Advanced Synthesis of Yellow-Green Fluorescent Helicenes for Next-Gen Optoelectronics
The landscape of organic optoelectronics is constantly evolving, driven by the demand for materials with precise emission profiles and robust stability. Patent CN102491936A introduces a significant advancement in this field by disclosing a novel class of conjugated compounds characterized by yellow-green fluorescence. These molecules are built upon a sophisticated skeleton comprising five aromatic rings rigidly fixed by two methylene groups, creating a large delocalized system with inherent helical chirality. This unique structural architecture not only enhances the conjugation effect but also leverages a push-pull electronic mechanism where electron-donating methoxy groups and electron-withdrawing imide moieties work in tandem to lower the energy gap for electron excitation. Consequently, these compounds exhibit extended excitation and emission wavelengths, making them highly suitable for next-generation display technologies and photochemical devices.
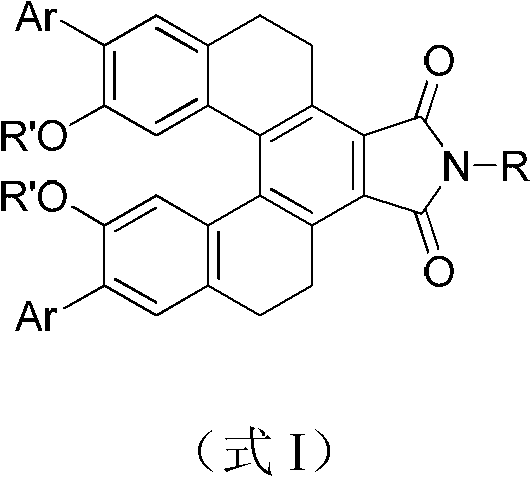
For procurement specialists and R&D teams seeking a reliable OLED material supplier, understanding the versatility of this chemical platform is crucial. The general formula allows for extensive derivatization at the aryl (Ar) and alkyl (R) positions, enabling fine-tuning of solubility and optical properties without compromising the core fluorescent efficiency. This adaptability ensures that manufacturers can tailor materials for specific device architectures, whether for small molecule OLEDs or as dopants in polymer matrices. The patent highlights that these compounds maintain their fluorescence even in solid-state thin films, a critical requirement for commercial device fabrication where solution-processability and film-forming capabilities are paramount for cost-effective manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance fluorescent materials has been plagued by complex multi-step routes that often rely on scarce precursors or harsh reaction conditions. Traditional red-emitting perylene diimides or blue-emitting pyrazoline derivatives frequently require rigorous purification protocols to remove trace metal impurities that can quench fluorescence or degrade device lifetime. Furthermore, many conventional fluorophores suffer from aggregation-caused quenching (ACQ) when transitioned from solution to solid state, limiting their utility in high-concentration applications. The lack of structural rigidity in some linear conjugated systems can also lead to non-radiative decay pathways, reducing overall quantum yield. For supply chain managers, these inefficiencies translate into higher production costs, longer lead times, and inconsistent batch-to-batch quality, posing significant risks for large-scale commercialization of optoelectronic components.
