Advanced Synthetic Route For Fondaparinux Sodium Pentasaccharide Intermediates And Commercial Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for complex anticoagulants, specifically targeting the production of fondaparinux sodium, a critical synthetic pentasaccharide used in preventing venous thromboembolism. As detailed in patent CN103601766A, a novel synthetic route has been developed that fundamentally alters the construction of the fondaparinux sodium pentasaccharide intermediate, addressing long-standing challenges in stereocontrol and purification efficiency. This innovation focuses on the preparation of a specific trisaccharide intermediate and its subsequent coupling to form the final pentasaccharide backbone, offering a viable solution for high-purity API manufacturing. The structural complexity of fondaparinux sodium, characterized by five distinct monosaccharide units linked by specific glycosidic bonds, demands precise chemical control to ensure therapeutic efficacy and safety. 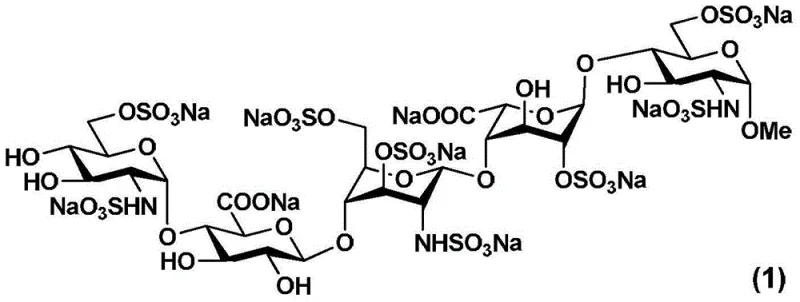
Traditional manufacturing processes for this critical cardiovascular medication have often been hindered by low overall yields and the formation of difficult-to-separate isomers, which directly impacts the cost of goods and supply reliability for global health systems. The new methodology described leverages a strategic redesign of the coupling sequence, moving away from conventional approaches that introduce the final monosaccharide unit at the very end of the synthesis. By reordering the assembly of the sugar units, the inventors have achieved a pathway with mild reaction conditions, excellent selectivity, and strong controllability, which are essential parameters for successful technology transfer from laboratory to commercial plant. This report analyzes the technical merits of this approach and its implications for procurement and supply chain stability in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fondaparinux sodium pentasaccharide intermediates has relied heavily on a 4+1 coupling strategy, where a tetrasaccharide acceptor is coupled with a monosaccharide donor in the final step. This conventional approach suffers from significant drawbacks, primarily due to the steric and electronic environment of the coupling site, which frequently leads to the formation of substantial amounts of isomeric byproducts. In many documented cases using the 4+1 scheme, the isomer proportion in the final coupling step can approach 20%, creating a severe bottleneck for purification efforts. Separating these closely related stereoisomers often requires extensive column chromatography, which is not only time-consuming and solvent-intensive but also results in significant material loss, driving down the overall yield. Furthermore, the accumulation of impurities throughout the lengthy synthetic sequence complicates the final crystallization of the API, potentially compromising the stringent purity specifications required for injectable anticoagulants. The high cost associated with these inefficiencies has historically limited the widespread application and affordability of this life-saving medication.
The Novel Approach
In contrast, the novel approach presented in the patent data introduces a 3+2 coupling strategy that fundamentally mitigates the risks associated with the final glycosylation step. By synthesizing a stable trisaccharide intermediate first and coupling it with a disaccharide unit, the chemical environment is optimized to favor the desired alpha-glycosidic linkage while suppressing isomer formation. This strategic shift ensures that the complex pentasaccharide backbone is assembled with higher fidelity, reducing the burden on downstream purification processes. The new route demonstrates that by altering the order of monosaccharide introduction, specifically bringing the E unit forward in the sequence, the reaction selectivity is markedly improved. This results in a cleaner reaction profile where the target pentasaccharide intermediate can be isolated with greater ease, often requiring only standard purification techniques rather than exhaustive chromatographic separation. Consequently, this method offers a pathway to significantly lower production costs and higher throughput, making it an attractive option for industrial-scale manufacturing.
