Advanced Synthesis of Fondaparinux Sodium Intermediates for Commercial Scale-up
Introduction to Advanced Anticoagulant Intermediate Synthesis
The pharmaceutical landscape for anticoagulants has been revolutionized by the development of synthetic pentasaccharides, specifically Fondaparinux sodium, which serves as a selective inhibitor of Factor Xa. As detailed in patent CN102942601A, the synthesis of this complex molecule traditionally involves over fifty polystep reactions, creating significant bottlenecks in production capacity and cost efficiency. This new technical disclosure presents a breakthrough methodology for preparing key intermediates, specifically Compound D7, starting from the abundant and cost-effective raw material N-acetyl glucosamine. By streamlining the synthetic pathway, this approach addresses the critical industry need for reliable pharmaceutical intermediates supplier capabilities that can meet the rigorous demands of modern drug manufacturing. The structural complexity of the target molecule, as illustrated below, requires precise stereochemical control and robust protecting group strategies to ensure the final API meets global pharmacopoeia standards.
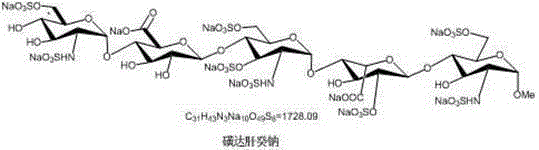
The strategic value of this patent lies in its ability to simplify the technology for synthesizing Fondaparinux sodium, thereby directly lowering production costs and enhancing supply chain stability. For R&D directors and procurement managers, understanding the nuances of this seven-step sequence is vital for evaluating potential partners who can deliver high-purity pharmaceutical intermediates. The process moves away from cumbersome traditional methods, utilizing optimized reaction conditions such as specific temperature ranges and catalytic systems to maximize yield at every stage. This report provides a deep dive into the mechanistic insights and commercial advantages of this novel approach, positioning it as a superior choice for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of Fondaparinux sodium has been plagued by excessive step counts and low overall yields, often requiring specialized reagents that are difficult to source on a large scale. Traditional routes frequently rely on intricate protecting group manipulations that necessitate harsh conditions, leading to epimerization or degradation of the sensitive carbohydrate backbone. These inefficiencies result in prolonged lead times and inflated costs, making the final anticoagulant medication less accessible for widespread clinical use. Furthermore, the reliance on multiple chromatographic purifications in older methods generates substantial chemical waste, posing environmental challenges and complicating regulatory compliance for green chemistry initiatives. The bottleneck in production capacity has limited the extensive use of this life-saving medicine, highlighting the urgent need for process innovation.
The Novel Approach
In stark contrast, the method disclosed in CN102942601A leverages a convergent strategy that begins with N-acetyl glucosamine, a renewable feedstock that offers significant economic advantages. The novel approach introduces a streamlined sequence where key transformations, such as the introduction of the azido group and benzyl protections, are executed with high regioselectivity and minimal byproduct formation. By optimizing parameters like reaction time and temperature, for instance maintaining 60°C for the initial glycosidation, the process achieves superior conversion rates compared to legacy methods. This efficiency translates directly into a more robust supply chain, as fewer unit operations mean reduced equipment occupancy and faster batch turnover. The result is a scalable protocol that maintains the structural integrity of the sugar moieties while drastically simplifying the downstream processing requirements.
Mechanistic Insights into Triflate-Mediated Azidation and Functionalization
A critical juncture in this synthesis is the conversion of Compound D3 to Compound D4, where a hydroxyl group is replaced by an azide functionality, a transformation essential for the biological activity of the final heparinoid. The patent describes a sophisticated activation strategy using trifluoromethanesulfonic anhydride to generate a highly reactive triflate intermediate in situ, which is then displaced by sodium azide. This mechanism allows for inversion of configuration or retention depending on the specific stereoelectronic environment, ensuring the correct alpha or beta linkage required for the pentasaccharide sequence. The use of a biphasic system involving toluene and water, along with careful pH control between 5 and 7, prevents the hydrolysis of the sensitive glycosidic bonds while facilitating the nucleophilic attack. Such precise control over reaction conditions is indicative of a mature process designed for high-purity pharmaceutical intermediates production.
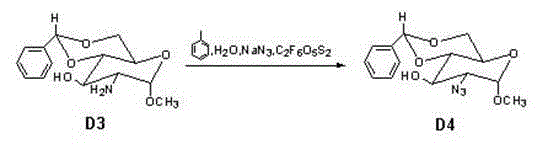
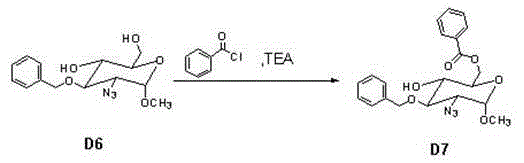
Following the azidation, the synthesis proceeds through benzylation and selective deprotection steps to install the necessary aromatic groups that serve as temporary masks for hydroxyl functionalities. The final transformation involves the benzoylation of Compound D6 to yield Compound D7, a key building block for the assembly of the full pentasaccharide. This step utilizes benzoyl chloride and triethylamine in toluene, a classic acylation reaction that is highly exothermic and requires careful thermal management to avoid side reactions. The ability to recrystallize the final product from normal heptane indicates a high degree of crystallinity and purity, which is paramount for reducing the burden on analytical quality control labs. Understanding these mechanistic details allows supply chain heads to appreciate the technical sophistication involved in producing these complex molecules reliably.
