Advanced Chiral Resolution Strategy for Scalable Optically Pure Tetrandrine Production
Advanced Chiral Resolution Strategy for Scalable Optically Pure Tetrandrine Production
The pharmaceutical industry constantly seeks robust synthetic routes for complex natural products, and the total synthesis of optically pure tetrandrine represents a significant challenge due to its intricate bisbenzylisoquinoline structure. Patent CN111440121B introduces a groundbreaking method for the chemical resolution of two critical racemic intermediates, enabling the efficient production of the S-configuration required for biological activity. This technology shifts the paradigm from low-yield plant extraction to high-precision chemical synthesis, addressing the global demand for this potent calcium channel blocker and calmodulin antagonist. By leveraging specific chiral resolving agents, manufacturers can now access high-purity intermediates that were previously difficult to obtain in bulk quantities.
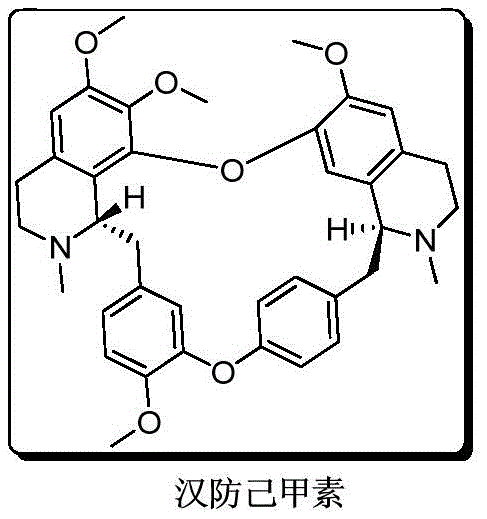
Tetrandrine, a bioactive alkaloid extracted from Stephania tetrandra, possesses remarkable pharmacological properties including anticancer and antifibrotic effects, yet its natural abundance is critically low at approximately 1% of the dry root weight. Conventional extraction methods are plagued by inefficiency, massive solvent consumption, and inconsistent quality, making them unsuitable for modern large-scale pharmaceutical manufacturing. The transition to total synthesis offers a sustainable alternative, but the creation of the two essential chiral centers with S-configuration remains the bottleneck. The disclosed invention solves this by providing a reliable pathway to resolve racemic precursors into their optically active forms with exceptional stereocontrol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of tetrahydroisoquinoline derivatives has relied on classical resolving agents such as camphorsulfonic acid, tartaric acid, or mandelic acid, which often fail when applied to the specific steric environment of tetrandrine intermediates. As illustrated by the structural diversity in prior art, minor changes in the molecular framework can drastically alter the crystallization behavior of diastereomeric salts, rendering standard protocols ineffective. Experimental data indicates that using these traditional acids results in poor enantiomeric excess values, often below 40%, and necessitates repeated recrystallization cycles that destroy overall yield. Furthermore, the reliance on multiple different solvents and agents for different steps complicates the waste stream and increases the environmental footprint of the process.
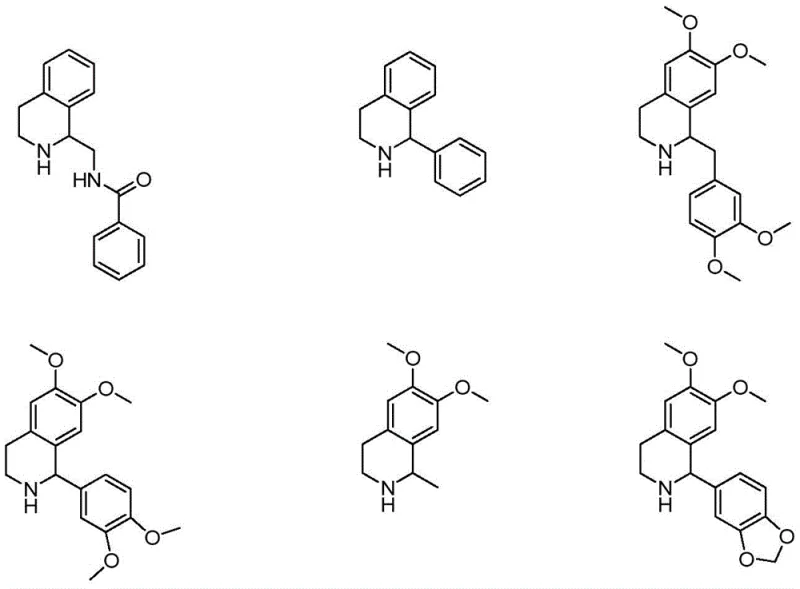
The Novel Approach
The innovative strategy presented in this patent utilizes N-acetyl amino acids, specifically N-Ac-L-cysteine, to achieve unprecedented resolution efficiency for both key intermediates simultaneously. Unlike the rigid structures of traditional acids, the flexible side chain of N-Ac-L-cysteine allows for optimal spatial arrangement during salt formation, facilitating the selective crystallization of the desired S-enantiomer. This approach eliminates the need for mixed resolving agents or complex solvent systems, as a single agent and a common solvent like ethanol can drive the reaction to completion. The result is a streamlined process that delivers yields exceeding 80% with optical purity surpassing 99%, effectively bypassing the limitations that have hindered previous synthetic efforts.
