Industrial Scale-Up of High-Purity d-Biotin via Novel Lewis Acid Catalysis
Introduction to Next-Generation d-Biotin Manufacturing
The global demand for high-purity vitamins and nutritional supplements continues to surge, placing immense pressure on manufacturers to optimize synthetic routes for critical nutrients like d-biotin, also known as Vitamin H or Vitamin B7. A significant technological breakthrough in this sector is documented in patent CN112390815B, which discloses a novel preparation method for d-biotin utilizing Lewis acid-mediated debenzylation. This innovation addresses long-standing challenges in the chemical synthesis of this essential cofactor, offering a pathway that combines operational simplicity with exceptional product quality. By shifting away from traditional harsh acidic conditions or toxic boron halides, this method leverages specific Lewis acids to achieve selective deprotection, ensuring that the delicate heterocyclic core of the biotin molecule remains intact throughout the transformation. For industry stakeholders, this represents a pivotal shift towards safer, more efficient, and highly scalable manufacturing protocols that align with modern green chemistry principles and rigorous regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of d-biotin has relied heavily on the Goldberg-Sternbach total synthesis route, where the removal of benzyl protecting groups is a critical bottleneck. Traditional methods often employ concentrated hydrobromic acid for azeotropic debenzylation, a process fraught with significant chemical drawbacks. The aggressive nature of strong mineral acids frequently leads to incomplete debenzylation and, more critically, induces the hydrolysis of the urea moiety within the imidazolidone ring. This side reaction generates ring-opened diamine byproducts, specifically (2S,3S,4S)-5-(3,4-diamino-tetrahydrothiophene-2-yl)pentanoic acid, which necessitates a subsequent and hazardous re-cyclization step using virulent phosgene under alkaline conditions. Furthermore, alternative approaches utilizing boron trihalides, while theoretically milder, have demonstrated issues with reproducibility, high toxicity, and severe corrosiveness that pose substantial risks to equipment integrity and operator safety in a large-scale plant environment.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN112390815B introduces a robust one-step debenzylation strategy using dried Lewis acids such as aluminum chloride, zinc chloride, or ferric chloride. This approach operates effectively in anhydrous organic solvents like halogenated hydrocarbons or aromatic hydrocarbons at moderate temperatures ranging from 50°C to 100°C. The key innovation lies in the chemoselectivity of the Lewis acid catalyst, which facilitates the cleavage of the N-benzyl bonds without triggering the ring-opening degradation observed with hydrobromic acid. Consequently, the need for toxic phosgene re-cyclization is entirely eliminated, streamlining the workflow into a single, continuous transformation. This not only simplifies the process flow but also drastically reduces the generation of hazardous waste and improves the overall atom economy of the synthesis, making it an ideal candidate for modern, sustainable pharmaceutical manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Debenzylation
The success of this synthetic route hinges on the precise interaction between the Lewis acid catalyst and the dibenzyl biotin substrate. Unlike protic acids that donate protons and can attack nucleophilic sites indiscriminately, Lewis acids function as electron pair acceptors, coordinating specifically with the nitrogen atoms of the imidazolidone ring. This coordination weakens the carbon-nitrogen bond of the benzyl group, facilitating its cleavage under thermal conditions without compromising the stability of the adjacent carbonyl group. This mechanistic nuance is crucial because it prevents the formation of the diamine impurity, thereby ensuring that the cyclic structure of the biotin skeleton is preserved throughout the reaction. The use of anhydrous conditions further supports this mechanism by preventing the hydrolysis of the Lewis acid and minimizing side reactions associated with moisture, leading to a cleaner reaction profile and higher crude purity before any purification steps are undertaken.
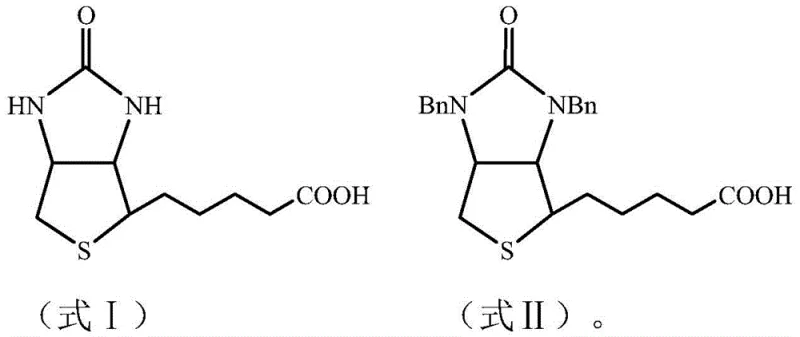
Furthermore, the control of impurities is inherently built into the reaction design through the selection of specific solvent systems and stoichiometric ratios. The patent data indicates that maintaining a molar ratio of dibenzyl biotin to Lewis acid between 1:2 and 1:4 optimizes the reaction kinetics while preventing excessive complexation that could complicate downstream processing. By avoiding the formation of ring-opened byproducts, the impurity profile of the crude product is significantly simplified, consisting primarily of unreacted starting material or mono-benzyl intermediates which are easily removed during subsequent crystallization. This high level of selectivity translates directly into a final product that consistently achieves purity levels exceeding 99%, with individual related substances controlled below 0.1%, satisfying the stringent acceptance criteria of the European Pharmacopoeia EP 10.0 without the need for extensive chromatographic purification.
