Scalable Convergent Synthesis of Optically Pure Tetrandrine for Global Pharmaceutical Supply
Introduction to Advanced Tetrandrine Manufacturing
The pharmaceutical industry continuously seeks robust, scalable alternatives to natural product extraction, particularly for complex alkaloids like tetrandrine. Patent CN111518108B introduces a groundbreaking total synthesis method for optically pure tetrandrine, addressing critical supply chain vulnerabilities associated with plant-based sourcing. This technology leverages a convergent synthesis strategy that drastically reduces reaction steps while maintaining exceptional stereochemical integrity. By shifting from a dependency on Stephania tetrandra root extraction, which suffers from low alkaloid content and seasonal variability, this chemical approach ensures a consistent, high-purity supply of this potent anti-cancer and anti-inflammatory agent. The methodology represents a significant leap forward in process chemistry, transforming a historically difficult natural product into a reliably manufacturable fine chemical intermediate.
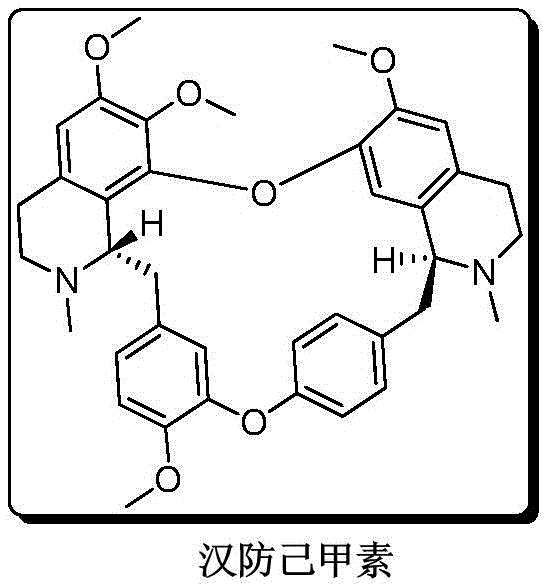
For R&D directors and procurement specialists, the implications of this patent are profound. It moves the production paradigm from an agricultural bottleneck to a controlled chemical engineering environment. The ability to synthesize the target molecule with yields reaching up to 38.9%—a tens-fold improvement over prior art—suggests a dramatic reduction in cost of goods sold (COGS). Furthermore, the preservation of optical purity (ee > 99%) throughout the synthetic sequence eliminates the need for costly downstream chiral separations, streamlining the path to commercial API production. This report analyzes the technical merits and commercial viability of this novel route, positioning it as a cornerstone for future tetrandrine supply strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of tetrandrine has been plagued by inefficiencies inherent to natural product extraction. The root of Stephania tetrandra contains a total alkaloid content of merely 1.5-2.3%, with tetrandrine itself constituting only about 1% of the dry weight. This scarcity necessitates the processing of massive quantities of raw plant material, leading to severe ecological pressure and supply instability. From a manufacturing perspective, the extraction process is operationally complex, requiring extensive solvent usage and generating substantial hazardous waste, which complicates environmental compliance. Moreover, previous attempts at total chemical synthesis were equally problematic; existing literature describes linear routes spanning nine distinct steps with a dismal overall yield of approximately 1.67%. These legacy methods produced racemic mixtures rather than the biologically active enantiomer, rendering them commercially non-viable for high-value pharmaceutical applications where specific chirality dictates efficacy.
The Novel Approach
The methodology disclosed in CN111518108B revolutionizes this landscape through a 'convergent' synthesis design. Instead of a long, fragile linear chain, the process builds the complex bis-benzylisoquinoline skeleton by coupling two advanced chiral intermediates. This strategic shift reduces the synthesis from the starting chiral fragments to the final product to just three high-efficiency steps. The core of this innovation lies in the sequential application of Ullmann coupling reactions—first intermolecular to link the two halves, and subsequently intramolecular to close the macrocyclic ether bridge. This approach not only simplifies the operational workflow but also inherently boosts the cumulative yield. By minimizing the number of unit operations, the process reduces material loss at each stage, directly translating to higher throughput and lower waste generation. The result is a scalable, gram-scale capable protocol that delivers the natural optically pure configuration without the need for resolution at the final stage.
