Revolutionizing Aripiprazole Manufacturing: A High-Purity, Low-Impurity Commercial Strategy
Revolutionizing Aripiprazole Manufacturing: A High-Purity, Low-Impurity Commercial Strategy
The global demand for second-generation antipsychotics continues to surge, placing immense pressure on supply chains to deliver Active Pharmaceutical Ingredients (APIs) with uncompromising purity profiles. In this context, the technical disclosure found in patent CN109180577B represents a significant leap forward in the process chemistry of Aripiprazole. This patent outlines a sophisticated synthetic strategy that fundamentally re-engineers the construction of the molecule's core scaffold, specifically targeting the notorious issue of dimer impurity formation that has plagued previous generations of manufacturing protocols. By shifting the synthetic logic to construct the piperazine moiety prior to the ether linkage, the inventors have achieved a level of impurity control that directly translates to reduced downstream processing costs and higher overall yields. For pharmaceutical manufacturers and procurement strategists, understanding the nuances of this pathway is critical for securing a competitive advantage in the generic drug market.
The Limitations of Conventional Methods vs. The Novel Approach
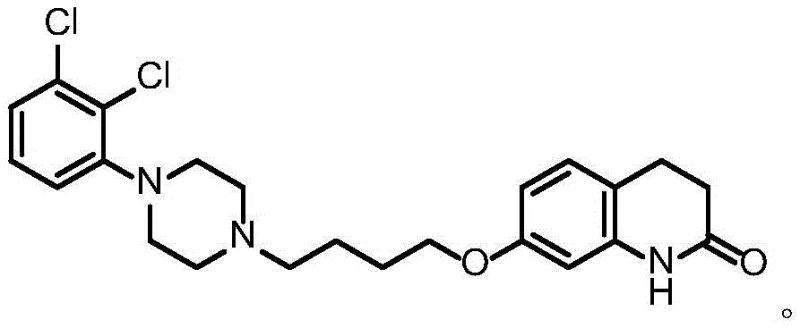
The Limitations of Conventional Methods
Historically, the industrial synthesis of Aripiprazole has been hindered by the reliance on etherification strategies that utilize symmetrical dihaloalkanes, such as 1,4-dibromobutane or 1,4-dichlorobutane. As illustrated in earlier prior art like patent CN1028104C, these conventional routes necessitate the use of a vast excess of the dihaloalkane—often up to 3 to 5 equivalents—to drive the reaction forward. This stoichiometric imbalance creates a statistical probability where both halogen ends of the alkane react, leading to the formation of diether dimer byproducts. These dimers are structurally similar to the target API, making them exceptionally difficult to remove via standard crystallization or chromatography, often resulting in final purities that struggle to meet stringent regulatory limits without extensive and costly reprocessing. Furthermore, the use of reagents like sodium azide in alternative ring-expansion routes introduces severe safety liabilities, including explosion risks, while the handling of large quantities of allergenic bromo-compounds poses significant occupational health challenges for plant operators.
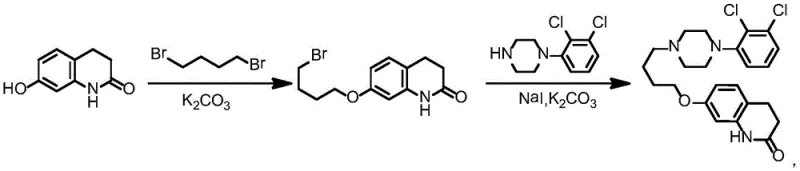
The Novel Approach
The methodology presented in CN109180577B circumvents these inherent flaws by adopting a convergent synthesis that prioritizes the formation of the piperazine ring system before introducing the butoxy linker. Instead of reacting a phenol with a dihaloalkane, this novel route begins with a palladium-catalyzed coupling of 2,3-dichlorobromobenzene with bis-2-chloroethylamine. This strategic inversion allows for the precise construction of the 1-(2,3-dichlorophenyl)piperazine core with high fidelity. Subsequent cyclization with 4-amino-1-butanol and chlorination generates a reactive chlorobutyl intermediate that is then coupled with the quinolinone fragment. This approach drastically reduces the required equivalents of raw materials, moving from a wasteful 3:1 ratio to a near-stoichiometric 1:1.2 ratio. The result is a process that not only minimizes the generation of the problematic dimer impurity but also enhances the atom economy of the entire sequence, offering a cleaner, safer, and more economically viable path to the final drug substance.
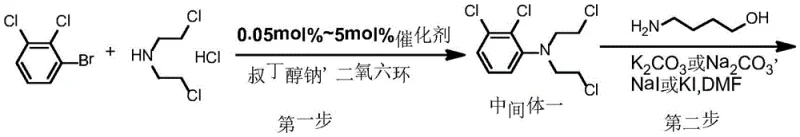
Mechanistic Insights into Pd-Catalyzed Coupling and Controlled Cyclization
The cornerstone of this improved synthesis lies in the initial palladium-catalyzed arylation step, which utilizes bis(dibenzylideneacetone)palladium as the catalyst and sodium tert-butoxide as the base in a dioxane solvent system. This specific catalytic system facilitates the nucleophilic attack of the amine nitrogen onto the electron-deficient aryl ring activated by the ortho-chlorine substituents. The choice of catalyst loading, optimized between 0.05 mol% and 5 mol%, ensures rapid conversion while allowing for potential catalyst recovery, a crucial factor for cost management in precious metal catalysis. Following this, the cyclization step employs 4-amino-1-butanol in the presence of an iodide catalyst (KI or NaI). The iodide acts as a nucleophilic catalyst, facilitating the displacement of the chlorine atoms on the ethyl chains to close the piperazine ring efficiently. This intramolecular cyclization is kinetically favored over intermolecular polymerization, which is the primary mechanism by which dimer impurities are suppressed in this route compared to the intermolecular etherification of older methods.
