Advanced Phase-Transfer Catalysis for High-Purity 7-Chloroquinaldine Production and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust, scalable pathways for critical intermediates, and the synthesis of 7-chloroquinaldine stands as a pivotal challenge in the production of leukotriene receptor antagonists like Montelukast. Patent CN103524408A introduces a transformative approach to this synthesis, leveraging a sophisticated phase-transfer catalytic reaction that fundamentally alters the economic and technical landscape of manufacturing this key building block. By utilizing m-chloroaniline and crotonaldehyde as primary feedstocks within a novel water and 2-butanol reaction system, this method achieves exceptional yields reaching up to 89%, significantly outperforming historical benchmarks. The core innovation lies in the synergistic application of lauryl sodium sulfate (SDS) as a surfactant and phosphomolybdic acid as a multifunctional oxidant, which collectively suppress the formation of troublesome 5-position isomers while eliminating the need for hazardous, traditional acid solvents. For R&D directors and procurement strategists alike, this patent represents a critical opportunity to optimize supply chains for high-purity pharmaceutical intermediates, offering a route that is not only chemically superior but also inherently safer and more cost-effective for commercial deployment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 7-chloroquinaldine has been plagued by significant technical hurdles that compromise both yield and operational safety, creating bottlenecks for reliable API intermediate suppliers. The classical Skraup reaction and Doebner-Miller modifications, while foundational, frequently result in complex mixtures containing substantial amounts of 5-position isomers, necessitating arduous purification steps involving zinc chloride, tartrates, or 4-nitrophthalic acid to isolate the desired product. Furthermore, prior art such as US Patent 5126456 relies on chloranil as an oxidant in aqueous isopropanol with hydrogen chloride, a process that not only yields a modest 67% but also demands the use of large quantities of flammable tetrahydrofuran (THF) for separation, introducing severe safety risks and high solvent recovery costs. Other attempts, such as those disclosed in CN 101638382 using alkylphenol polyoxyethylene catalysts, suffer from complicated post-reaction treatments requiring continuous vacuum distillation, leading to serious product loss and rendering them unsuitable for efficient suitability for industrialized production. These legacy methods impose heavy burdens on manufacturing facilities through excessive waste generation, high energy consumption for solvent recovery, and inconsistent batch quality due to isomer contamination.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN103524408A offers a streamlined, high-efficiency pathway that directly addresses the痛点 (pain points) of cost reduction in pharmaceutical intermediates manufacturing. By introducing Sodium Dodecyl Sulfate (SDS) into the reaction matrix, the process dramatically enhances the emulsibility of the reaction liquid, creating a micro-environment that kinetically favors the formation of the 7-isomer while effectively inhibiting the generation of the unwanted 5-isomer byproduct. Simultaneously, the inclusion of phosphomolybdic acid serves a dual purpose: it acts as a potent oxidant to drive the cyclization forward while possessing the general properties of an acid, thereby completely avoiding the use of corrosive traditional acid solvents. This elegant chemical design allows the reaction to proceed smoothly in a water-based system, which drastically simplifies the aftertreatment process; instead of complex distillations, the product can be isolated through simple layering, washing, and recrystallization. The result is a robust protocol that delivers yields up to 89%, providing a compelling value proposition for any organization seeking a reliable 7-chloroquinaldine supplier with a focus on sustainability and operational excellence.
Mechanistic Insights into SDS-Mediated Phase-Transfer Oxidative Cyclization
To fully appreciate the technical superiority of this route, one must delve into the mechanistic interplay between the surfactant and the oxidant within the biphasic system. The addition of SDS is not merely a solubility aid; it functions as a critical phase-transfer agent that organizes the reactants at the interface of the aqueous and organic phases. In this micellar environment, the local concentration of m-chloroaniline and crotonaldehyde is significantly increased, facilitating the initial condensation steps required to form the dihydroquinoline intermediate. More importantly, the specific steric and electronic environment created by the SDS micelles appears to exert a directing effect on the cyclization, sterically hindering the attack at the 5-position and thus suppressing the formation of the isomeric impurity that plagues conventional acid-catalyzed routes. This selectivity is paramount for achieving high-purity 7-chloroquinaldine without the need for downstream chromatographic separations, which are often cost-prohibitive at scale.
Complementing the surfactant is the role of phosphomolybdic acid, a heteropoly acid that acts as a green, reusable oxidant. Unlike chloranil, which generates stoichiometric amounts of chlorinated organic waste, phosphomolybdic acid participates in the redox cycle while maintaining the acidic pH necessary for the dehydration and aromatization steps of the quinoline ring formation. The mechanism likely involves the oxidation of the dihydro-intermediate to the aromatic quinoline system, driven by the high oxidation potential of the molybdenum centers. Because this reagent also provides the requisite protons for the reaction, the system avoids the accumulation of inorganic salts typically associated with neutralizing strong mineral acids post-reaction. This dual functionality simplifies the reaction mass balance and reduces the E-factor of the process, aligning perfectly with modern green chemistry principles demanded by top-tier pharmaceutical buyers.
