Advanced Manufacturing of Dronedarone: Technical Insights for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with industrial feasibility, particularly for complex antiarrhythmic agents like Dronedarone. Patent CN102770420A introduces a transformative methodology for the preparation of Dronedarone and its salts, addressing critical bottlenecks in yield and purification that have historically plagued large-scale manufacturing. This novel approach leverages a strategic sequence of catalytic reduction and simultaneous functionalization to bypass tedious chromatographic steps, ensuring a streamlined path to high-quality Active Pharmaceutical Ingredients (APIs). By integrating efficient unit operations with precise chemical control, this process offers a compelling solution for reliable pharmaceutical intermediate supplier networks aiming to secure stable inventory levels. The technical breakthrough lies not just in the chemistry itself, but in the holistic optimization of the workflow, which minimizes waste generation and maximizes throughput.
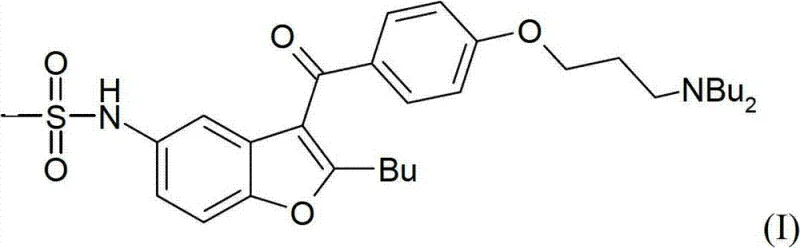
Furthermore, the ability to produce Dronedarone with an HPLC purity exceeding 99% directly through crystallization rather than column chromatography represents a significant leap forward in process chemistry. This advancement is crucial for meeting stringent regulatory standards regarding impurity profiles and residual solvents. For procurement specialists and supply chain managers, understanding the underlying mechanics of this synthesis is vital for assessing long-term vendor viability and cost structures. The patent details a route that transforms complex molecular architectures into manageable industrial processes, thereby reducing the risk of supply disruptions caused by low-yielding or unstable synthetic steps. As we delve deeper into the technical specifics, it becomes evident how this methodology aligns with modern Green Chemistry principles while delivering substantial economic advantages.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dronedarone has been fraught with challenges related to multi-step protection and deprotection strategies that inherently lower overall yields and increase production costs. Conventional routes, such as those described in prior art like WO2009/044143, often require the introduction of different functional groups onto the benzofuran ring through sequential reactions that demand rigorous purification after each stage. These traditional methods frequently rely on chromatographic techniques to isolate intermediates, which are not only solvent-intensive but also difficult to scale up efficiently for multi-ton production campaigns. The reliance on chromatography introduces significant variability in batch-to-batch consistency and creates bottlenecks in manufacturing throughput, ultimately driving up the cost of goods sold (COGS). Additionally, the use of sensitive protecting groups necessitates harsh conditions for their removal, which can lead to the formation of difficult-to-remove impurities that compromise the final drug substance quality.
The Novel Approach
In stark contrast, the process disclosed in CN102770420A revolutionizes the synthesis by employing a convergent strategy that minimizes the number of isolation steps and eliminates the need for chromatography entirely. The core innovation involves the preparation of a key intermediate, 3-{4-[(2-butyl-5-nitro-1-benzofuran-3-yl)carbonyl]phenoxy}propan-1-ol, which serves as a robust starting point for the subsequent transformations. 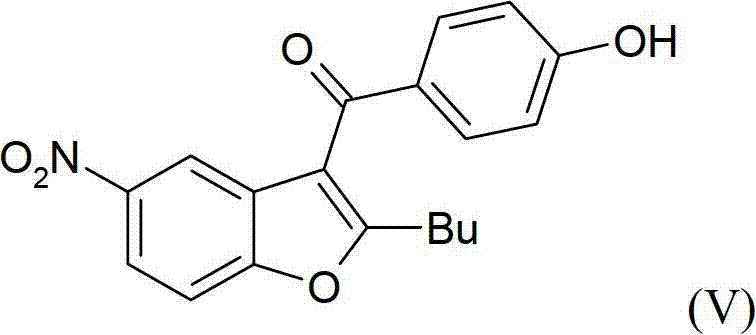
This novel route utilizes a direct alkylation of the phenolic hydroxyl group followed by a highly efficient reduction and activation sequence. By avoiding the cumbersome protection-deprotection cycles typical of older methodologies, the new process significantly reduces the consumption of reagents and solvents. The strategic design allows for the direct conversion of crude intermediates into the final product through simple crystallization, which is a far more scalable and cost-effective purification technique. This shift from chromatographic to crystalline purification is a game-changer for cost reduction in pharmaceutical intermediates manufacturing, as it drastically cuts down on processing time and hazardous waste disposal costs. The result is a synthesis that is not only chemically elegant but also commercially superior in terms of operational simplicity and environmental footprint.
