Advanced Manufacturing of High-Purity Aripiprazole Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Aripiprazole Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for antipsychotic medications is continuously evolving, driven by the demand for higher purity standards and more sustainable manufacturing processes. Patent CN115340494A introduces a groundbreaking synthesis method for aripiprazole, a third-generation antipsychotic known for its unique dopamine system stabilizing properties. This technical disclosure outlines a robust three-step convergent route that strategically bypasses the limitations of legacy synthetic pathways. By focusing on the optimization of reaction conditions, specifically the use of aqueous media in the initial cyclization and controlled addition rates during etherification, this method achieves exceptional purity levels exceeding 99.5%. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the quality of the active pharmaceutical ingredient while simultaneously addressing the critical need for cost reduction in pharmaceutical intermediates manufacturing. The following analysis dissects the technical merits and commercial viability of this innovative approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aripiprazole has been plagued by complex multi-step sequences that rely on hazardous reagents and generate difficult-to-remove impurities. As illustrated in prior art routes, such as the fifth route depicted below, traditional methods often necessitate the use of tributyl vinyl tin and carbon monoxide, introducing severe toxicity and environmental compliance challenges. These organometallic reagents not only escalate raw material costs but also require rigorous and expensive heavy metal removal processes to meet regulatory safety standards. Furthermore, conventional etherification steps frequently suffer from poor selectivity, leading to the formation of dimeric by-products that compromise the overall yield and purity of the final API. The reliance on high-boiling organic solvents in these legacy routes further complicates solvent recovery, resulting in substantial energy consumption and increased operational expenditures for chemical manufacturers.
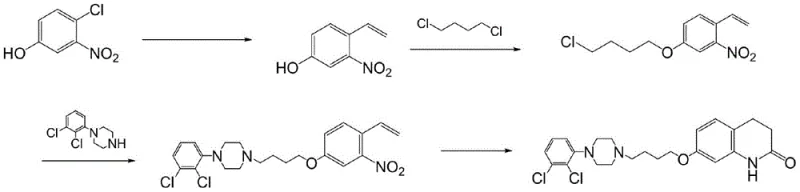
The Novel Approach
In stark contrast, the methodology disclosed in Patent CN115340494A offers a streamlined, three-step pathway that prioritizes safety, efficiency, and purity. The novel approach decouples the synthesis into two parallel intermediate preparations followed by a final convergent coupling, allowing for independent quality control of each fragment. A key differentiator is the substitution of toxic organometallics with benign alkyl halides and the strategic implementation of potassium iodide as a catalytic promoter. This catalytic system facilitates smoother nucleophilic substitutions under milder conditions. Moreover, the process engineering includes specific protocols, such as the slow dropwise addition of reactants, which kinetically suppresses the formation of dimer impurities. This results in a cleaner reaction profile that simplifies downstream processing and crystallization, directly translating to higher throughput and reduced waste generation for the reliable aripiprazole intermediate supplier.
Mechanistic Insights into KI-Catalyzed Nucleophilic Substitution
The core chemical innovation lies in the meticulous optimization of nucleophilic substitution reactions across all three steps, mediated by halide catalysis. In the initial cyclization step, 2,3-dichloroaniline reacts with bis(2-chloroethyl)amine hydrochloride. The presence of potassium iodide likely facilitates an in-situ Finkelstein-type halide exchange, converting the less reactive chloro-species into more reactive iodo-intermediates. This lowers the activation energy for the ring-closing nucleophilic attack by the amine nitrogen. The use of water as the solvent in this step is particularly ingenious; it leverages the hydrophobic effect to drive the reaction forward while allowing the product, 1-(2,3-dichlorophenyl)piperazine hydrochloride, to precipitate directly upon acidification. This phase separation mechanism eliminates the need for extractive workups with volatile organic solvents, showcasing a deep understanding of green chemistry principles applied to complex heterocycle synthesis.
Furthermore, the control of impurity profiles is achieved through precise kinetic management in the second step. During the etherification of 7-hydroxy-3,4-dihydro-2(1H)-quinolinone with 1,4-dibromobutane, the protocol mandates a slow, extended dropwise addition over seven hours. This low concentration of the phenolic nucleophile relative to the di-haloalkane minimizes the probability of double alkylation, which would lead to dimer formation. By maintaining a favorable stoichiometric ratio locally within the reaction vessel, the process ensures that the mono-alkylated product dominates. The subsequent coupling with the piperazine intermediate in acetonitrile utilizes triethylamine to scavenge the generated hydrobromic acid, driving the equilibrium towards the final aripiprazole product. This comprehensive mechanistic control ensures that the content of any single impurity remains below 0.1%, meeting the stringent requirements for high-purity OLED material and pharmaceutical grade standards alike.