Mechanistic Insights into N-Ac-L-Cysteine Mediated Chiral Resolution
The core of this technological breakthrough lies in the formation of highly stable diastereomeric salts between the basic nitrogen of the tetrahydroisoquinoline ring and the carboxylic acid group of the chiral resolver. When N-Ac-L-cysteine interacts with the racemic mixture, it creates two distinct diastereomers with different solubility profiles in polar protic solvents like ethanol. The thermodynamic stability of the salt formed with the S-configuration intermediate is significantly higher under the specified crystallization conditions, promoting its precipitation while the unwanted R-enantiomer remains in the mother liquor. This selective precipitation is driven by specific hydrogen bonding networks and steric interactions that are unique to the cysteine derivative, which are not replicated by simpler amino acids or organic acids.
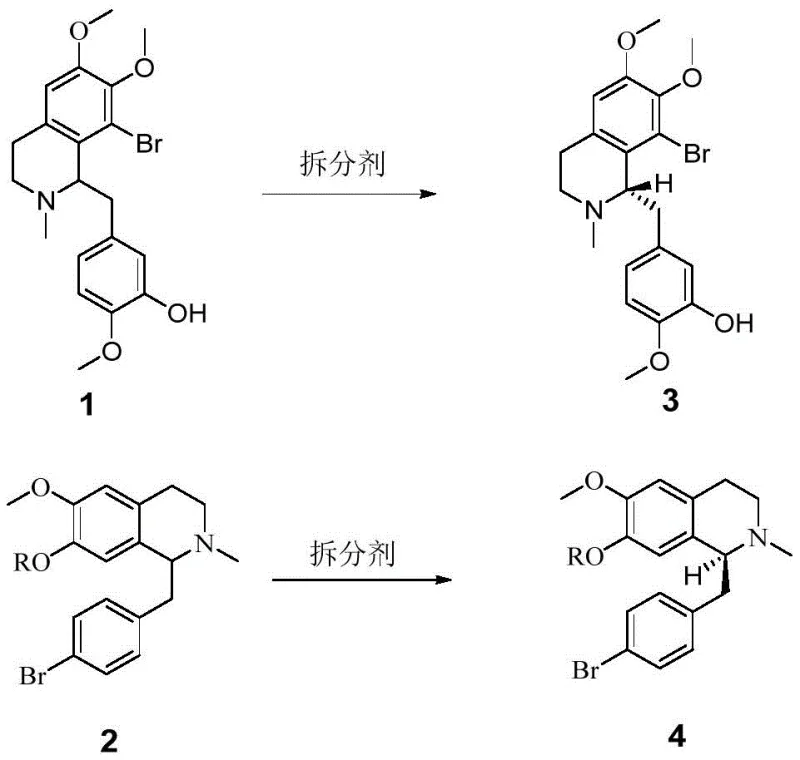
Impurity control is inherently built into this mechanism through the rigorous washing and refining steps that follow the initial salt formation. The process involves a hot filtration step where the crude salt is washed with warm ethanol, effectively dissolving any co-crystallized impurities or the minor enantiomer that may have been mechanically trapped. Subsequent dissociation of the purified salt using a mild base like sodium hydroxide ensures that the chiral integrity of the molecule is maintained without risking racemization. This multi-stage purification protocol guarantees that the final free base intermediates meet the stringent purity specifications required for downstream coupling reactions in the total synthesis of tetrandrine.
