Advanced Phase-Transfer Catalytic Synthesis of 7-Chloroquinaldine for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with environmental sustainability, a challenge vividly addressed in patent CN103524408A. This intellectual property discloses a groundbreaking method for preparing 7-chloroquinaldine, a critical intermediate for antiasthmatics like Montelukast and LTRA MK-0679, utilizing a sophisticated phase-transfer catalytic reaction. By employing m-chloroaniline and crotonaldehyde as raw materials within a unique water and 2-butanol reaction system, the inventors have achieved a remarkable yield of up to 89%. The core innovation lies in the synergistic application of lauryl sodium sulfate (SDS) as a surfactant and phosphomolybdic acid as a multifunctional oxidizing agent. This approach not only streamlines the synthetic pathway but also fundamentally alters the reaction kinetics to favor the desired 7-position isomer over unwanted byproducts. For global procurement teams and R&D directors, this patent represents a pivotal shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates without compromising on structural integrity or purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-chloroquinaldine has been plagued by significant technical hurdles that inflate production costs and complicate supply chains. Traditional methodologies, such as the classical Skraup reaction and Doebner-Miller condensation, rely on m-chloroaniline and crotonaldehyde but invariably generate substantial quantities of the 5-position isomer. Separating this isomeric impurity requires arduous purification steps involving zinc chloride, tartrates, or 4-nitrophthalic acid, which drastically reduces the overall yield and introduces heavy metal contaminants. Furthermore, alternative prior art, such as US Patent 5126456, utilizes chloranil as an oxidant in aqueous isopropanol; while achieving a moderate 67% yield, this route necessitates the use of large volumes of flammable tetrahydrofuran (THF) for separation and involves harsh dripping conditions that are difficult to control on a multi-ton scale. Other phase-transfer attempts, like those in CN 101638382, suffer from complex post-reaction treatments requiring continuous vacuum distillation, leading to severe product runoff and rendering them unsuitable for efficient industrial application.
The Novel Approach
The methodology outlined in CN103524408A offers a transformative solution by re-engineering the reaction environment through precise phase-transfer catalysis. By introducing Sodium Dodecyl Sulfate (SDS) into the reaction matrix, the process significantly enhances the emulsibility of the reaction liquid, creating a micro-environment that kinetically favors the formation of the target 7-chloroquinaldine while inhibiting the generation of the problematic 5-isomer. Additionally, the substitution of traditional acid solvents with phosphomolybdic acid serves a dual purpose: it acts as both the acidic catalyst and the oxidizing agent, thereby simplifying the reagent list and reducing chemical waste. This novel approach allows the reaction to proceed efficiently in a water-based system, which not only improves safety profiles by eliminating flammable organic solvents but also simplifies the downstream processing to a mere layer separation and recrystallization. The result is a robust, scalable process that delivers yields reaching 89%, representing a substantial leap forward in manufacturing efficiency for this key pharmaceutical building block.
Mechanistic Insights into SDS-Mediated Phase-Transfer Oxidative Cyclization
The success of this synthetic route hinges on the intricate interplay between the surfactant SDS and the polyoxometalate oxidant within a biphasic solvent system. In this mechanism, SDS molecules aggregate at the interface of the organic (2-butanol) and aqueous phases, forming micelles that solubilize the organic reactants, m-chloroaniline and crotonaldehyde, within the aqueous continuum. This dramatic increase in interfacial surface area facilitates a much higher frequency of effective molecular collisions between the reactants and the phosphomolybdic acid oxidant. The phosphomolybdic acid, possessing both acidic and oxidative properties, protonates the intermediate species to facilitate cyclization while simultaneously accepting electrons to drive the aromatization of the quinoline ring. This dual functionality eliminates the need for exogenous strong acids, which often lead to side reactions or equipment corrosion, and ensures a smoother progression through the transition states required for ring closure. The specific orientation of reactants within the SDS micelles is believed to sterically hinder the attack at the 5-position, thereby enforcing regioselectivity towards the thermodynamically stable 7-chloroquinaldine product.
Controlling the impurity profile is paramount for pharmaceutical intermediates, and this mechanism offers superior selectivity compared to homogeneous acid catalysis. In conventional acidic media, the high concentration of protons can lead to non-selective polymerization of crotonaldehyde or indiscriminate electrophilic attacks on the aniline ring, resulting in a complex soup of tars and isomers. However, the micellar environment provided by the phase-transfer catalyst creates a distinct local pH and polarity that stabilizes the specific transition state leading to the 7-isomer. Furthermore, the use of phosphomolybdic acid avoids the introduction of halide ions or heavy metals often associated with other oxidants like chloranil or nitrobenzene, resulting in a cleaner crude product that requires less aggressive purification. This inherent purity is critical for downstream coupling reactions in the synthesis of leukotriene receptor antagonists, where trace impurities can poison subsequent catalysts or fail stringent regulatory toxicology screens.