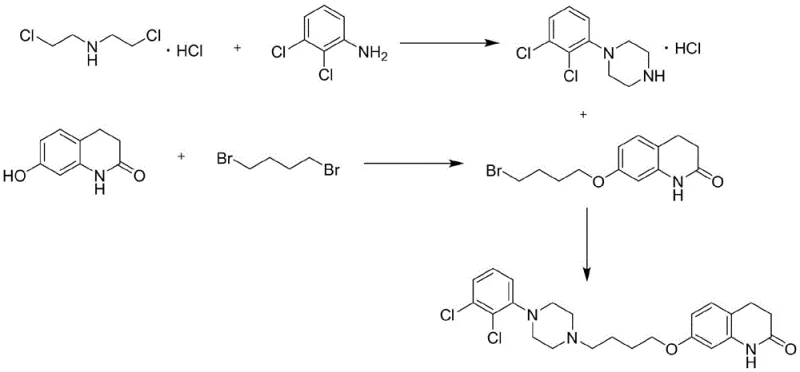
How to Synthesize Aripiprazole Efficiently
The implementation of this synthesis route requires strict adherence to the optimized reaction parameters defined in the patent to achieve the reported high yields and purity. The process is divided into three distinct operational units: the aqueous cyclization of the aniline derivative, the controlled etherification of the quinolinone core, and the final coupling reaction. Each step has been engineered to maximize atom economy and minimize waste. For process chemists looking to replicate this success, the key lies in the precise control of pH during the first step and the temperature modulation during the crystallization phases. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles, are outlined in the guide below to ensure reproducibility and quality consistency.
- Step 1: Cyclization of 2,3-dichloroaniline with bis(2-chloroethyl)amine hydrochloride in water using KOH and KI catalyst to form 1-(2,3-dichlorophenyl)piperazine hydrochloride.
- Step 2: Etherification of 7-hydroxy-3,4-dihydro-2(1H)-quinolinone with 1,4-dibromobutane in acetone, utilizing slow dropwise addition to suppress dimer formation.
- Step 3: Final coupling of the bromo-intermediate with the piperazine salt in acetonitrile using triethylamine to yield high-purity aripiprazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers transformative advantages for supply chain stability and cost management. The elimination of expensive and toxic reagents, such as organotin compounds and palladium catalysts found in alternative routes, fundamentally alters the cost structure of the manufacturing process. By relying on commodity chemicals like 2,3-dichloroaniline and 1,4-dibromobutane, the exposure to volatile raw material pricing is significantly mitigated. Additionally, the use of water as a solvent in the first step represents a massive reduction in solvent procurement costs and waste disposal fees. The ability to recover low-boiling solvents like acetone and acetonitrile further enhances the economic viability, ensuring that the commercial scale-up of complex pharmaceutical intermediates remains financially sustainable even under fluctuating market conditions.
- Cost Reduction in Manufacturing: The strategic replacement of high-cost organic solvents with water in the initial cyclization step leads to substantial cost savings in both raw material acquisition and waste treatment. The direct crystallization of the intermediate hydrochloride salt avoids energy-intensive distillation processes, thereby lowering the overall utility consumption of the plant. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, effectively increasing the overall yield and reducing the cost per kilogram of the final API. These factors combine to create a highly competitive cost position for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available, bulk commodity chemicals ensures a robust and resilient supply chain that is less susceptible to disruptions compared to routes requiring specialized reagents. The simplified post-treatment procedures, such as filtration and recrystallization, reduce the dependency on complex purification equipment, thereby shortening the production cycle time. This operational simplicity allows for greater flexibility in production scheduling and faster response to market demand fluctuations, ensuring a consistent supply of high-quality intermediates to downstream partners without compromising on delivery timelines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations that are easily transferable from pilot to commercial scale. The avoidance of heavy metals and toxic organics aligns perfectly with increasingly stringent global environmental regulations, reducing the regulatory burden and risk of compliance violations. The efficient solvent recovery systems and reduced waste generation contribute to a smaller environmental footprint, supporting corporate sustainability goals and enhancing the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new synthesis route improve impurity profiles compared to conventional methods?
A: The patented process specifically addresses dimer formation during the etherification step by employing a controlled slow-dropwise addition technique. This kinetic control, combined with the use of potassium iodide catalysis, ensures single impurity levels remain below 0.1% and overall purity exceeds 99.5%, significantly reducing downstream purification burdens.
Q: What are the environmental and cost benefits of using water in the first reaction step?
A: By utilizing water as the solvent for the initial cyclization of 2,3-dichloroaniline, the process eliminates the need for expensive and volatile organic solvents. This not only drastically reduces raw material costs but also simplifies post-treatment through direct crystallization of the hydrochloride salt, avoiding energy-intensive rectification processes required in traditional organic solvent systems.
Q: Is this synthesis method scalable for commercial production of aripiprazole intermediates?
A: Yes, the route is designed with industrial scalability in mind. It relies on readily available starting materials like 2,3-dichloroaniline and avoids toxic reagents such as organotin compounds found in older routes. The use of low-boiling point solvents like acetone and acetonitrile facilitates efficient solvent recovery, making the process highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
The technical advancements detailed in Patent CN115340494A underscore the potential for producing high-purity aripiprazole with superior efficiency and environmental stewardship. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by global regulatory bodies. We are committed to delivering value through technical excellence and operational reliability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and quality in your aripiprazole sourcing strategy.
