Revolutionizing 5-Isosorbide Mononitrate Production via Selective TMSCl-Mediated Nitration Technology
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for safer, more efficient synthesis of critical active ingredients. A pivotal advancement in this domain is detailed in patent CN110407846B, which discloses a highly selective preparation method for 5-isosorbide mononitrate (5-ISMN), a vital metabolite used in the treatment of angina pectoris and coronary heart disease. This technology addresses long-standing challenges in nitration chemistry, offering a pathway that bypasses the hazardous and low-yielding traditional routes. By leveraging a unique combination of trimethylchlorosilane (TMSCl) and aluminum trichloride, the inventors have achieved a breakthrough in regioselectivity, ensuring that the nitro group is installed exclusively at the 5-position of the isosorbide backbone. For global procurement leaders and R&D directors, this represents not just a chemical improvement, but a strategic opportunity to secure a more reliable supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-isosorbide mononitrate has been plagued by significant technical hurdles that impact both cost and safety profiles. The earliest methods, such as those described by Hayward L D, relied on direct nitration using fuming nitric acid. As illustrated in the reaction scheme below, this brute-force approach lacks specificity, generating a chaotic mixture of 2-isosorbide mononitrate, 5-isosorbide mononitrate, isosorbide dinitrate, and unreacted starting material.
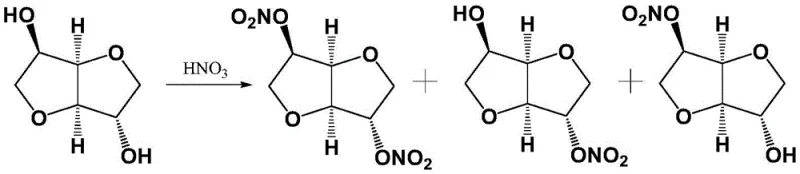
Separating the desired 5-isomer from this complex matrix traditionally requires energy-intensive column chromatography or repeated recrystallization, which drastically reduces overall yield and increases solvent waste. Furthermore, the formation of isosorbide dinitrate introduces severe safety risks, as these polynitrated species are thermally unstable and prone to explosion under heating or pressurization, rendering such processes unsuitable for modern, safe industrial manufacturing. Alternative esterification-nitration routes, while attempting to mitigate some issues, often involve multi-step sequences with expensive reagents like acetic anhydride and pyridine, followed by hydrolysis, which complicates the workflow and inflates production costs.
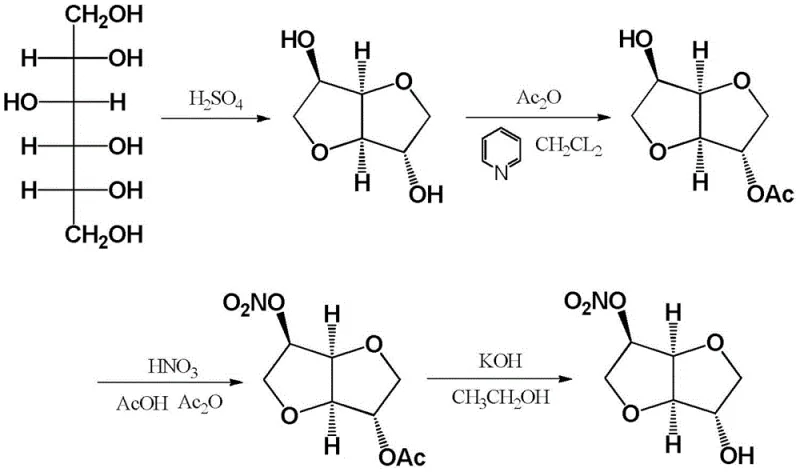
The Novel Approach
In stark contrast to these legacy methods, the technology protected under CN110407846B introduces a streamlined, one-pot strategy that fundamentally alters the reaction mechanism. Instead of relying on the aggressive nature of fuming nitric acid, this novel approach utilizes a milder nitrating system composed of TMSCl and a nitrate salt, catalyzed by aluminum trichloride. This shift allows for precise control over the reaction environment, operating effectively at low temperatures ranging from -20°C to 10°C. The result is a dramatic improvement in selectivity, where the 5-hydroxyl group is preferentially targeted, minimizing the formation of the unwanted 2-isomer and dinitrate byproducts. This precision eliminates the need for complex purification steps, allowing manufacturers to obtain high-purity products simply through extraction and concentration, thereby significantly enhancing the economic feasibility of large-scale production.
Mechanistic Insights into TMSCl-Mediated Selective Nitration
The core innovation of this process lies in the synergistic interaction between the silylating agent and the Lewis acid catalyst. Initially, isosorbide reacts with TMSCl in the presence of an organic base, such as triethylamine, to form a silylated intermediate. This step is crucial as it temporarily modifies the steric and electronic environment of the hydroxyl groups. When aluminum trichloride is subsequently introduced along with the nitrate source, it acts as a potent Lewis acid, activating the nitrate species for electrophilic attack. The specific geometry of the silylated intermediate favors nucleophilic substitution at the 5-position due to reduced steric hindrance compared to the 2-position. This mechanistic nuance ensures that the nitration occurs with high regioselectivity, a feat that is difficult to achieve with conventional acidic nitrating agents.