Mechanistic Insights into Catalytic Hydrogenation and Dual Mesylation
The heart of this improved synthesis lies in the precise execution of the reduction and activation steps, which dictate the purity and yield of the final API. The process begins with the catalytic hydrogenation of the nitro group in the presence of a heterogeneous catalyst such as Palladium on Carbon (Pd/C) or Platinum Oxide (PtO2). This reaction is conducted in lower alcohols like methanol or ethanol at moderate temperatures ranging from room temperature to approximately 50°C, typically under a hydrogen pressure of around 4 bar. The use of heterogeneous catalysis is particularly advantageous for industrial applications because the catalyst can be easily removed by filtration, preventing metal contamination in the product stream. Following the reduction, the resulting amino-alcohol intermediate undergoes a critical dual mesylation reaction. 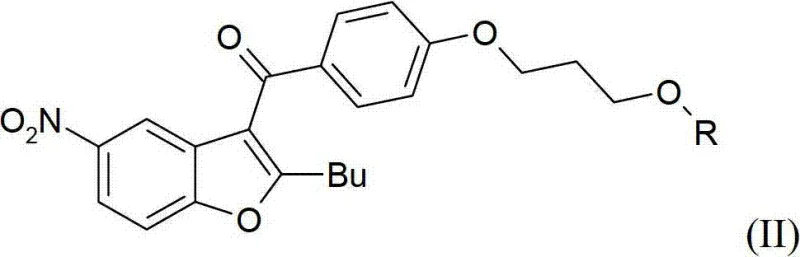
This step is mechanistically fascinating as it allows for the simultaneous introduction of the sulfonamide functionality and the activation of the primary alcohol as a mesylate leaving group in a single operational sequence. By reacting the amino-alcohol with an excess of methanesulfonyl chloride in a chlorinated solvent like dichloromethane or acetonitrile, both the amine and the hydroxyl groups are converted into their respective mesylated forms. This dual activation is pivotal because it sets up the molecule for the final nucleophilic substitution without requiring intermediate isolation or purification. The ability to perform this transformation in one pot significantly enhances the commercial scale-up of complex pharmaceutical intermediates by reducing the number of reactor turnovers and handling operations. Moreover, the reaction conditions are mild enough to preserve the integrity of the sensitive benzofuran core while ensuring complete conversion of the reactive sites.
Impurity control is rigorously maintained throughout this process through the strategic use of crystallization rather than chromatography. The dimesylated intermediate can be isolated as a solid with high purity simply by cooling the reaction mixture or adding an anti-solvent like toluene. This physical separation method is highly effective at rejecting soluble impurities and by-products that might otherwise carry through to the final step. The subsequent nucleophilic displacement with dibutylamine proceeds smoothly in polar aprotic solvents such as acetonitrile, where the activated mesylate group is readily displaced to form the tertiary amine side chain. 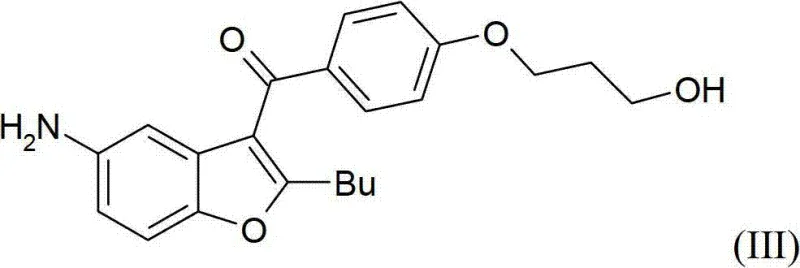
Because the preceding steps have already established a high level of purity, the final crude Dronedarone requires minimal workup before salt formation. The final conversion to the hydrochloride salt is achieved using standard acidification techniques, yielding a product with an HPLC purity of greater than 99%. This high level of purity is attained without the need for resource-intensive chromatographic columns, demonstrating the efficacy of the designed synthetic pathway. The mechanistic elegance of this route ensures that potential genotoxic impurities or heavy metal residues are kept well below regulatory thresholds, providing a safe and reliable supply of the active ingredient for downstream formulation.
