Advanced Phase-Transfer Catalysis for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic viability, particularly for critical intermediates like 7-Chloroquinaldine. Patent CN103524408A introduces a transformative approach to this synthesis, leveraging phase-transfer catalytic reactions to overcome longstanding limitations in yield and selectivity. This innovation utilizes m-chloroaniline and crotonaldehyde as primary feedstocks within a unique water and 2-butanol reaction system, facilitated by sodium dodecyl sulfate (SDS) and phosphomolybdic acid. By shifting away from traditional harsh acidic environments to a more controlled biphasic system, this method achieves reaction yields reaching up to 89%, a substantial improvement over historical benchmarks. For R&D directors and procurement specialists, this represents a pivotal opportunity to optimize the supply chain for antiasthmatic agents and LTRA MK-0679 precursors. The integration of surfactant-enhanced emulsification not only streamlines the reaction kinetics but also fundamentally alters the impurity profile, ensuring a cleaner crude product that requires less intensive downstream purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-Chloroquinaldine has relied heavily on classical Skraup or Doebner-Miller reactions, which are plagued by significant technical and economic inefficiencies. These traditional pathways typically involve reacting m-chloroaniline with crotonaldehyde under strongly acidic conditions, often necessitating the use of zinc chloride, tartrates, or 4-nitrophthalic acid to manage the reaction environment. A critical failure point in these legacy processes is the uncontrollable generation of the 5-position isomer, a structural analog that is chemically similar and notoriously difficult to separate from the desired 7-isomer. Prior art, such as US Patent 5126456, attempted to address oxidation using chloranil in aqueous isopropanol, yet this resulted in mediocre yields of approximately 67% and introduced severe safety hazards due to the requirement for large volumes of flammable tetrahydrofuran (THF) during the separation phase. Furthermore, the reliance on chloranil as an oxidant imposes a heavy cost burden due to its high market price and toxicological profile, creating substantial waste disposal challenges that conflict with modern green chemistry mandates.
The Novel Approach
The methodology disclosed in CN103524408A fundamentally reengineers the reaction landscape by introducing a phase-transfer catalytic system that operates efficiently in an aqueous medium. By incorporating sodium lauryl sulfate (SDS) as a surfactant, the process dramatically enhances the emulsibility of the reaction liquid, creating a micro-environment that favors the formation of the target 7-chloroquinaldine while kinetically inhibiting the generation of the unwanted 5-isomer byproduct. Simultaneously, the substitution of traditional acid solvents with phosphomolybdic acid serves a dual purpose: it provides the necessary acidic protons for cyclization while acting as a potent, reusable oxidant. This strategic combination allows the reaction to proceed in a water and 2-butanol mixture, eliminating the need for hazardous volatile organic compounds and simplifying the post-reaction workup to a straightforward phase separation and distillation. The result is a process that not only boosts yield to nearly 90% but also aligns with stringent environmental regulations, offering a reliable pharmaceutical intermediate supplier pathway that is both economically and ecologically superior.
Mechanistic Insights into SDS-Mediated Phase-Transfer Oxidative Cyclization
The core innovation of this synthesis lies in the synergistic interaction between the surfactant SDS and the polyoxometalate oxidant within the biphasic solvent system. In conventional homogeneous acid catalysis, the reactants often suffer from poor solubility matching, leading to localized hot spots that promote side reactions and isomerization. However, the addition of SDS creates stable micelles that encapsulate the organic reactants, effectively increasing their local concentration at the interface of the aqueous and organic phases. This emulsification effect ensures a more uniform distribution of the oxidizing species, phosphomolybdic acid, throughout the reaction matrix. The micellar environment appears to sterically hinder the transition states leading to the 5-position isomer, thereby directing the cyclization exclusively towards the thermodynamically favored 7-position. This selectivity mechanism is crucial for maintaining high purity without the need for complex chromatographic separations, which are often cost-prohibitive at an industrial scale.
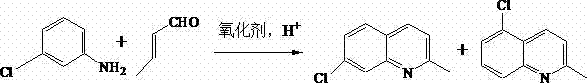
Furthermore, the role of phosphomolybdic acid extends beyond simple oxidation; its polyacidic nature allows it to buffer the reaction pH effectively, preventing the degradation of sensitive intermediates that often occurs with strong mineral acids. The redox potential of the phosphomolybdate cluster is sufficiently high to drive the oxidative aromatization of the dihydroquinoline intermediate but mild enough to avoid over-oxidation of the methyl group or the chloro-substituent. This precise control over the oxidation state minimizes the formation of tarry byproducts and polymeric residues, which are common issues in Skraup-type syntheses. For process chemists, understanding this mechanistic nuance is vital for scaling the reaction, as it suggests that maintaining the integrity of the micellar structure and the oxidation state of the catalyst is key to reproducing the high yields observed in the patent embodiments. The ability to recycle the aqueous phase containing the catalyst further enhances the atom economy of the process, reducing the overall E-factor of the manufacturing operation.