How to Synthesize d-Biotin Efficiently
The practical implementation of this Lewis acid-mediated debenzylation requires careful attention to reaction parameters to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of an anhydrous reaction environment, typically under a nitrogen or argon blanket, to protect the moisture-sensitive Lewis acid catalyst. Detailed operational procedures involve dissolving the Lewis acid in a suitable solvent such as 1,2-dichloroethane or chlorobenzene, heating the mixture to the target range of 60-80°C, and then slowly introducing the dibenzyl biotin solution. Following the reaction period of 2 to 4 hours, the mixture is quenched carefully with dilute hydrochloric acid at temperatures below 40°C to decompose the catalyst complexes and precipitate the product.
- Prepare the reaction system by mixing dried Lewis acid (e.g., AlCl3, ZnCl2) with an anhydrous organic solvent (e.g., 1,2-dichloroethane, chlorobenzene) under inert gas protection.
- Heat the mixture to 50-100°C and slowly add a solution of dibenzyl biotin dissolved in the same solvent, maintaining the temperature for 2-4 hours.
- Quench the reaction with dilute hydrochloric acid below 40°C, adjust pH to 1-3, and perform recrystallization using water or alcohol to obtain high-purity d-biotin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Lewis acid-based synthesis offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of phosgene from the process flow removes a major regulatory and safety burden, significantly lowering the capital expenditure required for specialized containment systems and emergency response infrastructure. This simplification of the safety profile allows for more flexible site selection and reduces insurance premiums associated with handling extremely hazardous substances. Additionally, the use of commercially available and relatively inexpensive Lewis acids like aluminum chloride or zinc chloride replaces costly and difficult-to-handle reagents, leading to a substantial reduction in raw material costs. The robustness of the reaction conditions also implies a wider operating window, which minimizes batch failures and ensures a more consistent supply of critical intermediates to downstream formulation units.
- Cost Reduction in Manufacturing: The streamlined one-step debenzylation process eliminates the need for a separate re-cyclization stage involving phosgene, which traditionally requires expensive reagents and complex waste treatment protocols. By consolidating the transformation into a single reactor operation, manufacturers can achieve significant savings in utility consumption, labor hours, and reactor occupancy time. The avoidance of ring-opening side reactions further enhances cost efficiency by maximizing the yield of the desired product and minimizing the loss of valuable starting materials to irrecoverable byproducts, thereby improving the overall cost-per-kilogram metric for d-biotin production.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable Lewis acids and common organic solvents mitigates the supply chain risks associated with sourcing highly regulated or toxic reagents like boron trihalides or phosgene. These standard chemicals are widely available from multiple global suppliers, ensuring continuity of supply even during market fluctuations. Furthermore, the mild reaction conditions reduce wear and tear on production equipment, extending the lifespan of reactors and piping systems and decreasing the frequency of maintenance shutdowns, which contributes to a more reliable and predictable production schedule for meeting customer delivery deadlines.
- Scalability and Environmental Compliance: From an environmental perspective, this method generates a cleaner waste stream devoid of heavy metal contaminants or highly corrosive acidic residues that are difficult to neutralize. The simplified workup procedure, involving straightforward crystallization and filtration, reduces the volume of solvent waste and the energy load associated with distillation or extraction processes. This alignment with green chemistry principles facilitates easier compliance with increasingly strict environmental regulations, allowing manufacturers to operate with a smaller ecological footprint while maintaining the capacity to scale production from pilot batches to multi-ton annual volumes without encountering significant engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in an industrial setting. Understanding these details is essential for R&D teams evaluating process transfer and for quality assurance personnel ensuring regulatory compliance. The answers provided are derived directly from the experimental data and technical specifications disclosed in the patent literature, offering a reliable foundation for decision-making.
Q: How does the Lewis acid method prevent ring-opening side reactions compared to HBr?
A: Unlike hydrobromic acid which causes harsh acidic conditions leading to ring-opened diamine byproducts, the Lewis acid method operates under milder conditions that selectively cleave the N-benzyl bond without attacking the urea carbonyl, thus preserving the cyclic structure.
Q: What represents the primary safety advantage of this process over traditional phosgene routes?
A: This method eliminates the need for re-cyclization using virulent phosgene, significantly reducing toxicity risks and simplifying the safety protocols required for industrial production facilities.
Q: Can this process meet European Pharmacopoeia standards?
A: Yes, the process yields d-biotin with a content exceeding 99% and controls individual impurities below 0.1%, fully meeting the stringent quality requirements of the European Pharmacopoeia EP 10.0.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable d-Biotin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global vitamin market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the Lewis acid debenzylation of d-biotin are translated efficiently from the laboratory to the manufacturing floor. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of d-biotin we produce meets or exceeds the requirements of the European Pharmacopoeia EP 10.0 and other international standards. We are committed to delivering high-quality pharmaceutical intermediates that empower our clients to bring safer and more effective nutritional products to market.
We invite you to collaborate with our technical procurement team to explore how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this greener, more efficient manufacturing protocol. We encourage you to contact us today to discuss your requirements,索取 specific COA data, and review our comprehensive route feasibility assessments to ensure a seamless partnership focused on quality, reliability, and mutual growth.
