Advanced Yellow-Green Fluorescent Conjugated Compounds for Commercial Optoelectronic Applications
Advanced Yellow-Green Fluorescent Conjugated Compounds for Commercial Optoelectronic Applications
The rapid evolution of the optoelectronic industry demands materials that combine high fluorescence efficiency with exceptional thermal and chemical stability. Patent CN102491936B introduces a novel class of conjugated compounds characterized by a unique helical structure fixed by methylene bridges, offering a robust solution for yellow-green emission applications. This technology addresses the critical need for stable emitters in organic electroluminescent devices (ELD) and solar cells (OPV), where traditional linear conjugated systems often suffer from efficiency roll-off. By leveraging a rigid five-aromatic-ring skeleton, this innovation ensures a large delocalized electron system that promotes superior fluorescence emission without the structural degradation common in flexible analogues. For procurement and R&D leaders, this represents a significant opportunity to enhance device longevity and performance consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of fluorescent organic materials often relies on linear conjugated systems or flexible helicenes that lack structural rigidity. These conventional molecules are prone to conformational changes in the excited state, leading to non-radiative decay and reduced quantum yield. Furthermore, many existing yellow-green emitters require complex multi-step syntheses involving expensive transition metal catalysts or harsh conditions that compromise scalability. The lack of steric hindrance in standard planar structures often results in aggregation-caused quenching (ACQ) in solid-state films, severely limiting their utility in commercial OLED panels. Supply chain managers frequently encounter difficulties in sourcing precursors for these complex architectures, leading to inconsistent batch quality and extended lead times for material qualification.
The Novel Approach
The methodology outlined in CN102491936B overcomes these barriers by employing a methylene-fixed conjugated system that enforces helical chirality. This structural constraint prevents the molecular flattening that typically quenches fluorescence, thereby maintaining high emission efficiency even in solid films. The synthesis utilizes readily available starting materials such as 7,7'-dimethoxy-3,4,3',4'-tetrahydro-1,1'-binaphthalene and maleic anhydride, significantly simplifying the supply chain compared to exotic heterocyclic precursors. The push-pull electron effect generated by the methoxy and imide groups facilitates easier excitation of ground-state electrons, resulting in longer excitation and emission wavelengths ideal for specific display spectra. This approach not only improves photophysical properties but also streamlines the manufacturing process through high-yield reactions that are amenable to large-scale production.
Mechanistic Insights into Helical Conjugated System Formation
The core of this technology lies in the precise construction of a five-aromatic-ring skeleton locked by two methylene groups, creating a rigid helical chirality that is essential for stable fluorescence. 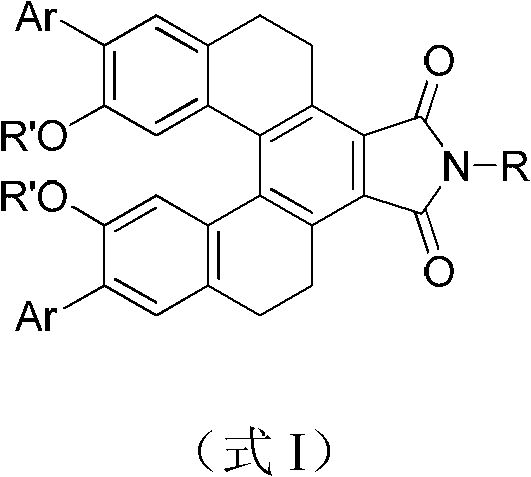 . The electronic architecture is designed to maximize the delocalized system, where the methoxy groups act as electron donors and the imide moiety serves as a strong electron acceptor. This intramolecular push-pull effect lowers the energy gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), facilitating efficient fluorescence emission in the yellow-green region. The rigidity imposed by the methylene bridges minimizes vibrational relaxation pathways, ensuring that the absorbed energy is released primarily as photons rather than heat, which is critical for high-efficiency optoelectronic devices.
. The electronic architecture is designed to maximize the delocalized system, where the methoxy groups act as electron donors and the imide moiety serves as a strong electron acceptor. This intramolecular push-pull effect lowers the energy gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), facilitating efficient fluorescence emission in the yellow-green region. The rigidity imposed by the methylene bridges minimizes vibrational relaxation pathways, ensuring that the absorbed energy is released primarily as photons rather than heat, which is critical for high-efficiency optoelectronic devices.
Impurity control is inherently managed through the high selectivity of the Suzuki coupling reaction used in the final functionalization step. The use of specific palladium catalysts and base conditions ensures that the aromatic boronic acids couple exclusively at the brominated positions without affecting the sensitive imide or methoxy functionalities. This chemoselectivity reduces the formation of side products that could act as quenching sites or charge traps in the final device. The crystallization and purification steps described in the patent, such as recrystallization from acetic anhydride, further ensure that the final product meets stringent purity specifications required for electronic grade materials. For R&D directors, this mechanistic robustness translates to reliable reproducibility and consistent batch-to-batch performance in downstream device fabrication.
