Scalable Synthesis of Rivaroxaban Intermediate 4-(4-Aminophenyl)Morpholine-3-One for Global API Production
The pharmaceutical industry's relentless demand for efficient anticoagulant therapies has placed significant pressure on the supply chains of key intermediates like 4-(4-aminophenyl)morpholine-3-one, a critical precursor for Rivaroxaban (CAS: 366789-02-8). As detailed in the recent patent disclosure CN114380763A, a novel synthetic methodology has emerged that fundamentally restructures the production landscape for this high-value molecule. This technical breakthrough addresses long-standing bottlenecks in process stability and environmental compliance, offering a robust alternative to legacy routes that have plagued manufacturers with high waste generation and unpredictable impurity profiles. By leveraging a streamlined five-step sequence starting from 2-haloethoxyethanol, this innovation delivers a pathway characterized by exceptional operational simplicity and economic viability. For global stakeholders, this represents a pivotal shift towards more sustainable and cost-effective pharmaceutical intermediate manufacturing, ensuring that the supply of this vital anticoagulant component remains secure and scalable for future market demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
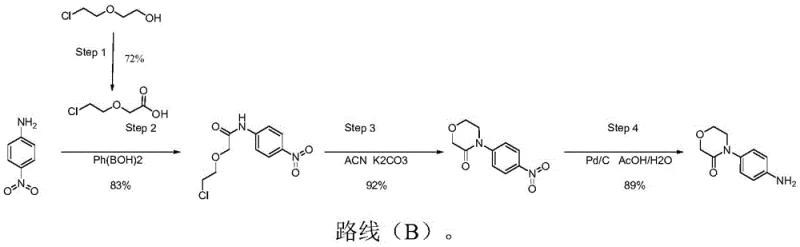
Historically, the synthesis of 4-(4-aminophenyl)morpholine-3-one has relied on routes exemplified by patent WO2019138362A1, which utilize p-nitroaniline and 2-chloroethoxyethanol but suffer from severe process inefficiencies. As illustrated in the conventional Route (B), the second step necessitates the use of phenylboronic acid, a reagent that is not only prohibitively expensive but also subject to volatile market availability and limited production capacity. Furthermore, the reliance on acetonitrile as a solvent in subsequent cyclization steps creates a significant downstream burden; acetonitrile is fully miscible with water, making the separation and recovery of solvents during post-treatment exceptionally energy-intensive and costly. These factors combine to create a process with poor stability, where the generation of uncontrollable impurities poses a constant risk to product quality, ultimately rendering such methods unsuitable for large-scale industrial application where consistency is paramount.
The Novel Approach
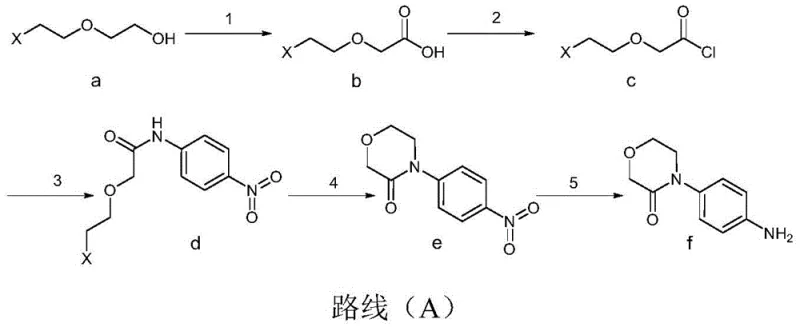
In stark contrast, the novel Route (A) disclosed in the present invention introduces a paradigm shift by utilizing 2-haloethoxyethanol as a foundational building block, proceeding through a logical sequence of oxidation, chlorination, acylation, ring closure, and reduction. This approach elegantly bypasses the need for expensive coupling reagents like phenylboronic acid, instead employing cost-effective oxidants such as sodium hypochlorite in the initial step to generate the carboxylic acid intermediate with high efficiency. The strategic selection of oxalyl chloride for the chlorination step ensures that no sulfur residues are introduced, thereby protecting the integrity of the catalyst in the final reduction stage. Moreover, the process allows for the direct concentration of reaction solutions between steps, eliminating tedious isolation procedures and significantly reducing the overall processing time. This streamlined architecture not only enhances the total yield but also drastically simplifies the operational workflow, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Cyclization
The core of this synthetic innovation lies in the precise control of reaction conditions to maximize chemoselectivity and minimize byproduct formation. The initial oxidation step employs a TEMPO/sodium hypochlorite system, a green chemistry approach that operates effectively at mild temperatures (0-60°C) to convert the primary alcohol to the corresponding carboxylic acid without over-oxidation or degradation of the ether linkage. This is followed by a chlorination reaction using oxalyl chloride, which activates the acid for nucleophilic attack while avoiding the introduction of sulfur contaminants that could irreversibly poison the palladium catalyst used later. The subsequent acylation with p-nitroaniline, catalyzed by DMAP, proceeds smoothly to form the amide bond, which then undergoes intramolecular cyclization in the presence of a base like triethylamine. This cascade effectively constructs the morpholine-3-one core with high regioselectivity, ensuring that the nitrogen atom is correctly positioned for the final pharmacological activity. The mechanistic robustness of this sequence ensures that impurity levels remain low, typically achieving purities around 98% before the final reduction.