How to Synthesize Dronedarone Efficiently
The implementation of this novel synthesis route requires careful attention to reaction parameters such as temperature control, stoichiometry, and solvent selection to maximize efficiency and safety. The process is designed to be robust, allowing for the direct use of crude intermediates in subsequent steps, which simplifies the overall workflow and reduces material loss. Detailed standard operating procedures for each stage, from the initial hydrogenation to the final salt formation, are essential for maintaining consistency across different production batches. 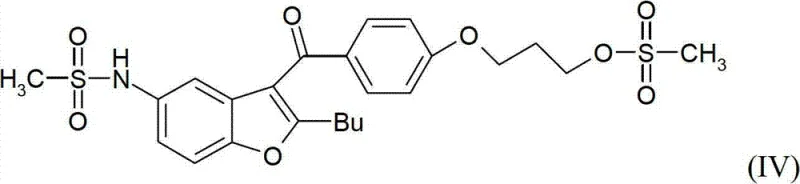
- Catalytic hydrogenation of the nitro-benzofuran precursor using Pd/C in alcohol solvents to generate the amino-alcohol intermediate.
- Simultaneous dimesylation of the amine and hydroxyl groups using methanesulfonyl chloride to activate the molecule for substitution.
- Nucleophilic displacement with dibutylamine followed by salt formation to yield high-purity Dronedarone hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis process translates into tangible strategic benefits that extend beyond simple chemical yield. The elimination of chromatographic purification steps represents a massive reduction in operational complexity and cost, as chromatography is notoriously expensive due to the high volume of solvents required and the specialized equipment needed for large-scale separation. By replacing this with crystallization, the process significantly lowers the variable costs associated with solvent purchase, recovery, and disposal. This shift also reduces the physical footprint required for manufacturing, as large chromatography columns and associated pumping systems are no longer necessary. Consequently, the production capacity of existing facilities can be effectively increased without major capital investment in new infrastructure, allowing for faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The streamlined nature of this process drives down manufacturing costs through several key mechanisms, primarily by removing the need for expensive protecting groups and chromatographic media. The use of commodity chemicals such as methanesulfonyl chloride and dibutylamine, combined with common solvents like acetonitrile and toluene, ensures that raw material costs remain stable and predictable. Furthermore, the high yields obtained in each step—such as the 91% yield in the reduction step and 92% in the mesylation step—accumulate to provide a superior overall yield compared to traditional multi-step routes. This efficiency means less raw material is wasted per kilogram of final product, directly improving the margin profile for the manufacturer and offering potential pricing advantages to buyers.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly enhanced by the robustness of the synthetic steps, which rely on well-understood unit operations like filtration and distillation rather than delicate separation techniques. The ability to isolate intermediates as stable crystalline solids allows for the creation of strategic inventory buffers at various stages of the synthesis, mitigating the risk of production stoppages due to equipment failure or raw material delays. Additionally, the process does not rely on exotic or hard-to-source reagents, reducing the vulnerability of the supply chain to geopolitical or logistical disruptions. This reliability is critical for pharmaceutical companies that require guaranteed continuity of supply to meet patient needs and regulatory commitments without interruption.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern sustainability goals by minimizing solvent usage and waste generation. The reduction in solvent intensity directly lowers the E-factor (mass of waste per mass of product), making the process more environmentally friendly and easier to permit in regions with strict environmental regulations. The scalability is further supported by the fact that crystallization is a linearly scalable process, meaning that moving from pilot plant to commercial scale does not introduce the non-linear complexities often seen with chromatographic separations. This ease of scale-up ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable, allowing manufacturers to ramp up production quickly in response to global health demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel Dronedarone synthesis route. These insights are derived directly from the patent data and practical experience in scaling similar chemical processes, providing a clear picture of what partners can expect. Understanding these details helps stakeholders make informed decisions about sourcing strategies and technology transfer protocols.
Q: How does the new process improve purity compared to conventional methods?
A: The novel process eliminates the need for chromatographic purification of intermediates. By utilizing crystallization for isolation and a simultaneous dimesylation strategy, the method achieves HPLC purity greater than 99% while significantly reducing processing time and solvent waste.
Q: What are the key cost drivers reduced in this synthesis route?
A: Major cost reductions are achieved by avoiding expensive protecting group strategies and chromatography columns. The use of standard heterogeneous catalysts like Pd/C and common solvents like acetonitrile and toluene further lowers the raw material expenditure and simplifies waste treatment.
Q: Is this process scalable for commercial API production?
A: Yes, the process is designed for industrial expediency. It relies on robust unit operations such as filtration, crystallization, and standard reflux reactions, which are easily transferable from pilot scale to multi-ton commercial manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dronedarone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles described in patent CN102770420A can be reliably replicated on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Our commitment to quality assurance means that every gram of Dronedarone we supply meets the exacting requirements necessary for global regulatory filings and patient safety.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements and logistical constraints. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal benchmarks. Together, we can build a resilient and efficient supply chain for Dronedarone that supports the continued availability of this vital antiarrhythmic medication.