The Novel Approach
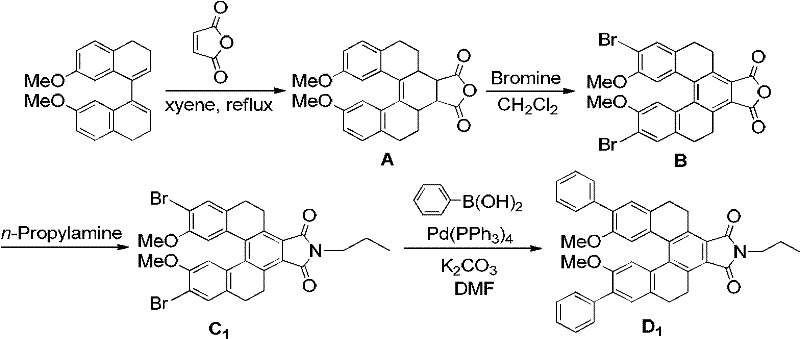
The methodology outlined in CN102491936A offers a transformative solution by utilizing a modular synthetic strategy centered on a Diels-Alder cycloaddition followed by palladium-catalyzed cross-coupling. This approach begins with readily available starting materials like 7,7'-dimethoxy-3,4,3',4'-tetrahydro-1,1'-binaphthalene and maleic anhydride, which are inexpensive and commercially accessible. The subsequent bromination and imidization steps construct the rigid helicene-like core with high regioselectivity, ensuring a consistent impurity profile. The final Suzuki-Miyaura coupling step allows for the late-stage introduction of diverse aryl groups, providing unparalleled flexibility in property tuning. This streamlined pathway significantly simplifies the manufacturing process, reducing the number of isolation steps and minimizing solvent waste, which aligns perfectly with modern green chemistry principles and cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Cyclization
The core of this synthesis lies in the precise construction of the conjugated backbone through a sequence of well-controlled organic transformations. The initial Diels-Alder reaction between the binaphthyl derivative and maleic anhydride proceeds via a concerted [4+2] cycloaddition mechanism, forming the central six-membered ring that locks the two naphthalene units into a fixed spatial arrangement. This rigidity is essential for preventing rotational freedom that could otherwise dissipate excited state energy as heat. Following this, the electrophilic aromatic substitution with bromine selectively functionalizes the positions ortho to the methoxy groups, activated by their electron-donating nature. This regioselectivity is critical for ensuring that the subsequent cross-coupling occurs at the correct sites to extend the conjugation length effectively. The use of Pd(PPh3)4 in the final step facilitates the transmetallation and reductive elimination cycles typical of Suzuki coupling, efficiently linking the electron-rich boronic acid derivatives to the electron-deficient brominated core.
From an impurity control perspective, the choice of reagents and conditions plays a pivotal role in achieving high-purity OLED material. The use of anhydrous conditions during the imidization step prevents hydrolysis of the anhydride intermediate, while the argon protection during the palladium-catalyzed step minimizes oxidative degradation of the catalyst and the sensitive organoboron species. The patent specifies the use of potassium carbonate as a mild base, which effectively activates the boronic acid without promoting side reactions such as dehalogenation or homocoupling. Furthermore, the ability to purify intermediates via recrystallization or column chromatography at each stage ensures that the final product meets stringent purity specifications required for electronic applications. This meticulous attention to reaction parameters guarantees that the resulting conjugated system possesses the intended electronic properties without contamination from catalytic residues or unreacted starting materials.
How to Synthesize Conjugated Fluorescent Compound Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these high-value fluorescent compounds with reproducible results. The process is divided into four distinct stages, each optimized for yield and purity, starting from the formation of the anhydride adduct to the final coupling reaction. Operators should pay close attention to temperature controls, particularly during the reflux steps in xylene and the subsequent heating in DMF, as these parameters directly influence reaction kinetics and product distribution. The detailed experimental procedures emphasize the importance of stoichiometric precision, especially regarding the molar ratios of the palladium catalyst and boronic acid, to maximize conversion rates. For those looking to implement this technology, the following guide outlines the standardized operational steps derived directly from the patent examples.
- Perform a Diels-Alder reaction between 7,7'-dimethoxy-3,4,3',4'-tetrahydro-1,1'-binaphthalene and maleic anhydride in xylene under reflux to form the anhydride addition product.
- Conduct oxidative addition by reacting the anhydride product with liquid bromine in dichloromethane at room temperature to obtain the dibromo intermediate.
- React the dibromo intermediate with n-propylamine or n-dodecylamine in DMF under argon protection to form the lactim intermediate.
