Advanced Synthesis and Purification of 5-Isosorbide Mononitrate for Industrial Scale-Up
The global demand for cardiovascular therapeutics continues to drive the need for efficient, scalable manufacturing processes for key active pharmaceutical ingredients (APIs) and their intermediates. Patent CN103641840A, published in March 2014, introduces a significant technological advancement in the synthesis and purification of 5-isosorbide mononitrate (5-ISMN), a critical vasodilator used extensively in the treatment of angina pectoris and heart failure. This patent addresses long-standing challenges in the industry regarding yield optimization, impurity control, and process safety. By shifting away from complex multi-step protection strategies or expensive catalytic reductions, the disclosed method utilizes a direct nitration approach coupled with a novel purification sequence involving selective crystallization and salt precipitation. For R&D directors and process chemists, this represents a viable pathway to enhance production efficiency while maintaining the rigorous purity profiles required by regulatory bodies such as the FDA and EMA.
For procurement managers and supply chain leaders, the implications of this technology are profound. The reliance on commodity chemicals such as nitric acid, acetic anhydride, and sodium hydroxide reduces dependency on specialized, high-cost catalysts that often suffer from supply volatility. Furthermore, the elimination of chromatographic purification steps drastically lowers the operational expenditure (OpEx) associated with solvent consumption and waste disposal. As the pharmaceutical industry increasingly prioritizes green chemistry and cost-effective manufacturing, the methodology outlined in CN103641840A offers a robust framework for the commercial scale-up of complex pharmaceutical intermediates. This report analyzes the technical nuances of this process, contrasting it with conventional routes to highlight its potential for reducing lead time for high-purity pharmaceutical intermediates and ensuring a stable supply chain for downstream API manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 5-isosorbide mononitrate has been plagued by inefficiencies inherent in traditional chemical routes. One prevalent method involves an indirect nitration strategy, often depicted in prior art such as 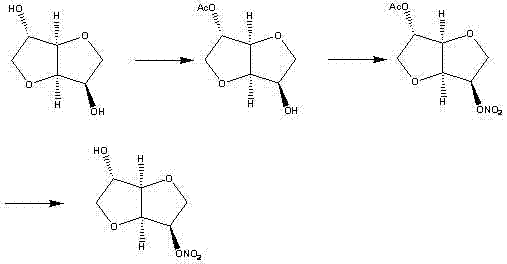 . This approach typically requires the protection of one hydroxyl group on the isosorbide backbone, followed by nitration of the remaining free hydroxyl, and finally, a deprotection step to reveal the target mononitrate. While chemically sound, this route is excessively long, involving multiple unit operations that accumulate yield losses at each stage. The use of protecting groups necessitates additional reagents and generates substantial chemical waste, complicating the environmental footprint of the manufacturing process. Moreover, the selective acylation required in the initial steps is difficult to control precisely, often leading to mixtures of mono- and di-substituted products that are challenging to separate without resorting to costly purification techniques.
. This approach typically requires the protection of one hydroxyl group on the isosorbide backbone, followed by nitration of the remaining free hydroxyl, and finally, a deprotection step to reveal the target mononitrate. While chemically sound, this route is excessively long, involving multiple unit operations that accumulate yield losses at each stage. The use of protecting groups necessitates additional reagents and generates substantial chemical waste, complicating the environmental footprint of the manufacturing process. Moreover, the selective acylation required in the initial steps is difficult to control precisely, often leading to mixtures of mono- and di-substituted products that are challenging to separate without resorting to costly purification techniques.
Another common avenue explored in the literature is the selective reduction of isosorbide dinitrate, as illustrated in 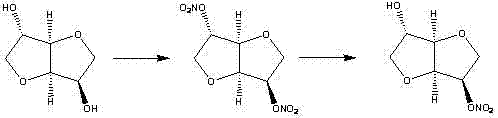 . This route attempts to convert the readily available dinitrate into the desired mononitrate using specific reducing agents or catalysts. However, achieving perfect chemoselectivity is notoriously difficult; incomplete reduction often leaves residual dinitrate or produces the unwanted 2-isosorbide mononitrate isomer. Separating these structural isomers typically demands column chromatography, a technique that is economically prohibitive on a multi-ton scale due to high solvent usage and low throughput. Additionally, some reduction methods employ expensive transition metal catalysts or hazardous reagents, introducing safety risks and supply chain vulnerabilities that are unacceptable for modern, sustainable pharmaceutical manufacturing.
