Scalable Total Synthesis of Racemic Tetrandrine for Commercial API Manufacturing
Scalable Total Synthesis of Racemic Tetrandrine for Commercial API Manufacturing
The pharmaceutical industry is constantly seeking robust, scalable routes for complex alkaloids that traditionally rely on unsustainable extraction methods. A pivotal advancement in this domain is detailed in patent CN113045578A, which discloses a novel total synthesis method for racemic tetrandrine. This bisbenzylisoquinoline alkaloid, historically extracted from the root tuber of Stephania tetrandra, possesses potent pharmacological properties including anti-inflammatory, anti-silicosis, and anti-tumor activities. However, natural extraction is plagued by a content yield of only about 1%, leading to excessive raw material consumption and severe environmental discharge. The new synthetic approach described in the patent utilizes cheap and easily obtainable starting materials, specifically 5-bromovanillin and 4-hydroxyphenylacetic acid, to construct the complex molecular architecture through a concise and efficient pathway.
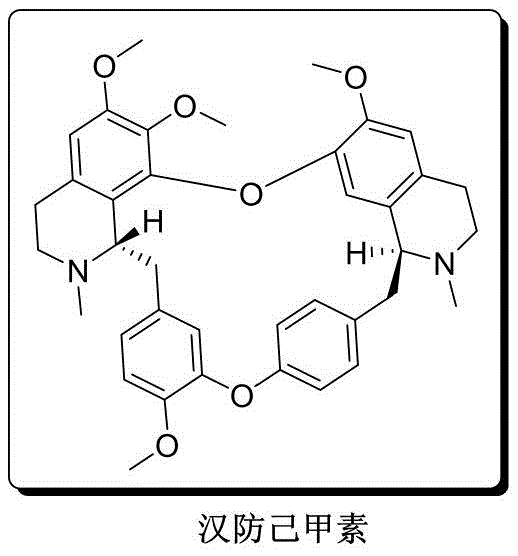
This technological breakthrough represents a significant shift from linear, low-yield academic syntheses to a convergent strategy optimized for industrial feasibility. By establishing a reliable pharmaceutical intermediate supplier capability for such complex molecules, manufacturers can secure the supply chain for downstream API production. The method not only addresses the scarcity of natural sources but also provides a platform for producing optical pure derivatives in the future, laying a critical foundation for the development of next-generation calcium antagonists and anti-fibrotic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of tetrandrine has been bottlenecked by the inefficiencies of plant extraction and the impracticality of early synthetic routes. Industrial extraction requires processing vast quantities of plant matter to isolate minute amounts of the active ingredient, resulting in high costs and substantial ecological footprints due to solvent waste. On the synthetic front, prior art such as the linear strategy reported in J. Chem. Soc. (1969) suffered from excessively long reaction sequences (14 steps) and harsh conditions that exceeded solvent boiling points, yielding a dismal total yield of approximately 1%. Furthermore, more recent academic approaches, while shorter, often rely on expensive starting materials and dangerous reagents like lithium diisopropylamide or lithium aluminum hydride, which pose significant safety risks and cost barriers for cost reduction in pharmaceutical intermediates manufacturing. These factors render traditional methods unsuitable for the commercial scale-up of complex alkaloids required by the global market.
The Novel Approach
The methodology outlined in CN113045578A overcomes these hurdles through a cleverly designed convergent synthesis that maximizes atom economy and operational simplicity. Instead of a long linear chain, the route synthesizes a versatile common intermediate, Compound 10, which is then divergently processed into two key fragments, Compounds 12 and 14. These fragments are subsequently united through strategic intermolecular and intramolecular Ullmann reactions to form the target macrocycle. This approach drastically simplifies the operational complexity, allowing for the use of milder conditions and recyclable solvents. By avoiding the need for cryogenic temperatures or pyrophoric reagents in the critical coupling steps, the process enhances safety and reduces the barrier to entry for large-scale production, effectively solving the supply continuity issues associated with botanical sourcing.
