Advanced Meropenem Intermediate Synthesis: A Green Chemistry Breakthrough for Commercial Scale-Up
Advanced Meropenem Intermediate Synthesis: A Green Chemistry Breakthrough for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical antibiotics like meropenem. A recent technological advancement detailed in patent CN113929684B introduces a novel synthesis method for meropenem intermediates that fundamentally shifts the activation strategy from traditional phosphorus-based reagents to greener chloroformate compounds. This innovation addresses long-standing challenges in carbapenem manufacturing, specifically targeting the elimination of non-recyclable phosphorus waste and the reduction of complex purification steps. By replacing diphenyl chlorophosphate with readily available chloroformates such as ethyl chloroformate or isopropyl chloroformate, this process not only enhances the ecological profile of the synthesis but also significantly streamlines the operational workflow. For global supply chain leaders and R&D directors, this represents a pivotal opportunity to optimize production costs while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of meropenem intermediates has relied heavily on the use of diphenyl chlorophosphate as an activating agent for the azabicyclo parent nucleus. As illustrated in the conventional synthetic route, this method involves reacting the starting material with diphenyl chlorophosphate to form an enol phosphate intermediate, which is subsequently coupled with the side chain. 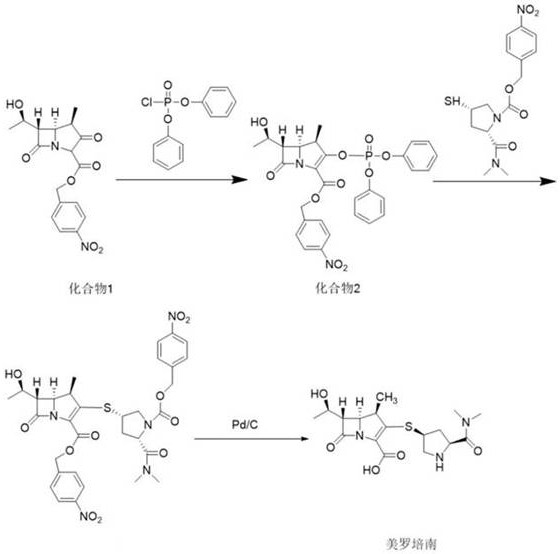 However, this traditional approach suffers from severe technical and economic drawbacks. The primary byproduct, diphenyl phosphate, is notoriously difficult to remove from the reaction mixture, necessitating multiple washing and purification steps that consume vast amounts of solvents and water. Furthermore, phosphorus-containing reagents are generally not recyclable in this context, leading to substantial solid and liquid waste generation that poses significant treatment costs and environmental hazards. The reaction times associated with this older methodology are also considerably longer, often requiring up to 5 hours for the activation step alone, which bottlenecks overall production throughput and limits scalability.
However, this traditional approach suffers from severe technical and economic drawbacks. The primary byproduct, diphenyl phosphate, is notoriously difficult to remove from the reaction mixture, necessitating multiple washing and purification steps that consume vast amounts of solvents and water. Furthermore, phosphorus-containing reagents are generally not recyclable in this context, leading to substantial solid and liquid waste generation that poses significant treatment costs and environmental hazards. The reaction times associated with this older methodology are also considerably longer, often requiring up to 5 hours for the activation step alone, which bottlenecks overall production throughput and limits scalability.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes chloroformate compounds to activate the azabicyclo compound, creating a much cleaner and more efficient reaction pathway. This method transforms the activation step into a rapid process that can be completed in as little as 0.5 hours under mild conditions, typically between -25°C and -20°C. Instead of generating persistent phosphorus waste, the byproducts of this reaction are simple alcohols like ethanol or isopropanol, which are volatile and easily removed during concentration without the need for complex aqueous workups. 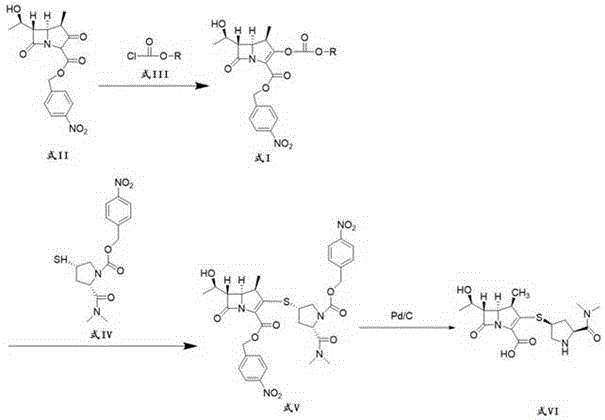 The subsequent coupling with the meropenem side chain is equally efficient, with reaction times reduced to approximately 1 hour. This drastic reduction in processing time, combined with the elimination of difficult-to-remove impurities, results in a process that is not only faster but also inherently safer and more cost-effective for large-scale manufacturing operations.
The subsequent coupling with the meropenem side chain is equally efficient, with reaction times reduced to approximately 1 hour. This drastic reduction in processing time, combined with the elimination of difficult-to-remove impurities, results in a process that is not only faster but also inherently safer and more cost-effective for large-scale manufacturing operations.