. This route attempts to convert the readily available dinitrate into the desired mononitrate using specific reducing agents or catalysts. However, achieving perfect chemoselectivity is notoriously difficult; incomplete reduction often leaves residual dinitrate or produces the unwanted 2-isosorbide mononitrate isomer. Separating these structural isomers typically demands column chromatography, a technique that is economically prohibitive on a multi-ton scale due to high solvent usage and low throughput. Additionally, some reduction methods employ expensive transition metal catalysts or hazardous reagents, introducing safety risks and supply chain vulnerabilities that are unacceptable for modern, sustainable pharmaceutical manufacturing.
The Novel Approach
The methodology presented in CN103641840A circumvents these bottlenecks through a clever manipulation of solubility and reactivity. Instead of avoiding the formation of the dinitrate entirely, the process allows for direct nitration of isosorbide using a mixed acid system (nitric acid, acetic acid, and acetic anhydride) under controlled low-temperature conditions. The breakthrough lies in the workup procedure: upon quenching the reaction with water and cooling the mixture to 0-5°C, the 2,5-isosorbide dinitrate byproduct selectively crystallizes out of the solution. This physical separation allows for the removal of the major impurity via simple filtration, bypassing the need for complex chromatographic columns. The remaining filtrate, enriched in the mononitrate, is then treated with sodium hydroxide to form a sodium salt complex, which precipitates as a white solid. This salt formation step serves as a second purification barrier, effectively isolating the target molecule from other organic impurities before the final hydrolysis and recrystallization steps yield the high-purity product.
Mechanistic Insights into Direct Nitration and Selective Precipitation
From a mechanistic perspective, the success of this synthesis relies on the differential reactivity of the two hydroxyl groups on the isosorbide scaffold—isosorbide possesses both an endo and an exo hydroxyl group. The exo-hydroxyl group is sterically more accessible and generally more reactive towards electrophilic attack by the nitronium ion generated in the acetic anhydride/nitric acid mixture. By carefully controlling the molar ratio of nitric acid to isosorbide (optimized between 1:1 and 1.5:1) and maintaining the reaction temperature between 5°C and 20°C, the process kinetically favors the formation of the mononitrate while minimizing over-nitration. However, since some dinitrate is inevitably formed, the subsequent purification strategy is critical. The addition of water serves a dual purpose: it quenches the excess nitrating agent and alters the solvent polarity, drastically reducing the solubility of the more lipophilic dinitrate species. This induces the crystallization of the 2,5-dinitrate, allowing it to be mechanically separated from the mononitrate which remains in the aqueous/organic phase.
Following the removal of the dinitrate, the conversion of the mononitrate into its sodium salt is a masterstroke of process chemistry. The isosorbide mononitrate reacts with sodium hydroxide to form a water-soluble sodium alkoxide intermediate which, under the specific conditions described (low temperature and high pH), precipitates as a hydrate complex. This step effectively 'locks' the mononitrate into a solid form while leaving neutral organic impurities in the solution. The subsequent filtration of this salt ensures that the material entering the final hydrolysis step is already of high purity. Finally, acidification with hydrochloric acid regenerates the free alcohol/nitrate form, which is then extracted and recrystallized. The patent specifies the use of ethyl acetate and petroleum ether or dichloromethane for recrystallization, solvents chosen for their ability to form needle-like crystals with high lattice purity, effectively excluding trace isomers and achieving purity levels exceeding 99% as confirmed by HPLC analysis in the experimental examples.
How to Synthesize 5-Isosorbide Mononitrate Efficiently
The synthesis protocol detailed in the patent provides a clear, step-by-step guide for replicating this high-efficiency route in a pilot or production setting. The process begins with the in-situ preparation of the nitrating agent, ensuring fresh reactivity and minimizing the storage hazards associated with pre-mixed nitrating acids. Operators must strictly adhere to the temperature controls during the addition of nitric acid to the acetic anhydride/acetic acid mixture to prevent thermal runaway. Following the nitration of isosorbide, the quenching and crystallization phases require precise temperature management to maximize the recovery of the dinitrate byproduct, thereby driving the equilibrium towards the desired mononitrate in the filtrate. The detailed standardized synthesis steps see the guide below.
