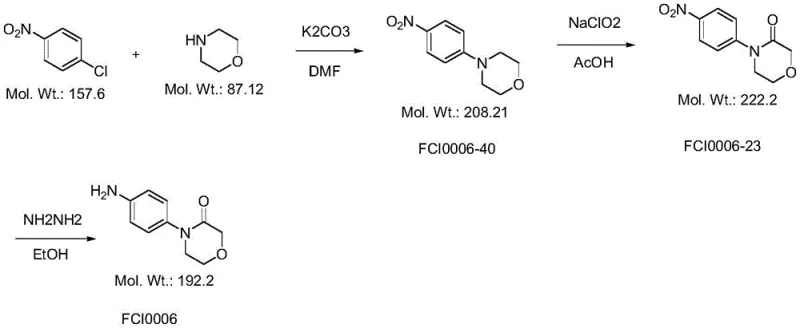
The Novel Approach
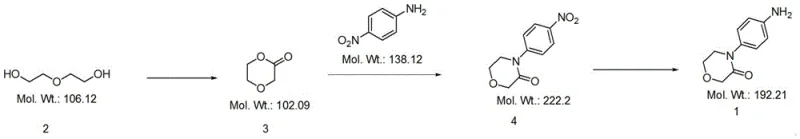
In stark contrast to these problematic legacy methods, the innovation disclosed in CN115260117A establishes a robust three-step sequence that fundamentally reimagines the construction of the morpholinone scaffold. The process initiates with the oxidative cyclization of diethylene glycol, a cheap and abundant commodity chemical, to directly form the lactone ring (Compound 3). This intermediate is then subjected to an acid-catalyzed condensation with p-nitroaniline to install the aromatic moiety, followed by a selective reduction of the nitro group to the corresponding amine. This strategy not only eliminates the need for hazardous sodium chlorite but also avoids the use of heavy metal catalysts in the ring-forming steps, thereby simplifying downstream purification and enhancing the overall purity profile of the final API intermediate.
Mechanistic Insights into Oxidative Cyclization and Catalytic Reduction
The cornerstone of this new synthetic strategy lies in the initial oxidative cyclization of diethylene glycol. Utilizing a catalytic system comprising TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) and an oxidant such as trichloroisocyanuric acid, the reaction selectively oxidizes the terminal hydroxyl groups to facilitate intramolecular esterification, forming the six-membered lactone ring with high precision. This mechanism avoids the over-oxidation or degradation often seen with harsher oxidizing agents, ensuring that the carbon backbone remains intact while establishing the necessary cyclic structure. The subsequent condensation step leverages the electrophilicity of the lactone carbonyl, which is activated by strong organic acids like p-toluenesulfonic acid, allowing the nucleophilic p-nitroaniline to attack and open the ring before reclosing to form the stable morpholinone architecture.
Impurity control is meticulously managed throughout the final reduction stage, where the nitro group is converted to the primary amine. By employing a hydrazine hydrate and ferric chloride system, the reaction proceeds under mild thermal conditions (50-70°C), which prevents the formation of azo-byproducts or hydrodehalogenation side reactions that can occur with more aggressive reducing agents. The use of activated carbon in conjunction with the iron catalyst further aids in adsorbing colored impurities and residual metal species, resulting in a crude product that requires minimal recrystallization to achieve pharmaceutical-grade purity. This mechanistic elegance ensures that the final 4-(4-aminophenyl)morpholine-3-one is produced with a clean impurity profile, essential for meeting the rigorous regulatory standards of the global pharmaceutical market.
How to Synthesize 4-(4-Aminophenyl)morpholine-3-one Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the careful addition of oxidants to diethylene glycol at low temperatures to manage the exotherm, followed by a controlled warm-up to complete the cyclization. The subsequent condensation requires high-temperature reflux in polar aprotic solvents to drive the equilibrium toward the desired product, while the final reduction must be monitored to ensure complete conversion of the nitro group without affecting the sensitive lactone ring. For laboratory and pilot-scale operations, adhering to the specific stoichiometric ratios and temperature profiles outlined in the patent is critical for reproducibility.