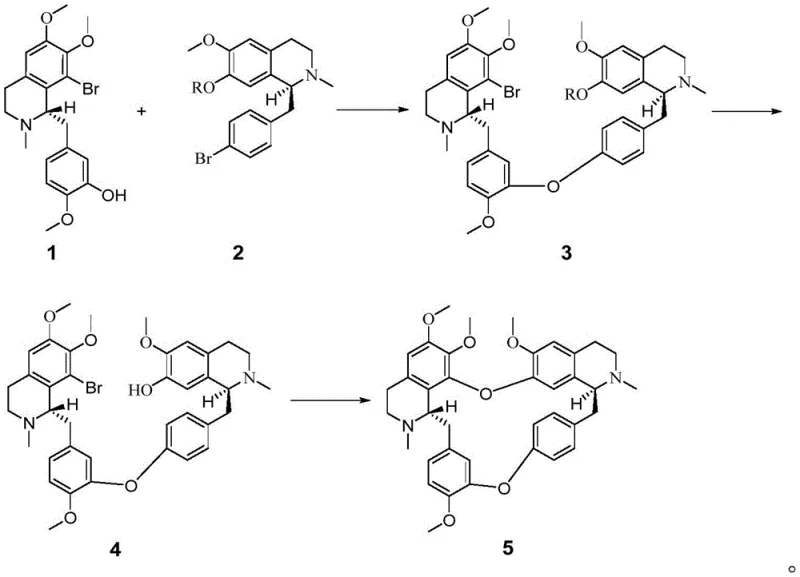
Mechanistic Insights into Copper-Catalyzed Ullmann Coupling
The success of this synthetic route hinges on the precise execution of copper-catalyzed Ullmann etherifications, a transformation renowned for constructing aryl-oxygen bonds. In the first critical step, an intermolecular Ullmann reaction joins Compound 1 and Compound 2. This reaction utilizes a copper(I) source, such as cuprous bromide, in conjunction with a base like potassium carbonate in a high-boiling solvent like toluene. The mechanism likely involves the oxidative addition of the aryl halide to the copper center, followed by ligand exchange with the phenoxide and subsequent reductive elimination to form the diaryl ether linkage. The choice of ligands and solvent polarity is crucial here to facilitate the coupling of these sterically hindered substrates without compromising the sensitive chiral centers located on the isoquinoline rings. The robustness of this catalytic system allows the reaction to proceed at elevated temperatures (around 100°C) to overcome activation barriers, ensuring complete conversion of the starting materials.
Following the initial coupling and a straightforward acidic deprotection to unveil the second phenolic handle, the synthesis culminates in an intramolecular Ullmann cyclization. This step is mechanistically more demanding due to the entropic penalty of forming a large macrocyclic ring. To drive this equilibrium, the process employs stronger bases like cesium carbonate and polar aprotic solvents such as DMF or DMSO, often with cuprous iodide as the catalyst. The high dilution principles typically associated with macrocyclization are managed here through optimized concentration and extended reaction times (up to 80 hours), allowing the flexible linker to adopt the necessary conformation for ring closure. Crucially, the reaction conditions are tuned to be chemoselective, avoiding racemization of the chiral benzylic positions. This meticulous control over the catalytic cycle ensures that the final product retains the high enantiomeric excess established in the starting materials, delivering a pharmacologically active substance ready for biological evaluation.
How to Synthesize Optically Pure Tetrandrine Efficiently
The implementation of this synthesis requires strict adherence to the optimized parameters regarding catalyst loading, base selection, and thermal profiles to maximize yield and purity. The process begins with the preparation of high-enantiomeric purity starting materials via resolution with N-Ac-L-cysteine, setting the foundation for the entire sequence. Subsequent coupling and cyclization steps must be monitored closely to prevent over-reaction or degradation. For detailed operational procedures, including specific molar ratios, workup protocols, and purification techniques, please refer to the standardized guide below.
- Perform an intermolecular Ullmann reaction between chiral Compound 1 and Compound 2 using a copper catalyst (e.g., CuBr) and base (e.g., K2CO3) in toluene at elevated temperatures to form the ether-linked intermediate Compound 3.