- Execute a Suzuki-Miyaura coupling using Pd(PPh3)4 catalyst, potassium carbonate base, and aromatic boronic acid to yield the final conjugated fluorescent compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic route offers substantial strategic benefits for organizations focused on cost reduction in electronic chemical manufacturing and supply chain resilience. The reliance on commodity chemicals such as maleic anhydride and simple alkyl amines drastically lowers the raw material cost basis compared to routes requiring exotic heterocycles or precious metal complexes. Moreover, the modularity of the Suzuki coupling step means that a single batch of the brominated intermediate can be diversified into multiple final products simply by changing the boronic acid reagent. This flexibility allows manufacturers to respond rapidly to market demands for different emission colors or solubility profiles without retooling the entire production line, thereby enhancing supply chain reliability and reducing inventory risks associated with specialized intermediates.
- Cost Reduction in Manufacturing: The elimination of complex protecting group strategies and the use of standard reflux conditions significantly lower energy consumption and operational overhead. By avoiding cryogenic temperatures or ultra-high vacuum requirements, the process becomes compatible with existing industrial reactor infrastructure, facilitating immediate scale-up without capital-intensive equipment upgrades. The high yields reported in the patent examples indicate efficient atom economy, meaning less waste disposal cost and higher throughput per batch. Additionally, the catalyst loading is kept relatively low, reducing the expense associated with palladium recovery and purification, which contributes to a more favorable overall cost structure for high-volume production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are widely available from global chemical suppliers, mitigating the risk of supply disruptions common with niche reagents. The robustness of the reaction conditions, particularly the tolerance of the Suzuki coupling to various functional groups, ensures consistent quality even with slight variations in reagent grades. This reliability is crucial for maintaining continuous production schedules in the fast-paced electronics industry. Furthermore, the ability to store stable intermediates like the brominated anhydride allows for strategic stockpiling, buffering against potential logistical delays and ensuring a steady flow of finished goods to downstream device manufacturers.
- Scalability and Environmental Compliance: The process utilizes solvents like xylene, dichloromethane, and DMF, which are well-understood in industrial settings with established recovery and recycling protocols. The absence of highly toxic reagents or explosive intermediates simplifies safety management and regulatory compliance. As production scales from laboratory to commercial quantities, the exothermic profiles of the reactions remain manageable, allowing for safe operation in large-scale reactors. The solid-state fluorescence of the final products also implies that they can be processed into films using standard coating techniques, reducing the need for specialized deposition equipment and further lowering the barrier to entry for mass manufacturing of optoelectronic devices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent compound technology. These insights are derived from the specific experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and handling requirements. Understanding these details is essential for R&D directors evaluating the material for device integration and procurement managers assessing its viability for long-term sourcing.
Q: What is the primary emission color of these conjugated compounds?
A: The compounds exhibit bright yellow-green fluorescence in solution, with excitation wavelengths around 420nm and emission wavelengths typically ranging from 474nm to 490nm depending on the substituents.
Q: What are the key applications for this technology?
A: These materials are specifically designed for optoelectronic applications, including organic light-emitting devices (OLEDs), organic photovoltaics (OPV), and as organic fluorescent dyes.
Q: How does the molecular structure enhance fluorescence?
A: The structure features a rigid conjugated system of five aromatic rings fixed by methylene groups, creating helical chirality. The push-pull electron effect between methoxy groups and the imide moiety facilitates ground state electron excitation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Conjugated Fluorescent Compound Supplier
As the demand for advanced optoelectronic materials continues to surge, partnering with an experienced CDMO is vital for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of conjugated fluorescent compound meets the exacting standards required for high-performance OLED and display applications. We understand the critical nature of impurity profiles in electronic materials and employ state-of-the-art analytical techniques to ensure product integrity.
We invite you to collaborate with our technical team to explore how this patented synthesis can be optimized for your specific production goals. By engaging with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic feasibility of scaling this route within your existing infrastructure. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Let us help you secure a competitive advantage in the electronic materials market with our reliable supply chain and deep technical expertise.