Impurity control is further enhanced by the choice of solvents and reagents throughout the pathway. Unlike previous methods that utilized acetonitrile-water mixtures which complicate extraction, this protocol favors dichloromethane and methanol, solvents that offer distinct phase separation properties and ease of removal. The final reduction step utilizes palladium on carbon under hydrogen atmosphere, a standard yet highly effective method for nitro group reduction. Crucially, because the preceding chlorination step avoided sulfur-based reagents, the Pd/C catalyst maintains its activity throughout the reaction, preventing the accumulation of partially reduced intermediates or dehalogenated byproducts. This attention to catalytic compatibility ensures that the final high-purity pharmaceutical intermediate meets stringent quality specifications required for downstream API synthesis, reducing the need for extensive recrystallization or chromatographic purification.
How to Synthesize 4-(4-Aminophenyl)Morpholine-3-One Efficiently
The implementation of this synthesis route requires careful adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process begins with the oxidation of 2-chloroethoxyethanol, followed by activation and coupling, culminating in a clean hydrogenation step. Each stage has been rigorously tested to identify optimal molar ratios, temperatures, and reaction times, allowing for a seamless transition from laboratory bench to pilot plant. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-efficiency process. For the detailed standardized synthesis steps, please refer to the guide below.
- Oxidation of 2-haloethoxyethanol using sodium hypochlorite and TEMPO catalyst to form the carboxylic acid intermediate.
- Chlorination of the acid intermediate using oxalyl chloride to generate the acyl chloride without sulfur contamination.
- Acylation with p-nitroaniline followed by base-mediated cyclization to form the morpholine ring structure.
- Catalytic hydrogenation using Pd/C in methanol to reduce the nitro group to the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. By fundamentally altering the raw material input and processing logic, the method addresses critical pain points related to cost volatility and logistical complexity. The elimination of scarce and expensive reagents like phenylboronic acid immediately stabilizes the bill of materials, shielding production budgets from market fluctuations. Furthermore, the simplification of workup procedures—specifically the ability to telescope reactions without intermediate isolation—drastically reduces labor hours and equipment occupancy time. These operational efficiencies translate directly into a more resilient supply chain capable of meeting tight delivery schedules without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of high-cost reagents with commodity chemicals and the reduction of solvent consumption. By replacing phenylboronic acid with inexpensive sodium hypochlorite and oxalyl chloride, the raw material cost per kilogram of product is significantly lowered. Additionally, the avoidance of acetonitrile eliminates the need for complex distillation setups required to separate it from water, thereby reducing energy consumption and waste treatment fees. The ability to concentrate reaction mixtures and proceed directly to the next step without purification further cuts down on solvent usage and loss, resulting in substantial overall cost savings for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on readily available starting materials such as 2-chloroethoxyethanol and p-nitroaniline ensures a stable and continuous supply of inputs. Unlike specialized coupling agents that may have single-source suppliers or long lead times, these bulk chemicals are produced globally in vast quantities, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further enhancing the reliability of the manufacturing output. This stability is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to conventional methods, aligning with increasingly strict global regulations on industrial emissions. The use of green oxidants and the minimization of heavy metal waste reduce the burden on wastewater treatment facilities, lowering the environmental compliance costs associated with production. The process generates fewer solid wastes and avoids the use of toxic sulfur-containing byproducts, making it safer for operators and the surrounding community. This eco-friendly design facilitates easier permitting for new production lines and supports the long-term sustainability goals of modern chemical enterprises, ensuring that commercial scale-up can proceed without regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on why this route is superior for industrial applications. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new process for their own production lines.
Q: Why is oxalyl chloride preferred over thionyl chloride in this synthesis?
A: Oxalyl chloride is selected to prevent sulfur contamination, which would otherwise poison the palladium on carbon catalyst used in the final hydrogenation step, ensuring consistent reaction kinetics and yield.
Q: How does this new route improve environmental compliance compared to prior art?
A: The process replaces expensive phenylboronic acid reagents and avoids acetonitrile-water mixtures that are difficult to separate, significantly reducing three-waste treatment costs and simplifying solvent recovery.
Q: What represents the key cost-saving advantage for procurement teams?
A: The use of readily available 2-haloethoxyethanol as a starting material and the elimination of complex post-treatment steps for intermediate isolation drastically lowers raw material and operational expenditures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Aminophenyl)Morpholine-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving anticoagulants depends on the uninterrupted supply of high-quality intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-(4-aminophenyl)morpholine-3-one performs consistently in your downstream synthesis. Our infrastructure is designed to handle complex organic transformations with precision, making us a trusted partner for global pharmaceutical companies seeking reliability and excellence.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your production economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the anticoagulant market through superior chemical manufacturing.
