Innovative Catalytic Process for High-Purity Roxadustat Intermediate Commercial Scale-Up
Patent CN106478504B presents a groundbreaking method for synthesizing the key Roxadustat intermediate, specifically 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline carboxylate (IV), which is critical for producing the hypoxia-inducible factor prolyl hydroxylase inhibitor used in treating chronic anemia. This innovative approach addresses significant limitations in existing manufacturing processes by utilizing a streamlined cyclization reaction that enhances both yield and purity while reducing operational complexity. The patent details a robust synthetic pathway starting from readily available precursors, enabling efficient scale-up from laboratory to commercial production volumes. By eliminating multi-step protection/deprotection sequences and hazardous reagents, this method establishes a new benchmark for sustainable pharmaceutical intermediate manufacturing. The strategic design ensures compatibility with current Good Manufacturing Practices (cGMP) and supports rapid adoption by global pharmaceutical manufacturers seeking reliable supply chains for Roxadustat production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Roxadustat intermediates suffer from multiple critical drawbacks that hinder commercial viability. Methods developed by FibroGen and other entities require numerous experimental steps to introduce the methyl group on the isoquinoline ring, often necessitating noble metal catalysis under ultralow temperatures which significantly increases production costs and complicates large-scale implementation. For instance, prior art processes involve complex protection/deprotection sequences for methyl group introduction, requiring specialized hydrogenation equipment and generating substantial waste streams that challenge environmental compliance standards. Additionally, certain routes employ hydrogen peroxide oxidation for hydroxyl group installation, creating serious safety hazards during industrial production due to potential exothermic reactions and instability of peroxide compounds. These limitations collectively result in low overall yields, high impurity profiles requiring extensive purification, and extended lead times that disrupt supply chain continuity for pharmaceutical manufacturers dependent on timely intermediate availability.
The Novel Approach
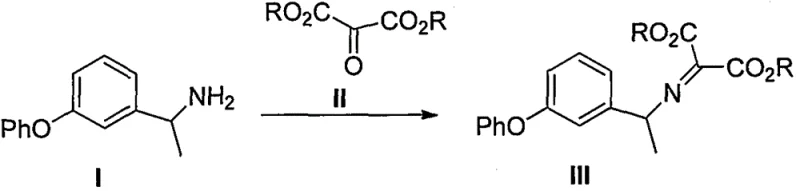
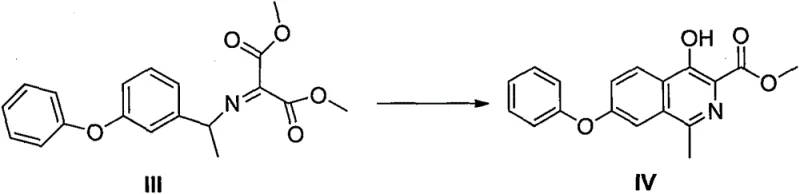
The patented method described in CN106478504B overcomes these challenges through an elegant single-step cyclization process that directly converts compound III into the target intermediate IV without requiring hazardous reagents or complex catalytic systems. By utilizing simple Lewis acid catalysts such as anhydrous aluminum trichloride or ferric trichloride in common solvents like ethylene glycol or xylene, this approach achieves high reaction yields exceeding 99% with minimal byproduct formation. The process eliminates multi-step sequences entirely by leveraging the inherent reactivity of the imino malonate precursor, enabling direct ring closure under controlled thermal conditions that avoid extreme temperatures or pressures. This streamlined methodology significantly reduces both capital expenditure through simpler equipment requirements and operational costs by minimizing raw material consumption and waste generation. Furthermore, the absence of transition metals or oxidizing agents ensures superior product purity without additional purification steps, making it ideal for pharmaceutical applications where stringent quality standards must be met.
Mechanistic Insights into Catalytic Cyclization Process
The core innovation lies in the thermal cyclization mechanism where compound III undergoes intramolecular condensation facilitated by Lewis acid catalysts to form the isoquinoline ring structure of intermediate IV. This reaction proceeds through a concerted mechanism where the imino group activates the malonate ester toward nucleophilic attack by the adjacent phenyl ring's ortho position, forming a new carbon-carbon bond that establishes the heterocyclic framework. The Lewis acid catalyst coordinates with carbonyl oxygen atoms, enhancing electrophilicity and lowering the activation energy barrier for ring closure while maintaining regioselectivity that prevents unwanted isomer formation. This catalytic system operates effectively across various solvent media including ethylene glycol and chlorobenzene, demonstrating remarkable versatility without requiring specialized reaction conditions or inert atmospheres typically needed in traditional methods.