- Execute a hydroxyl deprotection step on Compound 3 under acidic conditions (e.g., sulfuric acid in methanol) to reveal the phenolic group necessary for the final ring closure, yielding Compound 4.
- Conduct an intramolecular Ullmann reaction on Compound 4 using a copper catalyst (e.g., CuI) and cesium carbonate in a polar aprotic solvent like DMF to close the macrocycle and finalize the optically pure tetrandrine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to this convergent synthetic route offers transformative economic and logistical benefits. The primary advantage lies in the decoupling of production from agricultural constraints. By relying on chemically synthesized intermediates, manufacturers can guarantee year-round production schedules不受 seasonal harvest cycles or geopolitical issues affecting raw herb availability. This stability is critical for maintaining continuous API supply lines for downstream drug formulations. Furthermore, the drastic reduction in synthesis steps—from nine in older methods to just three in this patent—fundamentally alters the cost structure. Fewer steps mean less solvent consumption, reduced energy usage for heating and cooling, and lower labor costs per kilogram of output. While specific financial figures depend on local utility costs, the qualitative reduction in process complexity invariably leads to a significantly lower cost of goods sold.
- Cost Reduction in Manufacturing: The elimination of expensive and time-consuming extraction processes, combined with the high-yield nature of the convergent synthesis, drives substantial cost efficiencies. By avoiding the low-yield bottlenecks of traditional linear synthesis (which previously yielded less than 2%), this method maximizes atom economy. The use of readily available copper catalysts instead of precious metals further reduces raw material expenses. Additionally, the ability to recycle solvents and reagents in a closed-loop chemical process minimizes waste disposal costs, contributing to a leaner, more profitable manufacturing model that enhances competitiveness in the global generic drug market.
- Enhanced Supply Chain Reliability: Dependence on plant extraction creates a fragile supply chain vulnerable to crop failures and quality fluctuations. This synthetic approach mitigates those risks by utilizing stable, storable chemical intermediates. The scalability demonstrated in the patent, moving from milligram to gram scales with consistent quality, indicates a clear path to multi-kilogram and ton-scale production. This reliability allows pharmaceutical companies to secure long-term contracts with confidence, knowing that their supplier can meet demand spikes without the lead times associated with agricultural sourcing. It effectively transforms tetrandrine from a scarce natural resource into a dependable commodity chemical.
- Scalability and Environmental Compliance: Modern regulatory environments demand rigorous control over impurities and waste. This synthetic route offers superior control over the impurity profile compared to crude plant extracts, which often contain complex mixtures of related alkaloids that are difficult to separate. The defined chemical steps allow for precise purification via crystallization or chromatography, ensuring high purity specifications are met consistently. Moreover, the reduction in total reaction steps inherently lowers the environmental footprint by reducing the volume of hazardous waste generated per unit of product. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of optically pure tetrandrine using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on feasibility and quality standards.
Q: What is the primary advantage of this synthetic route over traditional extraction?
A: Traditional extraction from Stephania tetrandra roots yields only about 1% tetrandrine and faces raw material shortages. This synthetic route offers a convergent strategy that increases total yield to nearly 39%, ensuring supply continuity independent of agricultural variables.
Q: How is optical purity maintained during the synthesis?
A: Optical purity is secured at the starting material stage through chemical resolution using N-Ac-L-cysteine, achieving ee values exceeding 99.8%. The subsequent Ullmann coupling conditions are mild enough to preserve these chiral centers throughout the 3-step process.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent demonstrates gram-scale synthesis (1.3g-1.5g) with robust yields. The reduction from 9 linear steps in previous methods to just 3 convergent steps significantly simplifies process control and reduces material costs, making it viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Pure Tetrandrine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic routes like the one described in CN111518108B to secure the supply of critical oncology and anti-inflammatory agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of tetrandrine meets the highest international pharmacopoeia standards. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis and macrocyclization with precision, delivering products that empower your drug development pipelines.
We invite you to collaborate with us to optimize your supply chain for tetrandrine and related bis-benzylisoquinoline alkaloids. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner to bring this high-purity, cost-effective therapeutic agent to the market faster and more efficiently than ever before.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