- Perform oxidative cyclization on diethylene glycol using an oxidant such as TEMPO and trichloroisocyanuric acid to obtain the lactone intermediate (Compound 3).
- React Compound 3 with p-nitroaniline in the presence of an acid catalyst like p-toluenesulfonic acid at elevated temperatures to form the nitro-substituted morpholinone (Compound 4).
- Reduce the nitro group of Compound 4 using a reducing system such as hydrazine hydrate with ferric chloride catalyst to yield the final target product 4-(4-aminophenyl)morpholine-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple unit cost metrics. By shifting the raw material base to diethylene glycol, manufacturers can decouple their supply chains from the volatility associated with specialized halogenated reagents or complex phthalimide derivatives. This transition to commodity feedstocks inherently stabilizes sourcing logistics, ensuring consistent availability even during periods of global chemical supply disruption. Moreover, the elimination of hazardous oxidation steps reduces the regulatory burden and insurance costs associated with handling dangerous goods, contributing to a more resilient and compliant operational framework.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the utilization of low-cost starting materials and the simplification of the purification train. By avoiding expensive heavy metal catalysts and hazardous oxidants like sodium chlorite, the process significantly lowers the cost of goods sold (COGS). Furthermore, the high atom economy of the oxidative cyclization step minimizes waste disposal fees, while the efficient reduction protocol reduces solvent consumption and energy usage during workup, collectively driving down the total manufacturing cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Sourcing diethylene glycol and p-nitroaniline is far less risky than procuring specialized brominated precursors or phthalimide derivatives, as these commodities are produced at massive scales globally. This abundance ensures that production schedules are not held hostage by the lead times of niche chemical suppliers. Additionally, the robustness of the reaction conditions means that the process is less susceptible to batch failures caused by minor variations in reagent quality, thereby guaranteeing a steady flow of high-purity intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a significant advancement in green chemistry principles. The avoidance of toxic gas evolution and the reduction of heavy metal waste streams simplify the wastewater treatment process and lower the facility's environmental footprint. This alignment with sustainability goals not only facilitates easier permitting for capacity expansion but also enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain, appealing to multinational corporations with strict vendor codes of conduct.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of safety, yield, and scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for commercial production.
Q: What are the primary safety advantages of this new synthesis route compared to prior art?
A: Unlike previous methods that utilize sodium chlorite for oxidation—a reagent known to release hazardous yellow-green smoke and generate difficult-to-control exothermic reactions—this novel process employs safer oxidants like TEMPO/trichloroisocyanuric acid. This significantly mitigates thermal runaway risks and improves operational safety during industrial scale-up.
Q: Why is diethylene glycol preferred as a starting material over ethanolamine or phthalimide?
A: Diethylene glycol is a commodity chemical that is inexpensive and readily available in bulk quantities. Furthermore, it allows for a direct oxidative cyclization to form the morpholinone core in a single step, avoiding the complex multi-step sequences and heavy metal catalyst residues associated with ethanolamine or phthalimide-based routes.
Q: How does this method impact the purity profile of the final anticoagulant intermediate?
A: The process utilizes mild reduction conditions (hydrazine hydrate/ferric chloride) and avoids harsh heavy metal catalysts in the final steps. This results in a cleaner reaction profile with fewer metallic impurities, facilitating easier purification and ensuring the final product meets stringent pharmaceutical quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Aminophenyl)morpholine-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation anticoagulants depends on the availability of high-quality, cost-effective intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering 4-(4-aminophenyl)morpholine-3-one with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch against the highest international standards.
We invite you to collaborate with us to leverage this innovative synthesis route for your Rivaroxaban supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the confidence of a secure and optimized supply foundation.