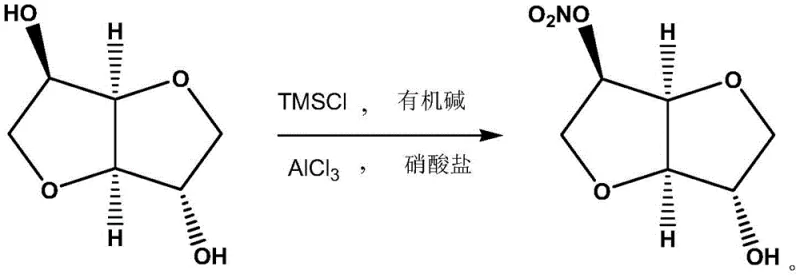
Furthermore, the reaction conditions are meticulously optimized to maintain stability throughout the transformation. The use of mild temperatures prevents the thermal degradation of sensitive intermediates and suppresses side reactions that typically lead to impurity profiles difficult to remove. Post-reaction, the workup is remarkably straightforward; quenching with ice water hydrolyzes any remaining silyl groups and dissolves inorganic salts, while the organic product can be easily extracted. The ability to recycle silicon-containing byproducts through phase separation adds another layer of efficiency, reducing raw material consumption. This robust mechanistic framework not only guarantees a purity exceeding 99% as confirmed by HPLC analysis but also ensures that the process remains consistent and reproducible across different batches, a critical requirement for GMP-compliant manufacturing.
How to Synthesize 5-Isosorbide Mononitrate Efficiently
Implementing this advanced synthesis route requires careful attention to reagent ratios and temperature control to maximize the benefits of the selective nitration mechanism. The process begins with the activation of isosorbide, followed by the controlled addition of the nitrating system. Detailed operational parameters, including specific mass ratios of TMSCl to isosorbide and the precise timing of reagent addition, are critical for achieving the reported high yields. For technical teams looking to adopt this methodology, the following guide outlines the standardized steps derived from the patent examples to ensure successful replication and scale-up.
- Protect and activate isosorbide by reacting with TMSCl and an organic base at low temperature to form a silylated intermediate.
- Introduce aluminum trichloride and a nitrate salt to the reaction mixture, maintaining strict temperature control to ensure selective nitration.
- Quench the reaction in ice water, adjust pH to neutral, extract with organic solvent, decolorize with activated carbon, and concentrate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The elimination of hazardous reagents and complex purification steps translates directly into a more resilient and cost-effective supply chain. By moving away from processes that require column chromatography or generate explosive byproducts, manufacturers can significantly reduce operational risks and insurance costs associated with handling dangerous chemicals. This shift enables a more stable production schedule, free from the delays often caused by safety incidents or the need for extensive waste treatment protocols.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the downstream processing. Traditional methods often incur heavy expenses due to the need for specialized equipment to handle corrosive acids and the high cost of solvents and stationary phases required for chromatographic purification. In this new method, the product can be isolated through simple filtration and extraction, removing the need for expensive silica gel columns and reducing solvent consumption substantially. Additionally, the high selectivity means that raw material utilization is maximized, as less isosorbide is wasted in forming unusable isomers, leading to a more efficient use of capital resources.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory and safety constraints of older nitration technologies. Because this process operates under mild conditions and avoids the accumulation of unstable dinitrates, it faces fewer regulatory hurdles and safety shutdowns. The use of common, commercially available reagents like TMSCl and aluminum trichloride ensures that raw material sourcing is not a bottleneck. This reliability allows for consistent delivery schedules, reducing the risk of stockouts for downstream pharmaceutical manufacturers who depend on a steady flow of high-quality intermediates for their final drug formulations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, particularly regarding waste disposal. This technology is inherently greener, as the post-treatment involves neutralization and extraction rather than generating large volumes of acidic waste or spent chromatography media. The ability to recycle silyl byproducts further minimizes the environmental footprint. For facilities aiming to meet stringent environmental standards, this process offers a clear pathway to compliance without sacrificing output volume, making it an ideal candidate for expansion from pilot scale to multi-ton commercial production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its potential impact on their operations. The following questions address common concerns regarding safety, purity, and scalability, drawing directly from the experimental data and technical disclosures found in the patent literature. These insights are intended to provide clarity for decision-makers assessing the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the TMSCl-mediated method improve selectivity compared to direct nitration?
A: Direct nitration often yields a complex mixture of 2-ISMN, 5-ISMN, and dinitrates requiring difficult separation. The TMSCl method utilizes steric and electronic effects to specifically target the 5-hydroxyl group, significantly reducing byproduct formation and eliminating the need for column chromatography.
Q: What are the safety advantages of this new nitration process?
A: Traditional methods using fuming nitric acid carry explosion risks due to unstable dinitrate byproducts. This novel route operates under mild conditions (-20°C to 10°C) and avoids the accumulation of hazardous energetic materials, enhancing overall process safety for industrial scale-up.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It features simple post-treatment steps like filtration and extraction rather than complex purification, uses recyclable silyl byproducts, and achieves high yields (>90%), making it economically viable for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Isosorbide Mononitrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis technologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensively analyzed the potential of the TMSCl-mediated nitration route and is well-positioned to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical, industrial setting. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 5-isosorbide mononitrate meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your cardiovascular drug projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of the most efficient and safe chemical processes available in the market.