Mechanistic Insights into Ag2CO3-Promoted Glycosylation and 3+2 Coupling
The core of this synthetic innovation lies in the precise preparation of the trisaccharide donor, specifically the conversion of intermediate EDC-1 to the activated trichloroacetimidate EDC-4. The process begins with the coupling of monosaccharide E-3 and disaccharide DC-2 using silver carbonate as a promoter in dichloromethane, yielding the trisaccharide EDC-1 with high stereocontrol. 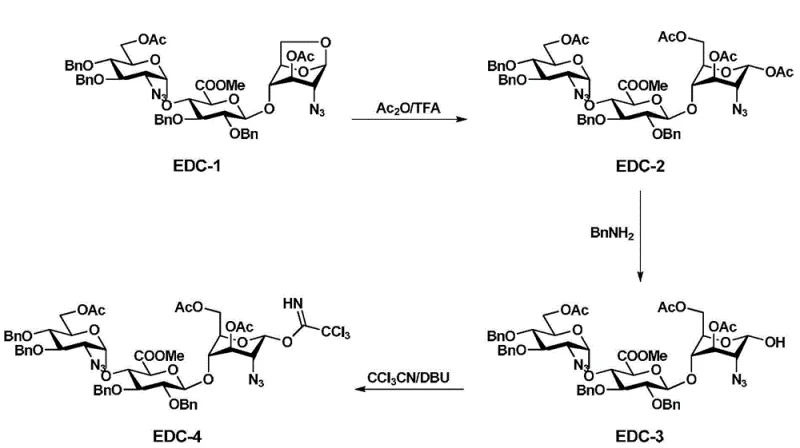
This intermediate EDC-1 is particularly valuable because its molecular structure contains an inner ether sugar moiety that imparts rigidity, facilitating purification via simple recrystallization to remove minor isomers. Subsequent transformation involves acetylation using acetic anhydride and trifluoroacetic acid, followed by selective deprotection and activation with trichloroacetonitrile in the presence of DBU. This sequence generates the highly reactive trisaccharide donor EDC-4, which is then poised for the critical 3+2 coupling reaction. The final step involves the coupling of EDC-4 with the disaccharide acceptor BA-2 using trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a catalyst at temperatures ranging from -30°C to 30°C. This specific catalytic system ensures smooth glycosidic bond formation with minimal side reactions, approaching a 1:1 reactant ratio which further enhances atom economy and reduces waste generation in the process.
The impurity control mechanism is inherently built into the choice of protecting groups and the coupling sequence. By utilizing benzoyl protection at specific positions rather than acetyl groups in certain precursors, the solubility and crystallization properties of the intermediates are tuned to favor the exclusion of impurities. The patent data indicates that the isomer proportion in the initial trisaccharide formation is reduced to lower than 5%, a stark improvement over the nearly 20% observed in alternative routes. This early-stage purity enhancement prevents the carryover of difficult-to-remove impurities into the final pentasaccharide stage. Moreover, the use of mild Lewis acids and controlled temperature profiles prevents degradation of the sensitive glycosidic linkages, ensuring that the structural integrity of the oligosaccharide chain is maintained throughout the synthesis. This rigorous control over the reaction landscape is what enables the production of high-purity intermediates suitable for pharmaceutical applications.
How to Synthesize Fondaparinux Sodium Intermediate Efficiently
The efficient synthesis of the fondaparinux sodium intermediate relies on the sequential assembly of protected monosaccharide building blocks into a trisaccharide donor, followed by coupling with a disaccharide acceptor. The process begins with the silver-promoted glycosylation of E-3 and DC-2 to form EDC-1, which is then purified and activated to EDC-4. Detailed standardized synthetic steps, including specific reagent quantities, solvent volumes, and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety in a GMP environment. 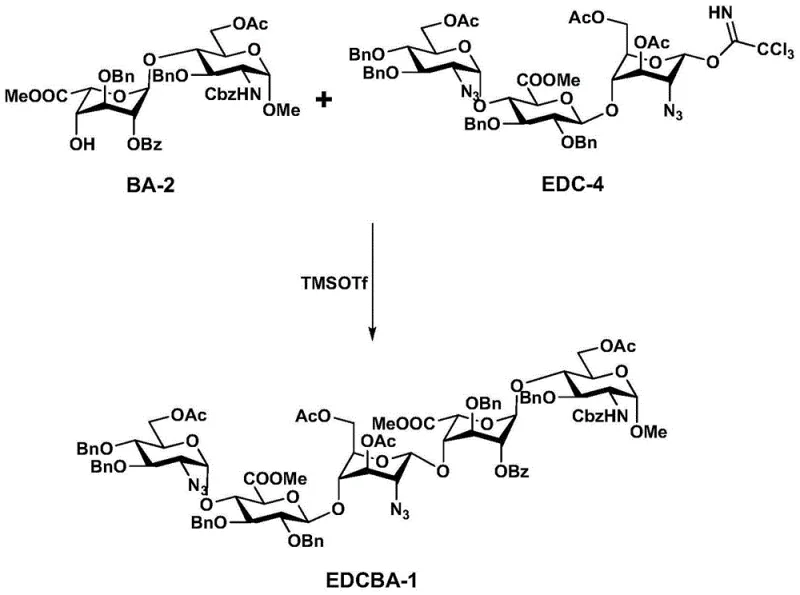
- Couple monosaccharide E-3 and disaccharide DC-2 using silver triflate to form trisaccharide EDC-1.