How to Synthesize 7-Chloroquinaldine Efficiently
The practical implementation of this phase-transfer catalytic method offers a clear roadmap for laboratories aiming to transition from bench-scale discovery to pilot production. The protocol begins with the dissolution of m-chloroaniline in 2-butanol, followed by the introduction of the surfactant and oxidant into the aqueous-organic mix, establishing the reactive micro-emulsion prior to the addition of the aldehyde. This sequential addition is critical to ensure the catalytic system is fully formed before the exothermic condensation begins. Detailed standardized synthesis steps, including precise molar ratios and temperature ramping profiles, are essential for maximizing the 89% yield potential described in the patent data. Operators must pay close attention to the droplet size of the emulsion and the rate of crotonaldehyde addition, as these parameters directly influence the isomer ratio and final purity.
- Dissolve m-chloroaniline in 2-butanol and add Sodium Dodecyl Sulfate (SDS) and phosphomolybdic acid to the aqueous-organic mixed solvent system.
- Dropwise add crotonaldehyde to the mixture and maintain reaction temperature between 80°C and 95°C for 3 to 4 hours.
- Separate the organic phase, recover 2-butanol, wash the aqueous layer with NaHCO3 solution, and recrystallize to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation strategies. The shift from expensive, hazardous reagents like chloranil and THF to commodity chemicals like SDS and phosphomolybdic acid drastically simplifies the raw material sourcing landscape. This reduction in specialized chemical dependency lowers the barrier to entry for multiple suppliers, fostering a more competitive and resilient supply chain for high-purity pharmaceutical intermediates. Moreover, the elimination of flammable solvents reduces the regulatory burden associated with storage and transport, lowering insurance premiums and facility compliance costs. The simplified workup procedure, which relies on basic distillation and washing rather than complex extraction or chromatography, significantly shortens the batch cycle time, allowing for higher throughput in existing manufacturing assets without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic impact of replacing chloranil with phosphomolybdic acid cannot be overstated, as the latter is not only cheaper but also serves a dual function that eliminates the need for additional acid catalysts. By avoiding the use of THF and other volatile organic solvents, the process reduces solvent recovery costs and minimizes losses due to evaporation, leading to substantial cost savings in the overall bill of materials. The high selectivity of the reaction means that less starting material is wasted on byproduct formation, directly improving the effective yield per kilogram of input. Additionally, the ability to operate in an aqueous system reduces the load on solvent recovery units and lowers the energy consumption associated with drying and distilling large volumes of organic waste.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically m-chloroaniline and crotonaldehyde, are bulk commodity chemicals with well-established global supply chains, ensuring consistent availability and price stability. Unlike specialized oxidants that may face supply bottlenecks, the reagents used in this phase-transfer process are readily accessible from multiple vendors, reducing the risk of production stoppages due to material shortages. The robustness of the aqueous reaction system also means that the process is less sensitive to minor fluctuations in raw material quality, providing a buffer against supply chain variability. This reliability is critical for maintaining continuous production schedules for downstream API manufacturing, ensuring that customers receive their orders on time without unexpected delays.
- Scalability and Environmental Compliance: From an environmental perspective, the move to a water-based system significantly reduces the volume of hazardous waste generated, simplifying effluent treatment and lowering disposal fees. The absence of heavy metal catalysts or toxic oxidants like chloranil aligns the process with increasingly strict environmental regulations, future-proofing the manufacturing site against tighter compliance standards. The simplicity of the workup, involving phase separation and crystallization, is inherently easier to scale than complex multi-step extractions, allowing for seamless transition from pilot plants to multi-ton commercial production. This scalability ensures that as demand for the final drug product grows, the supply of the intermediate can be ramped up quickly without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phase-transfer catalytic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy synthesis routes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new process for their specific manufacturing lines.
Q: How does the new method improve yield compared to traditional Skraup reactions?
A: Traditional methods often suffer from low yields (around 67%) due to the formation of difficult-to-separate 5-position isomers and the use of harsh conditions. The patented phase-transfer method utilizes SDS to enhance emulsification and suppress isomer formation, achieving yields up to 89%.
Q: What are the environmental advantages of using phosphomolybdic acid in this process?
A: Phosphomolybdic acid serves a dual function as both an acid catalyst and an oxidant. This eliminates the need for separate strong acid solvents and expensive, toxic oxidants like chloranil, significantly simplifying waste treatment and reducing hazardous chemical usage.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It operates in a water and 2-butanol system which avoids flammable solvents like THF used in prior art. The workup involves simple phase separation and distillation, making it robust for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Chloroquinaldine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-yield synthetic routes in the modern pharmaceutical landscape. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the phase-transfer synthesis of 7-Chloroquinaldine are translated into reliable industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API intermediate manufacturing. We understand that consistency is key, and our process engineering teams are dedicated to optimizing every variable, from emulsion stability to crystallization kinetics, to deliver a product that exceeds expectations.
We invite forward-thinking partners to collaborate with us on leveraging this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest standards of quality and efficiency available in the market.