Impurity control is achieved through precise reaction engineering where controlled temperature profiles prevent decomposition pathways that could generate side products. The absence of transition metals eliminates metal residue concerns that often complicate purification in conventional routes, while the single-step nature avoids accumulation of intermediates that might degrade under prolonged reaction conditions. Solvent selection plays a critical role in directing reaction selectivity; polar solvents like ethylene glycol facilitate proton transfer steps essential for aromatization, whereas non-polar solvents such as xylene promote cleaner product isolation through reduced solubility of impurities. This mechanistic understanding enables consistent production of intermediate IV with HPLC purity exceeding 99.5%, meeting pharmaceutical industry standards without requiring chromatographic purification that would increase manufacturing costs and reduce throughput.
How to Synthesize Roxadustat Intermediate Efficiently
This patented methodology provides a robust framework for producing high-purity Roxadustat intermediate at commercial scale through a simplified synthetic route that eliminates multiple processing steps required in conventional approaches. The process leverages readily available starting materials and standard manufacturing equipment to achieve exceptional yields while maintaining stringent quality specifications essential for pharmaceutical applications. By focusing on thermal cyclization rather than metal-catalyzed transformations, this approach significantly reduces both capital investment requirements and operational complexity across production facilities. Detailed standardized synthesis procedures are provided below to ensure consistent implementation across different manufacturing environments while maintaining optimal product quality and process efficiency.
- Combine compound III with a suitable solvent such as ethylene glycol in a reaction vessel under inert atmosphere.
- Heat the mixture to elevated temperatures (e.g., 190°C) with continuous stirring for several hours to facilitate cyclization.
- After reaction completion, cool the mixture, add water, filter the precipitate, and recrystallize from ethanol to obtain pure intermediate IV.
Commercial Advantages for Procurement and Supply Chain Teams
The innovative synthesis route described in patent CN106478504B directly addresses critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing organizations. By eliminating complex multi-step sequences and hazardous reagents, this method creates significant opportunities for cost optimization while enhancing supply chain resilience through simplified logistics and reduced dependency on specialized materials. The process design prioritizes operational flexibility that accommodates varying production volumes without requiring major equipment modifications, making it particularly valuable for companies managing fluctuating demand patterns in competitive therapeutic markets.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and hazardous reagents substantially reduces raw material expenses while avoiding costly purification steps required to remove metal residues from final products. Simplified reaction sequences decrease solvent consumption and waste generation, leading to significant savings in both material costs and environmental compliance expenditures without compromising product quality or yield.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials ensures consistent sourcing without dependency on specialized suppliers or restricted chemicals, reducing vulnerability to supply chain disruptions. The robust nature of the process maintains consistent performance across different production scales and locations, providing procurement teams with greater flexibility in vendor selection and inventory management while ensuring reliable delivery timelines.
- Scalability and Environmental Compliance: The straightforward thermal cyclization process demonstrates excellent scalability from laboratory to commercial production volumes without requiring specialized equipment modifications or safety protocols. Reduced waste generation through high atom economy and elimination of hazardous reagents significantly lowers environmental impact while simplifying regulatory compliance procedures across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis method for Roxadustat intermediate production. These responses are based on detailed analysis of the patent specifications and experimental data provided in the original disclosure, offering practical insights for manufacturing teams evaluating process adoption.
Q: How does this method overcome safety hazards of traditional hydrogen peroxide oxidation?
A: The patented process eliminates hydrogen peroxide by employing thermal cyclization instead of oxidation methods, removing explosion risks associated with peroxide compounds during industrial production.
Q: What cost benefits arise from avoiding noble metal catalysts?
A: Using simple Lewis acids like anhydrous aluminum trichloride eliminates expensive palladium catalysts and associated metal removal steps during purification, significantly reducing raw material expenses.
Q: How does the method ensure pharmaceutical-grade purity?
A: The single-step cyclization achieves >99.5% HPLC purity through straightforward recrystallization without chromatography, meeting stringent regulatory requirements without complex purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs and rigorous analytical protocols. Our technical expertise in catalytic cyclization processes ensures seamless transition from laboratory development to full-scale manufacturing with minimal process optimization requirements. As a trusted partner in pharmaceutical intermediate supply, we combine deep chemical knowledge with robust manufacturing capabilities to deliver consistent quality and reliable delivery schedules that support your critical drug development timelines.
Contact our technical procurement team today to request specific COA data and route feasibility assessments for your production needs. We offer a Customized Cost-Saving Analysis to demonstrate how our patented process can optimize your supply chain while ensuring uninterrupted access to high-purity Roxadustat intermediates essential for chronic anemia treatment development.