Mechanistic Insights into Copper-Catalyzed Ullmann Macrocyclization
The core technical innovation of this synthesis lies in the construction of the diaryl ether linkages that define the bisbenzylisoquinoline skeleton. The process employs a copper-catalyzed Ullmann-type coupling, a robust method for forming carbon-oxygen bonds between aryl halides and phenols. In the penultimate stages, Compound 12 (containing a phenolic hydroxyl group) and Compound 14 (containing an aryl bromide) undergo an intermolecular Ullmann reaction under alkaline and high-temperature conditions. This step is critical as it links the two halves of the molecule, requiring precise control over catalyst loading (e.g., cuprous iodide) and ligand selection (e.g., N,N-dimethylglycine) to ensure high selectivity and minimize homocoupling byproducts. The subsequent intramolecular Ullmann reaction closes the second ring, finalizing the macrocyclic structure of racemic tetrandrine.
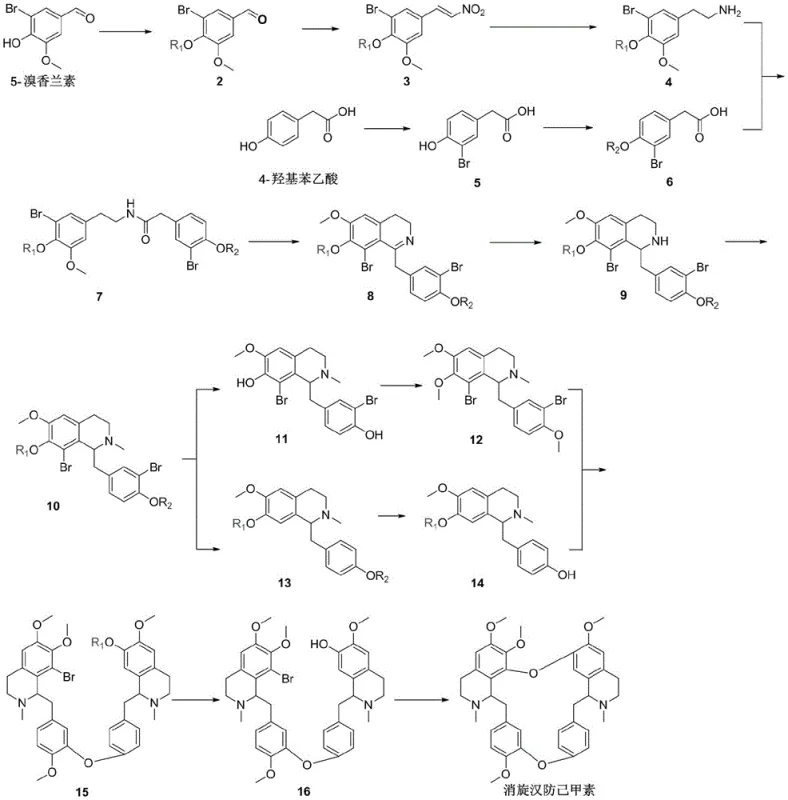
From an impurity control perspective, the choice of protecting groups plays a vital role in the purity profile of the final high-purity pharmaceutical intermediate. The patent details the use of benzyl or p-toluenesulfonyl groups to protect hydroxyl functionalities during the early condensation and cyclization steps (forming the tetrahydroisoquinoline rings). These groups are stable enough to withstand the rigorous conditions of the Bischler-Napieralski type cyclization (using phosphorus oxychloride) yet can be cleanly removed under specific acidic or alkaline conditions later in the sequence. This orthogonal protection strategy ensures that the reactive phenolic sites are only exposed when needed for the final Ullmann coupling, thereby preventing premature polymerization or side reactions that could compromise the quality of the final API precursor.
How to Synthesize Racemic Tetrandrine Efficiently
The synthesis protocol described in the patent offers a streamlined pathway that balances yield with operational ease, making it an ideal candidate for technology transfer. The process begins with the protection and functionalization of 5-bromovanillin, followed by a Henry reaction and reduction to generate the amine component. Simultaneously, 4-hydroxyphenylacetic acid is brominated and protected to form the acid component. These two streams converge via amide condensation, followed by dehydration and reduction to establish the tetrahydroisoquinoline core. For the detailed standardized operating procedures, including specific molar ratios, temperature profiles, and workup protocols for each of the 16 steps, please refer to the technical guide below.