- Prepare a nitrating agent by mixing acetic acid, acetic anhydride, and concentrated nitric acid at low temperatures (-5°C to 10°C), then react with isosorbide.
- Quench the reaction with water and cool to 0-5°C to selectively crystallize and filter out the 2,5-isosorbide dinitrate byproduct.
- Treat the filtrate with sodium hydroxide to precipitate the isosorbide mononitrate sodium salt, filter, hydrolyze with acid, and recrystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The most significant advantage lies in the drastic simplification of the downstream processing train. By eliminating the need for column chromatography, manufacturers can significantly reduce the volume of solvents required per kilogram of product. This reduction directly translates to lower costs associated with solvent purchase, recovery, and disposal, which are often the largest variable costs in fine chemical manufacturing. Furthermore, the removal of chromatography increases the throughput capacity of existing facilities, as batch cycles are shortened and equipment occupancy time is reduced. This efficiency gain allows for a more responsive supply chain capable of meeting fluctuating market demands for cardiovascular medications without the need for massive capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the substitution of expensive, specialized reagents with commodity chemicals. Traditional routes often rely on precious metal catalysts or complex protecting group reagents that carry high price tags and supply risks. In contrast, this method utilizes nitric acid, acetic anhydride, and sodium hydroxide, which are globally available bulk chemicals with stable pricing. Additionally, the ability to isolate the product via crystallization and filtration rather than distillation or chromatography reduces energy consumption. The overall process design minimizes waste generation, aligning with modern environmental regulations and reducing the financial burden of waste treatment compliance, thereby delivering substantial cost savings in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability of raw materials. Since this synthesis route avoids niche catalysts that might be sourced from single suppliers, it mitigates the risk of production stoppages due to raw material shortages. The robustness of the chemistry also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when sourcing from different vendors. This flexibility allows procurement teams to negotiate better terms with multiple suppliers, fostering a competitive purchasing environment. Moreover, the simplified workflow reduces the number of intermediate storage requirements, lowering inventory holding costs and minimizing the risk of degradation or contamination during prolonged storage periods.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden complexities, particularly regarding heat transfer and mixing in heterogeneous systems. This patent's method relies on homogeneous reactions followed by straightforward solid-liquid separations, which are inherently easier to scale than multiphase catalytic reactions or delicate chromatographic separations. The use of standard filtration and crystallization equipment ensures that the technology can be transferred to existing manufacturing lines with minimal modification. From an environmental standpoint, the reduction in solvent usage and the avoidance of heavy metal catalysts simplify the effluent treatment process. This facilitates easier compliance with increasingly stringent environmental discharge standards, reducing the regulatory risk profile for the manufacturing site and ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adoption. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios.
Q: How does this process remove the dinitrate impurity without chromatography?
A: The process utilizes a selective crystallization step where water is added to the reaction mixture at 0-5°C. Under these specific conditions, the 2,5-isosorbide dinitrate byproduct precipitates out as a solid and is removed by filtration before the mononitrate is isolated.
Q: What represents the key cost-saving advantage of this synthesis route?
A: The primary economic advantage is the elimination of column chromatography. Traditional methods often require expensive silica gel columns for purification, whereas this method relies on crystallization and salt formation, which are significantly cheaper and easier to scale industrially.
Q: What purity levels can be achieved with this recrystallization method?
A: Experimental data from the patent indicates that following the extraction and recrystallization steps using solvents like ethyl acetate and petroleum ether, the final product purity can exceed 99%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Isosorbide Mononitrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN103641840A process are fully realized in a GMP-compliant environment. We understand that consistency is key in the pharmaceutical supply chain, which is why our rigorous QC labs enforce stringent purity specifications on every batch of 5-isosorbide mononitrate we produce. By leveraging our state-of-the-art facilities and technical know-how, we can deliver this critical cardiovascular intermediate with the reliability and quality that global drug manufacturers demand.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for 5-ISMN and related compounds. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Whether you are looking to secure a secondary source for an existing product or develop a new supply line for a novel formulation, NINGBO INNO PHARMCHEM is positioned to be your strategic partner in growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