- Convert EDC-1 to trichloroacetimidate donor EDC-4 via acetylation and selective deprotection.
- Perform final 3+2 glycosylation between EDC-4 and disaccharide BA-2 to yield pentasaccharide EDCBA-1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible operational benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification workflow, which directly correlates to reduced processing time and lower consumption of expensive chromatography resins and solvents. By minimizing the formation of isomers, the manufacturing process becomes more predictable and robust, reducing the risk of batch failures that can disrupt supply continuity. This reliability is crucial for maintaining the steady flow of critical anticoagulant medications to the global market, especially in times of high demand. Furthermore, the use of readily available starting materials and common reagents ensures that the supply chain is not dependent on exotic or single-source catalysts, thereby mitigating sourcing risks.
- Cost Reduction in Manufacturing: The elimination of extensive purification steps and the improvement in overall yield lead to substantial cost savings in the production of fondaparinux intermediates. By avoiding the losses associated with separating difficult isomers, the effective cost per kilogram of the final API is significantly lowered. Additionally, the ability to use near-stoichiometric amounts of reactants in the final coupling step reduces raw material waste, contributing to a more economical process. These efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the pharmaceutical product, providing a clear financial advantage for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the 3+2 coupling strategy ensures consistent batch-to-batch quality, which is essential for regulatory compliance and long-term supply contracts. The reduced complexity of the synthesis means that production timelines are shorter and less prone to delays caused by troubleshooting purification issues. This predictability allows supply chain planners to optimize inventory levels and reduce the need for excessive safety stock. Moreover, the scalability of the process means that production capacity can be ramped up quickly to meet surges in demand, ensuring that the supply of this vital medication remains uninterrupted.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scale-up in mind, utilizing reaction conditions that are easily manageable in large reactors. The reduction in solvent usage and waste generation aligns with modern environmental standards and green chemistry principles, reducing the burden on waste treatment facilities. This environmental efficiency not only lowers disposal costs but also enhances the sustainability profile of the manufacturing operation. The process avoids the use of hazardous reagents where possible and operates at moderate temperatures, reducing energy consumption and improving overall workplace safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of fondaparinux sodium intermediates using this advanced methodology. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for technical teams evaluating this route for potential adoption. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing manufacturing frameworks.
Q: How does the 3+2 coupling strategy improve yield compared to traditional methods?
A: The 3+2 coupling strategy significantly reduces isomer formation during the final glycosylation step, lowering impurity levels to below 5% compared to nearly 20% in conventional 4+1 routes, thereby simplifying purification.
Q: What are the key purification advantages of the EDC-1 intermediate?
A: The EDC-1 intermediate possesses a rigid inner ether sugar structure that allows for effective purification through simple recrystallization, removing isomers without complex chromatography.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route utilizes mild reaction conditions ranging from -50°C to 30°C and avoids excessive reagent usage, making it highly controllable and cost-effective for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fondaparinux Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving anticoagulants like fondaparinux sodium. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch. Our capability to implement complex glycosylation strategies positions us as a strategic partner for pharmaceutical companies seeking to optimize their supply chains and reduce costs.
We invite you to contact our technical procurement team to discuss how this novel synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this more efficient manufacturing process. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and help you secure a reliable supply of high-purity fondaparinux intermediates for your global operations.