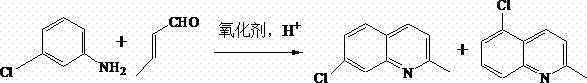
How to Synthesize 7-Chloroquinaldine Efficiently
Implementing this synthesis requires precise control over reagent ratios and thermal conditions to maximize the benefits of the phase-transfer system. The process begins by dissolving m-chloroaniline in 2-butanol, followed by the addition of SDS and phosphomolybdic acid to create a stable emulsion with water. Crotonaldehyde is then introduced dropwise to manage the exotherm and ensure controlled addition, with the reaction maintained between 80°C and 95°C for a duration of 3 to 4 hours. Following the reaction, the mixture naturally separates into phases, allowing for the recovery of the organic solvent and the isolation of the product from the aqueous layer through simple basification and recrystallization. For detailed standard operating procedures and specific parameter optimization, please refer to the step-by-step guide below.
- Dissolve m-chloroaniline in 2-butanol, then add Sodium Dodecyl Sulfate (SDS) and phosphomolybdic acid to form a mixed solvent system with water.
- Dropwise add crotonaldehyde to the mixture while maintaining a temperature between 80°C and 95°C for 3 to 4 hours.
- Separate the organic phase, recover 2-butanol, wash the aqueous residue with NaHCO3 solution, and recrystallize to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology offers profound strategic advantages that extend far beyond simple yield improvements. The elimination of expensive and hazardous reagents like chloranil and THF directly translates to substantial cost savings in raw material procurement and waste disposal, addressing the critical need for cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the shift to an aqueous-based system significantly lowers the fire hazard profile of the manufacturing facility, potentially reducing insurance premiums and regulatory compliance burdens associated with volatile organic compound (VOC) emissions. The simplified workup procedure, which avoids energy-intensive vacuum distillation steps, enhances the overall throughput of the production line, allowing for faster turnaround times and greater flexibility in meeting fluctuating market demands for high-purity API intermediates.
- Cost Reduction in Manufacturing: The replacement of costly oxidants like chloranil with phosphomolybdic acid, combined with the use of inexpensive surfactants like SDS, drastically lowers the bill of materials for each production batch. Additionally, the ability to recycle the 2-butanol solvent and the avoidance of complex purification columns for isomer removal significantly reduce operational expenditures, ensuring a more competitive pricing structure for the final product without compromising on quality standards.
- Enhanced Supply Chain Reliability: By utilizing readily available commodity chemicals such as m-chloroaniline and crotonaldehyde, this process mitigates the risk of supply disruptions often associated with specialty reagents. The robustness of the aqueous system ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed quality control tests, thereby securing a steady flow of materials for downstream API synthesis and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The inherent safety of the water-based reaction medium facilitates easier commercial scale-up of complex pharmaceutical intermediates, as heat management and mixing are more predictable compared to heterogeneous organic systems. Moreover, the reduction in hazardous waste generation and the absence of halogenated solvents align with increasingly stringent global environmental regulations, future-proofing the supply chain against evolving sustainability mandates and enhancing the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phase-transfer catalytic method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this technology resolves historical manufacturing challenges. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific supply chain requirements.
Q: How does this method improve upon traditional Skraup reactions for 7-chloroquinaldine?
A: Traditional Skraup reactions often suffer from the formation of 5-position isomers and require harsh acidic conditions with difficult separations. This patented method utilizes SDS to inhibit isomer formation and phosphomolybdic acid as a dual-function oxidant/acid, allowing for a cleaner reaction in a water-based system with yields up to 89%.
Q: What are the environmental benefits of using phosphomolybdic acid in this process?
A: Phosphomolybdic acid serves as both the oxidant and the acid source, eliminating the need for separate strong mineral acids and expensive oxidants like chloranil. This simplifies the waste stream, reduces the reliance on hazardous organic solvents like THF, and facilitates easier aqueous workup procedures.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It avoids complex vacuum distillation steps required by previous phase-transfer methods (like those using OP-10) and utilizes common, inexpensive reagents like SDS and 2-butanol, making it economically viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Chloroquinaldine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN103524408A is crucial for maintaining a competitive edge in the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield, aqueous-phase process are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 7-chloroquinaldine meets the exacting standards required for the synthesis of life-saving medications like Montelukast, providing our partners with absolute confidence in material quality.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this greener, more efficient route. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your production of critical quinoline intermediates is both economically optimized and technically secure for the long term.