Furthermore, the conversion of the terminal hydroxyl group to a chloride using thionyl chloride is executed under strictly controlled low-temperature conditions (below 15°C) followed by reflux. This precision is vital to prevent the degradation of the sensitive piperazine ring or the formation of elimination byproducts. The resulting chlorobutyl intermediate is highly reactive towards the phenolic oxygen of the 3,4-dihydro-7-hydroxy-2(1H)-quinolinone in the final step. By performing this final coupling in an alkaline aqueous medium, the process leverages the phase transfer capabilities to drive the reaction to completion without requiring hazardous organic solvents in large volumes. The mechanistic elegance of this sequence ensures that impurities generated in early steps do not propagate through to the final API, thereby simplifying the purification profile and enabling the achievement of HPLC purities exceeding 99.8% as demonstrated in the patent examples.
How to Synthesize Aripiprazole Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the exothermic chlorination step and the maintenance of anhydrous conditions during the palladium coupling. The patent provides a robust framework for scaling this reaction, detailing specific molar ratios and solvent choices that maximize yield while minimizing waste. For process chemists looking to implement this technology, the key lies in the sequential addition of reagents and the rigorous monitoring of intermediate conversion via TLC or HPLC to prevent over-reaction. The detailed standardized synthesis steps below outline the precise operational protocol required to replicate the high-purity results reported in the intellectual property.
- Couple 2,3-dichlorobromobenzene with bis-2-chloroethylamine using a Pd catalyst and sodium tert-butoxide to form Intermediate I.
- React Intermediate I with 4-amino-1-butanol in the presence of alkali and KI/NaI catalyst to cyclize and form Intermediate II.
- Convert the hydroxyl group of Intermediate II to a chloride using thionyl chloride to obtain Intermediate III.
- Perform the final substitution reaction between Intermediate III and 3,4-dihydro-7-hydroxy-2(1H)-quinolinone in alkaline aqueous solution.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages for supply chain stability and cost structure optimization. Traditional methods that rely on excessive amounts of 1,4-dibromobutane not only inflate raw material costs but also create significant waste disposal burdens due to the toxicity and difficulty in recovering the excess reagent. By contrast, the stoichiometric efficiency of the new method means that procurement teams can secure lower volumes of starting materials for the same output of API, directly impacting the cost of goods sold (COGS). Additionally, the elimination of hazardous reagents like sodium azide removes the need for specialized explosion-proof infrastructure and complex safety protocols, further reducing the overhead costs associated with manufacturing compliance and insurance.
- Cost Reduction in Manufacturing: The most significant economic driver here is the drastic reduction in material equivalents. Moving from a 3-to-5-fold excess of alkylating agents to a near 1-to-1 ratio represents a massive saving in raw material expenditure. Furthermore, the ability to recover and reuse the palladium catalyst and solvents like dioxane adds another layer of cost efficiency. The simplified purification process, necessitated by the lower impurity load, reduces the consumption of recrystallization solvents and energy, leading to substantial operational savings without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable starting materials. Unlike routes dependent on unstable halo-alcohols that tend to cyclize into tetrahydrofuran upon storage, the reagents used in this pathway are robust and have long shelf lives. This stability ensures consistent batch-to-batch quality and reduces the risk of production delays caused by raw material degradation. Moreover, the avoidance of specialty reagents that may be subject to export controls or supply shortages ensures a more predictable and continuous manufacturing schedule for global distribution.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly lighter than its predecessors. The reduction in waste generation aligns with modern green chemistry principles, making it easier for manufacturers to meet increasingly strict environmental regulations. The aqueous workup in the final step minimizes the use of volatile organic compounds (VOCs), and the high atom economy ensures that a greater proportion of input mass ends up in the final product rather than in waste streams. This scalability and environmental compatibility make the process ideal for multi-ton production campaigns required to meet global market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is superior for industrial application. Understanding these specifics is essential for technical teams evaluating technology transfer opportunities.
Q: How does this new route control dimer impurities compared to traditional methods?
A: Traditional methods often use excess 1,4-dihalobutanes which statistically generate diether dimers (>10%). This novel route builds the piperazine ring first and uses a controlled chlorobutyl chain, significantly reducing dimer formation and simplifying purification.
Q: What are the safety advantages of this synthesis pathway?
A: Unlike routes requiring sodium azide for ring expansion or large excesses of allergenic dibromobutanes, this method utilizes standard coupling and chlorination reagents, reducing explosion risks and occupational health hazards.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process demonstrates high yields (over 90% in key steps) and uses recoverable catalysts. The reduction in material equivalents and simplified workup procedures makes it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
The technical potential of the synthesis route described in CN109180577B is immense, offering a clear pathway to high-quality, cost-effective Aripiprazole production. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of meeting stringent purity specifications, guaranteeing that every batch of API intermediate or finished drug substance adheres to the highest international standards. We understand the critical nature of impurity control in antipsychotic medications and have the expertise to manage complex reaction profiles effectively.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthetic route, we can help you reduce your overall manufacturing costs while securing a supply of high-purity materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering capabilities can support your long-term supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