How to Synthesize Fondaparinux Intermediate Efficiently
The execution of this synthetic route requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety, particularly when handling reactive species like triflic anhydride and sodium azide. The process begins with the acid-catalyzed methanolysis of N-acetyl glucosamine to form the methyl glycoside, followed by a series of protection and functionalization steps that build molecular complexity incrementally. Operators must monitor reaction progress closely, utilizing techniques such as TLC or HPLC to determine endpoints before quenching and workup. The detailed standardized synthesis steps see the guide below, which breaks down the specific molar ratios, solvent volumes, and thermal profiles required for each of the seven stages. Mastery of these unit operations is essential for any contract development and manufacturing organization aiming to support the production of next-generation anticoagulants.
- Perform methanolysis of N-acetyl glucosamine using acidic resin to obtain Compound D1.
- Protect the diol system with benzaldehyde dimethyl acetal to form Compound D2, followed by deacetylation to D3.
- Activate the hydroxyl group with trifluoromethanesulfonic anhydride and substitute with sodium azide to yield Compound D4.
- Execute benzylation and subsequent hydrolysis to remove protecting groups, finishing with benzoylation to obtain Compound D7.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound benefits for procurement managers seeking to optimize the cost structure of their API supply chains. By utilizing N-acetyl glucosamine as a starting material, the process taps into a widely available and inexpensive commodity chemical, effectively decoupling production costs from the volatility of exotic reagent markets. The elimination of numerous intermediate isolation steps and the reduction in overall reaction count lead to substantially lower labor and utility consumption per kilogram of output. Furthermore, the high yields reported in the embodiments, such as the 97% yield in the benzylation step, imply a significant reduction in raw material waste, directly contributing to improved gross margins for the final drug product. These factors combine to create a compelling value proposition for partners looking for cost reduction in API manufacturing without compromising on quality.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive transition metal catalysts and reduces the consumption of organic solvents typically associated with lengthy purification sequences. By simplifying the workflow, manufacturers can achieve significant operational expenditure savings, as fewer reactor vessels are tied up for shorter durations, increasing overall plant throughput. The use of standard reagents like tosic acid and benzoyl chloride ensures that sourcing remains straightforward and competitive, avoiding single-supplier dependencies that often drive up prices. Consequently, the total cost of goods sold for the intermediate is drastically lowered, allowing for more aggressive pricing strategies in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate moderate variations in temperature and time without catastrophic failure, enhances the reliability of supply for critical medical ingredients. Since the starting materials are bio-based and produced on a massive global scale, the risk of raw material shortages is minimized, ensuring continuous production even during market disruptions. The process design also facilitates easier technology transfer between different manufacturing sites, providing flexibility in case of regional logistical challenges or capacity constraints. This resilience is crucial for maintaining the uninterrupted availability of anticoagulant therapies, which are often life-dependent for patients undergoing surgical procedures.
- Scalability and Environmental Compliance: The protocol is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without requiring fundamental changes to the chemistry. The waste profile is improved due to higher atom economy and the ability to recycle solvents like toluene and acetonitrile, aligning with increasingly stringent environmental regulations. By avoiding the generation of heavy metal waste and reducing the volume of aqueous effluent, the process lowers the burden on wastewater treatment facilities and reduces the carbon footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's background and beneficial effects sections. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for large-scale intermediate production. Stakeholders are encouraged to review these points to understand how this technology differentiates itself from conventional carbohydrate chemistry approaches. The answers reflect the specific improvements in yield, purity, and process simplicity documented in the intellectual property.
Q: What are the key advantages of this synthesis route for Fondaparinux intermediates?
A: The patented method significantly simplifies the traditional 50-step synthesis by utilizing N-acetyl glucosamine as a readily available starting material, reducing reaction steps and improving overall yield through optimized protection and deprotection strategies.
Q: How does the azidation step ensure high purity in the intermediate?
A: The process employs a specific activation strategy using trifluoromethanesulfonic anhydride under controlled pH conditions, which minimizes side reactions and ensures high regioselectivity during the nucleophilic substitution with sodium azide.
Q: Is this process scalable for industrial production of anticoagulant APIs?
A: Yes, the method utilizes common solvents like toluene and acetonitrile and avoids exotic catalysts, making it highly suitable for commercial scale-up while maintaining stringent quality control standards required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fondaparinux Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes like the one described in CN102942601A for securing the global supply of essential anticoagulants. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex carbohydrate intermediates are delivered with consistent quality and timing. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Our commitment to technical excellence means we can navigate the intricacies of azidation and glycosylation chemistry safely and effectively, providing a secure foundation for your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this optimized route. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability as a trusted partner in the fine chemical industry. Let us collaborate to bring cost-effective and high-quality Fondaparinux intermediates to the market, supporting better health outcomes worldwide.