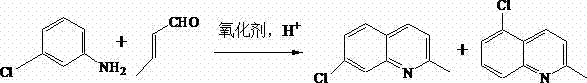
How to Synthesize 7-Chloroquinaldine Efficiently
Implementing this phase-transfer catalytic protocol requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the micellar system. The process begins by dissolving m-chloroaniline in 2-butanol, followed by the addition of the surfactant and oxidant into the aqueous mixture, establishing the biphasic catalytic environment. Subsequent dropwise addition of crotonaldehyde at controlled temperatures between 80°C and 95°C ensures that the exothermic cyclization proceeds without runaway reactions, maintaining the integrity of the micellar structure. Detailed standard operating procedures regarding exact mixing times, layer separation techniques, and recrystallization solvents are essential for reproducibility. For a comprehensive, step-by-step guide on executing this synthesis in a pilot or production plant setting, please refer to the standardized protocol below.
- Dissolve m-chloroaniline in 2-butanol, then add Sodium Dodecyl Sulfate (SDS) and phosphomolybdic acid to the aqueous/organic mixed solvent system.
- Dropwise add crotonaldehyde to the mixture at 80-95°C and maintain reaction for 3-4 hours to ensure complete cyclization.
- Separate layers, recover organic phase, neutralize aqueous phase with NaHCO3, and recrystallize to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phase-transfer catalytic method translates directly into tangible operational efficiencies and risk mitigation. The shift from hazardous, flammable solvents like THF to a predominantly water-based system significantly lowers the cost of goods sold by reducing expenditure on specialized solvent recovery infrastructure and fire safety compliance measures. Moreover, the elimination of expensive and toxic oxidants like chloranil in favor of phosphomolybdic acid, coupled with the high atom economy of the reaction, drives down raw material costs per kilogram of finished product. The simplified workup procedure, which avoids energy-intensive continuous vacuum distillation, further reduces utility consumption and shortens the batch cycle time, allowing for increased throughput within existing reactor vessels. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands for asthma medication intermediates without the bottlenecks associated with complex purification trains.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the reagent profile and the drastic reduction in waste treatment costs. By utilizing SDS, a commodity surfactant, to replace expensive phase-transfer catalysts like OP-10 or complex separating agents like tartrates, the direct material cost is significantly lowered. Additionally, the high yield of 89% minimizes the loss of valuable starting materials, ensuring that a greater proportion of input mass is converted into saleable product. The avoidance of heavy metal salts for purification also reduces the cost associated with hazardous waste disposal and environmental compliance fees, leading to substantial overall cost savings in pharma intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents or solvents with volatile market prices, but this method utilizes widely available, bulk chemicals. Sodium dodecyl sulfate and 2-butanol are commodity chemicals with stable global supply chains, reducing the risk of production stoppages due to raw material shortages. The robustness of the water-based system also means that the process is less sensitive to minor fluctuations in moisture content or ambient conditions, ensuring consistent batch-to-bquality. This reliability allows supply chain planners to forecast production schedules with greater confidence, reducing lead time for high-purity pharmaceutical intermediates and securing long-term contracts with downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new engineering challenges, yet this methodology is inherently designed for expansion due to its benign solvent system. The use of water as the primary reaction medium eliminates the massive fire hazards associated with scaling up reactions in flammable organic solvents, simplifying the regulatory approval process for new production lines. Furthermore, the reduced generation of organic waste and the absence of heavy metal contaminants align perfectly with modern green chemistry principles and strict environmental regulations. This ease of scalability ensures that commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly, moving from laboratory grams to multi-ton annual production without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply networks. The following questions address common concerns regarding isomer control, safety protocols, and industrial feasibility based on the specific disclosures in the patent literature. These insights are intended to clarify how this technology outperforms legacy methods in both chemical efficiency and operational safety. For further technical validation, our team can provide detailed comparative data sheets upon request.
Q: How does this method improve upon traditional Skraup reactions for 7-Chloroquinaldine?
A: Traditional Skraup reactions often suffer from the formation of 5-position isomers, requiring complex separation steps using ZnCl2 or tartrates. This novel phase-transfer catalytic method utilizes SDS to enhance emulsification, effectively suppressing the generation of the 5-isomer byproduct and significantly improving reaction efficiency and yield up to 89%.
Q: What are the safety advantages of using phosphomolybdic acid over chloranil?
A: Prior art methods utilizing chloranil as an oxidant often require flammable solvents like tetrahydrofuran (THF) and involve harsh processing conditions. The use of phosphomolybdic acid in this invention serves a dual function as both an acid and an oxidant, eliminating the need for traditional acid solvents and allowing the reaction to proceed safely in a water-based system.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for industrial scale-up. Unlike previous phase-transfer methods that required continuous vacuum distillation leading to product loss, this method features a simple post-treatment involving layer separation and recrystallization. The use of water as a primary solvent also reduces environmental impact and waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Chloroquinaldine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this phase-transfer catalytic method are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature profiles required for this synthesis, maintaining stringent purity specifications to meet the exacting standards of the global pharmaceutical industry. With our rigorous QC labs and commitment to process optimization, we guarantee a supply of 7-chloroquinaldine that is not only cost-competitive but also consistently reliable for your critical drug development pipelines.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of this essential pharmaceutical intermediate.