Mechanistic Insights into Chloroformate-Mediated Enol Activation
The core chemical innovation lies in the selective activation of the enol hydroxyl group on the bicyclic beta-lactam nucleus using chloroformates. In this mechanism, the chloroformate reacts with the azabicyclo compound in the presence of a tertiary amine base, such as triethylamine, to form a mixed carbonate ester intermediate (Formula I). This intermediate is highly reactive towards nucleophilic attack by the thiol group of the meropenem side chain, facilitating the formation of the critical C-S bond. The use of chloroformates ensures that the leaving group during the coupling step is a carbonate species that decomposes into harmless alcohol and carbon dioxide, thereby driving the reaction forward and preventing the accumulation of acidic byproducts that could degrade the sensitive beta-lactam ring. This mechanistic elegance ensures high stereochemical integrity, preserving the crucial (4R, 5S, 6S) configuration required for biological activity.
Furthermore, the choice of base and solvent plays a critical role in controlling the impurity profile. The patent data highlights that while bases like N,N-diisopropylethylamine can lead to oily residues and incomplete reactions, the use of triethylamine or tetramethylguanidine in solvents like dichloromethane or ethyl acetate promotes clean conversion. The reaction is conducted at low temperatures to minimize epimerization and beta-lactam ring opening, common degradation pathways for carbapenems. By carefully controlling the stoichiometry and addition rate, the process achieves high purity levels, often exceeding 98% by HPLC, directly after crystallization. This high level of control over the reaction mechanism translates directly into reduced downstream processing requirements and higher overall yields for the final API.
How to Synthesize Meropenem Intermediate Efficiently
The synthesis of the meropenem intermediate via this chloroformate route is designed for robustness and ease of execution in a GMP environment. The process begins with the activation of the azabicyclo compound at low temperatures, followed by a direct telescoped coupling with the side chain, minimizing the isolation of unstable intermediates. The detailed standardized synthesis steps, including specific reagent grades, addition rates, and crystallization parameters, are outlined in the guide below to ensure reproducibility and quality compliance.
- Activate the azabicyclo compound (Formula II) by reacting it with a chloroformate compound (Formula III) and an organic base in a solvent like dichloromethane at low temperatures (-25°C to -20°C) to form the meropenem intermediate (Formula I).
- Couple the resulting meropenem intermediate with the meropenem side chain (Formula IV) using a second organic base in acetonitrile, followed by washing, concentration, and crystallization to obtain undeprotected meropenem (Formula V).
- Perform a catalytic reduction reaction on the undeprotected meropenem using a catalyst such as palladium on carbon to remove protecting groups and yield the final meropenem product (Formula VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chloroformate-based synthesis offers transformative advantages that extend far beyond simple yield improvements. The shift away from phosphorus chemistry fundamentally alters the cost structure of manufacturing by eliminating the need for expensive waste treatment protocols associated with phosphate disposal. Additionally, the reduction in reaction time from several hours to less than one hour per step significantly increases plant capacity utilization, allowing for greater output without capital expenditure on new reactors. The simplicity of the workup procedure, which avoids multiple aqueous washes, also leads to substantial savings in solvent consumption and wastewater treatment costs, aligning perfectly with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of diphenyl chlorophosphate removes a significant cost center related to hazardous waste management. Since the byproducts are volatile alcohols, the energy-intensive drying and purification steps required to remove phosphorus salts are no longer necessary. This simplification of the downstream process reduces utility consumption and labor hours, leading to a leaner and more cost-efficient production model that enhances margin potential for high-volume antibiotic manufacturing.
- Enhanced Supply Chain Reliability: Chloroformate reagents such as ethyl chloroformate and isopropyl chloroformate are commodity chemicals with stable global supply chains, unlike specialized phosphorus reagents which can be subject to regulatory scrutiny and supply volatility. The robustness of the reaction conditions, which tolerate standard industrial solvents like ethyl acetate and acetonitrile, ensures that production can be maintained consistently across different geographic locations. This reliability minimizes the risk of production stoppages due to raw material shortages, ensuring a steady flow of critical intermediates to the final API production lines.
- Scalability and Environmental Compliance: The green chemistry nature of this process makes it highly scalable and compliant with increasingly strict environmental regulations. The absence of persistent organic pollutants and heavy phosphorus loads in the effluent simplifies the permitting process for new manufacturing facilities and reduces the liability associated with environmental compliance. The mild reaction conditions and short cycle times facilitate easy scale-up from pilot plants to multi-ton commercial production, allowing manufacturers to respond rapidly to market demand fluctuations for essential carbapenem antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel meropenem intermediate synthesis. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits and operational parameters of the technology.
Q: Why is the chloroformate method superior to the traditional diphenyl chlorophosphate route?
A: The traditional route generates diphenyl phosphate byproducts which are difficult to remove, require extensive washing, and create significant environmental waste. The new chloroformate method produces simple alcohol byproducts (ethanol or isopropanol) that are easily removed, drastically simplifying purification and reducing environmental impact.
Q: What represent the key quality metrics for this new intermediate synthesis?
A: According to the patent data, this method achieves high two-step molar yields ranging from 91% to 93% and HPLC purities exceeding 98%, demonstrating excellent reaction efficiency and product quality suitable for pharmaceutical applications.
Q: Which organic bases are recommended for optimal reaction performance?
A: The patent identifies triethylamine and tetramethylguanidine as highly effective bases. Comparative examples showed that using N,N-diisopropylethylamine resulted in poor reaction outcomes and oily residues, highlighting the importance of base selection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in patent CN113929684B to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity meropenem intermediates that meet stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical techniques to verify every batch.
We invite you to collaborate with us to leverage this green chemistry breakthrough for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and sustainability for your organization.