How to Synthesize Optically Pure Tetrandrine Intermediates Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for converting readily available racemic starting materials into high-value chiral building blocks. The procedure is designed to be operationally simple, requiring standard laboratory equipment and avoiding extreme temperatures or pressures that would complicate scale-up. By adhering to the specific molar ratios and solvent volumes defined in the examples, manufacturers can reproduce the high yields and optical purities reported in the intellectual property. The detailed standardized synthesis steps below outline the precise conditions for salt formation, refining, and dissociation to ensure consistent batch-to-batch quality.
- Dissolve racemic Compound 1 or 2 in ethanol and add the chiral resolving agent N-Ac-L-cysteine at room temperature.
- Crystallize the resulting diastereomeric salt at 0°C for 12-24 hours to isolate the desired stereoisomer.
- Perform base dissociation using sodium hydroxide and dichloromethane extraction to recover the free base with >99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this resolution technology offers substantial strategic benefits by simplifying the raw material portfolio and reducing processing complexity. The ability to use a single resolving agent, N-Ac-L-cysteine, for two different critical intermediates consolidates purchasing requirements and minimizes the risk of supply disruptions associated with sourcing multiple specialized chiral acids. Additionally, the preference for ethanol as the primary solvent aligns with green chemistry principles, reducing the costs associated with hazardous waste disposal and solvent recovery compared to processes relying on chlorinated or aromatic solvents. This operational simplicity translates directly into a more resilient and cost-effective supply chain for the production of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and inefficient resolving agents like dibenzoyl-tartaric acid significantly lowers the direct material costs per kilogram of product. By achieving high yields in a single resolution step rather than requiring multiple recrystallizations, the process reduces energy consumption and labor hours associated with extended processing times. The recovery and reuse of the mother liquor containing the unwanted enantiomer further enhance the economic viability of the process by maximizing atom economy.
- Enhanced Supply Chain Reliability: Utilizing commercially abundant amino acid derivatives as resolving agents ensures a stable supply base that is not subject to the volatility of niche chemical markets. The robustness of the reaction conditions, which proceed effectively at room temperature or mild heating, reduces the dependency on specialized high-pressure or cryogenic equipment, thereby increasing the number of qualified contract manufacturing organizations capable of executing the synthesis. This flexibility allows for faster scaling and more reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The use of ethanol, a benign and renewable solvent, simplifies regulatory compliance regarding volatile organic compound emissions and worker safety. The high efficiency of the resolution means less waste is generated per unit of product, supporting corporate sustainability goals and reducing the burden on wastewater treatment facilities. The process is inherently scalable from gram-level laboratory optimization to multi-ton industrial production without significant re-engineering of the unit operations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this chiral resolution technology in an industrial setting. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their own tetrandrine synthesis projects. The answers are derived directly from the experimental data and comparative examples provided in the patent documentation to ensure accuracy and relevance.
Q: Why are traditional resolving agents like tartaric acid ineffective for these Tetrandrine intermediates?
A: Traditional agents such as tartaric acid or mandelic acid result in poor resolution efficiency and low optical purity for these specific bisbenzylisoquinoline structures due to insufficient steric differentiation during salt formation.
Q: What is the optical purity achieved using the N-Ac-L-cysteine resolution method?
A: The patented method consistently achieves an enantiomeric excess (ee) value of greater than 99%, ensuring the production of high-quality intermediates suitable for final API synthesis.
Q: Can the same resolving agent be used for both key intermediates in the synthesis?
A: Yes, N-Ac-L-cysteine serves as a universal resolving agent for both Compound 1 and Compound 2, significantly simplifying the supply chain and waste treatment processes compared to using multiple different agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrandrine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to verify the enantiomeric excess of every batch we produce. Our commitment to quality assurance guarantees that the tetrandrine intermediates we supply meet the exacting standards required for clinical and commercial applications.
We invite potential partners to contact our technical procurement team to discuss how this advanced resolution technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this synthetic route. We are prepared to provide specific COA data and route feasibility assessments to support your regulatory filings and process validation efforts, ensuring a successful partnership focused on innovation and reliability.