How to Synthesize Helical Conjugated Fluorescent Compounds Efficiently
The synthesis pathway described in the patent offers a streamlined four-step process that balances chemical complexity with operational simplicity, making it highly suitable for industrial adoption. The initial Diels-Alder type addition followed by oxidative bromination sets up the core scaffold with high regioselectivity, minimizing the need for complex separation techniques. Subsequent imidation and palladium-catalyzed cross-coupling allow for diverse derivatization, enabling the tuning of electronic properties without altering the core stability. Detailed standardized synthesis steps see the guide below.
- Reflux 7,7'-dimethoxy-3,4,3',4'-tetrahydro-1,1'-binaphthalene with maleic anhydride in xylene at 140-150°C to form the anhydride addition product.
- Perform oxidative bromination of the anhydride intermediate using liquid bromine in acetic acid solution at room temperature to introduce leaving groups.
- Execute Suzuki coupling reaction between the brominated imide intermediate and aromatic boronic acids using Pd(PPh3)4 catalyst to finalize the conjugated system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages in cost reduction and supply chain reliability compared to traditional fluorescent material manufacturing. The reliance on commodity chemicals like xylene, maleic anhydride, and simple amines eliminates the dependency on scarce or highly regulated precursors that often bottleneck production. The high yields reported in the patent examples, particularly in the key coupling steps, indicate a process that minimizes raw material waste and maximizes output per batch. For procurement managers, this translates to a more predictable cost structure and reduced exposure to volatile raw material markets. The simplicity of the workup procedures, such as steam distillation and filtration, further reduces processing time and energy consumption, contributing to overall manufacturing efficiency.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps typically required in other catalytic processes significantly lowers downstream processing costs. By utilizing standard palladium catalysts in efficient coupling reactions, the process avoids the need for specialized ligands or extreme conditions that drive up operational expenses. The high atom economy of the reaction sequence ensures that a larger proportion of input materials are converted into the final product, reducing the cost per kilogram of the active pharmaceutical ingredient or electronic material. This economic efficiency allows for competitive pricing strategies without compromising on the quality or purity of the final optoelectronic compound.
- Enhanced Supply Chain Reliability: The use of widely available organic solvents and reagents ensures that production is not vulnerable to single-source supplier risks. The robustness of the chemical steps means that manufacturing can be easily transferred between facilities without significant re-optimization, providing flexibility in case of regional disruptions. The stability of the intermediates allows for potential stockpiling of key precursors, smoothing out production schedules and ensuring consistent delivery to downstream device manufacturers. This reliability is crucial for maintaining the continuous operation of high-volume display or lighting production lines that depend on steady material flows.
- Scalability and Environmental Compliance: The reaction conditions, primarily involving reflux in common solvents, are easily scalable from laboratory to multi-ton production using standard stainless steel reactors. The waste streams generated are primarily organic solvents which can be recovered and recycled, aligning with modern environmental compliance standards and reducing disposal costs. The absence of highly toxic heavy metals in the final product simplifies regulatory approval for use in consumer electronics. This scalability ensures that the technology can meet growing market demand for high-performance fluorescent materials without requiring capital-intensive new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent conjugated compound technology in industrial settings. These insights are derived directly from the patent data to provide accurate guidance for potential partners and licensees. Understanding these aspects is vital for integrating this material into existing supply chains and product development roadmaps.
Q: What is the primary stability advantage of this helical conjugated structure?
A: The structure is fixed by two methylene groups which create a rigid helical chirality, preventing conformational relaxation that typically quenches fluorescence in flexible conjugated systems.
Q: Can the emission wavelength be tuned for specific display applications?
A: Yes, the push-pull electron effect allows for derivatization at the aromatic positions, enabling tuning of excitation and emission wavelengths while maintaining the core yellow-green fluorescence.
Q: Is the synthesis route scalable for industrial production?
A: The process utilizes cheap raw materials and standard organic solvents like xylene and DMF, with reported yields exceeding 70% in key steps, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Conjugated Compound Supplier
NINGBO INNO PHARMCHEM stands ready to support your transition to this advanced fluorescent technology with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to optimize the methylene-fixed synthesis route for your specific volume requirements, ensuring stringent purity specifications are met through our rigorous QC labs. We understand the critical nature of optoelectronic materials and are committed to delivering batch consistency that meets the exacting standards of the display and lighting industries. Our facility is equipped to handle the specific solvent recovery and purification needs of this conjugated system, guaranteeing a supply that supports your long-term product lifecycle.
We invite you to initiate a dialogue with our technical procurement team to discuss how this technology can enhance your product portfolio. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your operation. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your application needs. Let us partner with you to bring this high-performance yellow-green fluorescent material from patent to production efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