- Synthesize the bis-tetrahydroisoquinoline core (Compound 10) by condensing protected 5-bromovanillin derivatives with brominated phenylacetic acid precursors, followed by cyclization and methylation.
- Diverge Compound 10 into two key fragments: Compound 12 (via deprotection and methylation) and Compound 14 (via debromination and deprotection).
- Execute the final macrocyclization through sequential intermolecular and intramolecular Ullmann etherifications using copper catalysts to form the bisbenzylisoquinoline skeleton.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to this specific synthetic route offers profound strategic benefits beyond mere technical feasibility. The primary advantage is the decoupling of production from agricultural variables; unlike plant extraction, which is subject to seasonal fluctuations, climate change, and geopolitical instability in sourcing regions, this chemical synthesis relies on bulk commodity chemicals. 5-bromovanillin and 4-hydroxyphenylacetic acid are widely available in the global fine chemical market, ensuring a stable and predictable supply chain. This reliability is crucial for maintaining continuous API manufacturing schedules and mitigating the risk of raw material shortages that frequently plague botanical supply chains.
- Cost Reduction in Manufacturing: The economic model of this synthesis is driven by the utilization of low-cost starting materials and the elimination of expensive, hazardous reagents found in previous academic routes. By avoiding reagents like lithium aluminum hydride in favor of safer, cheaper alternatives like sodium borohydride or catalytic hydrogenation where applicable, the direct material costs are significantly lowered. Furthermore, the patent highlights the recyclability of solvents such as dichloromethane, ethanol, and DMF. In an industrial setting, solvent recovery systems can capture and reuse the majority of these liquids, drastically reducing both the purchase cost of fresh solvents and the disposal costs associated with hazardous waste treatment, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: The convergent nature of the synthesis allows for the parallel production of intermediates. Compounds 12 and 14 can be synthesized in separate reactors simultaneously before being combined in the final coupling steps. This parallelization effectively shortens the critical path of the manufacturing timeline compared to a strictly linear sequence. Additionally, the robustness of the Ullmann coupling conditions means the process is less sensitive to minor variations in temperature or moisture, reducing the likelihood of batch failures. This consistency translates to higher on-time delivery rates for customers relying on a reliable pharmaceutical intermediate supplier for their drug development pipelines.
- Scalability and Environmental Compliance: As regulatory pressure on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key decision factor. This method significantly reduces the "three wastes" (waste water, waste gas, waste residue) compared to plant extraction, which generates massive amounts of biomass waste. The chemical process generates defined organic waste streams that are easier to treat and incinerate. Moreover, the avoidance of heavy metal catalysts in the early stages (using organic bases and mild dehydrating agents instead) simplifies the purification process, reducing the burden on downstream heavy metal scavenging units and ensuring the final product meets stringent ICH Q3D guidelines for elemental impurities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the feasibility and advantages for potential partners.
Q: Why is chemical synthesis preferred over plant extraction for Tetrandrine?
A: Plant extraction yields are extremely low (approximately 1%), requiring massive raw material inputs and generating significant environmental waste. Chemical synthesis offers a continuous, scalable alternative with lower costs and better supply chain stability.
Q: What are the critical reaction steps in this novel synthesis route?
A: The route relies on a convergent strategy where a common intermediate (Compound 10) is split into two fragments. The key technological breakthrough lies in the final intermolecular and intramolecular Ullmann reactions that close the macrocyclic structure efficiently.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the patent emphasizes the use of cheap, commercially available starting materials like 5-bromovanillin and avoids hazardous reagents like lithium aluminum hydride where possible, making it highly feasible for large-scale pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Racemic Tetrandrine Supplier
The novel total synthesis of racemic tetrandrine represents a paradigm shift in how complex bisbenzylisoquinoline alkaloids can be manufactured for the global market. At NINGBO INNO PHARMCHEM, we recognize the immense potential of this route to stabilize the supply of this critical therapeutic agent. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with the specialized reactors and containment systems necessary to handle the high-temperature Ullmann coupling steps safely and efficiently. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced HPLC and NMR techniques to verify the structural integrity and impurity profile of every batch.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize this pathway further or to initiate custom synthesis projects. Whether you require gram-scale quantities for preclinical studies or metric tons for commercial launch, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how we can support your supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
