Scalable Production of High-Purity Roxadustat Intermediates via Novel Cyclization
Introduction to Patent CN111499572A
The pharmaceutical landscape for treating renal anemia has been significantly transformed by Hypoxia-Inducible Factor Prolyl Hydroxylase (HIF-PH) inhibitors, with Roxadustat standing out as a pioneering oral therapy. However, the commercial viability of such potent drugs relies heavily on the efficiency of their supply chain, specifically the synthesis of critical intermediates. Patent CN111499572A discloses a groundbreaking preparation method for the Roxadustat intermediate, 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline formate. This technology represents a paradigm shift from legacy multi-step syntheses to a streamlined, high-yield process that begins with 3-oxo-3-(4-phenoxyphenyl) propionate. By optimizing the construction of the isoquinoline core through a novel cyclization strategy, this method addresses long-standing industry pain points regarding cost, safety, and environmental impact.
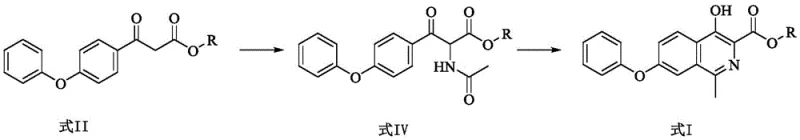
The core innovation lies in the strategic manipulation of the side chain prior to ring closure. Instead of building the heterocycle first and struggling with difficult functionalizations later, this route establishes the necessary nitrogen functionality early via a robust nitrosation-acetylation sequence. This approach not only simplifies the operational workflow but also drastically improves the overall atom economy. For procurement and R&D leaders, understanding this mechanistic departure is crucial, as it signals a move away from reliance on scarce precious metal catalysts towards abundant, commodity-grade reagents, ensuring a more resilient supply chain for this high-demand API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat intermediates has been plagued by inefficiencies that hinder large-scale production. The original research route, exemplified by world patent WO2004108681, involves a cumbersome eleven-step sequence starting from 3,4-dicyanonitrobenzene. This lengthy pathway necessitates multiple protection and deprotection steps, hazardous reagents like metallic sodium, and complex purification protocols that erode overall yield. Furthermore, optimized variations such as those in WO2013013609 attempted to shorten the route but introduced new bottlenecks, specifically relying on Palladium-catalyzed methylation. This step is notoriously problematic, often suffering from yields as low as 33%, which creates a massive material throughput deficit and introduces expensive heavy metal residues that require rigorous and costly removal processes to meet pharmaceutical standards.
The Novel Approach
In stark contrast, the methodology outlined in CN111499572A offers a concise and robust alternative that bypasses these historical hurdles. By selecting 3-oxo-3-(4-phenoxyphenyl) propionate as the starting material, the synthesis is reduced to essentially three critical transformations: nitrosation, acetylation/reduction, and cyclization. This novel approach eliminates the need for expensive Palladium catalysts entirely in the preferred embodiments, replacing them with cost-effective reagents like zinc powder or magnesium powder in acetic acid. The reaction conditions are notably mild, typically operating between 0°C and reflux temperatures in common solvents like toluene or DMF. This simplicity translates directly to operational excellence, allowing for easier temperature control, reduced energy consumption, and a significant reduction in the generation of hazardous waste streams associated with heavy metal catalysis.
Mechanistic Insights into Nitrosation and Cyclization
The chemical elegance of this process is rooted in the precise control of regioselectivity during the functionalization of the beta-keto ester backbone. The initial step involves the reaction of the starting ketone with sodium nitrite under acidic conditions (glacial acetic acid/water). This generates an alpha-oximino or hydroxylamino intermediate through an electrophilic substitution mechanism at the active methylene position. The subsequent transformation of this nitrogen-oxygen species into the required acetamido group is critical. In the preferred embodiment, this is achieved via a reductive acetylation where the N-O bond is cleaved and the nitrogen is simultaneously acylated. This one-pot or telescoped capability minimizes intermediate isolation losses and prevents the degradation of the sensitive beta-keto structure.
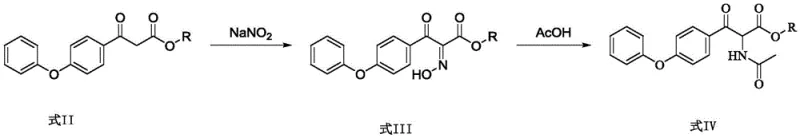
The final ring-closing step utilizes phosphorus oxychloride (POCl3) to effect a Bischler-Napieralski type cyclization. The acetamido carbonyl oxygen is activated by the phosphorus species, creating a highly electrophilic imidoyl chloride intermediate. This species undergoes an intramolecular electrophilic aromatic substitution with the pendant phenoxy phenyl ring, closing the isoquinoline system. The use of POCl3 is advantageous here as it serves both as a dehydrating agent and a chlorinating activator, driving the equilibrium towards the aromatic product. The resulting 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline formate is formed with high regiochemical fidelity, ensuring that the methyl group is correctly positioned at the 1-position without the formation of difficult-to-separate isomers that often plague alternative alkylation strategies.
How to Synthesize 4-Hydroxy-1-methyl-7-phenoxy-3-isoquinoline Formate Efficiently
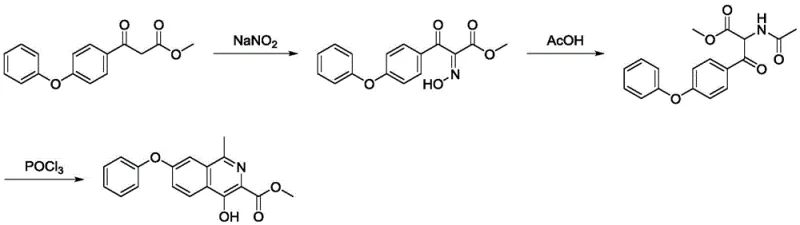
Implementing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the exothermic nitrosation phase. The process is designed to be flexible regarding the ester group (R), allowing for methyl, ethyl, benzyl, or even trifluoroethyl esters depending on downstream processing needs. The patent provides specific experimental data demonstrating that the intermediate 2-acetamido-3-oxo-3-(4-phenoxyphenyl) propionate can be isolated or carried forward directly, offering process engineers the option to telescope steps for maximum throughput. The following visual guide outlines the specific conditions used in Example 1, which achieved a remarkable 92.9% yield in the final cyclization step with 98.8% purity.
- Perform sodium nitrite hydroxylamination on 3-oxo-3-(4-phenoxyphenyl) propionate to generate the hydroxylamino intermediate.
- Execute reduction and acetylation reactions to convert the hydroxylamino group into an acetamido group, forming the key precursor.
- Conduct a cyclization reaction using phosphorus oxychloride to close the isoquinoline ring and obtain the final target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the global supply of pharmaceutical ingredients, the transition to this novel synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of precious metal catalysts is perhaps the most significant economic driver. Palladium prices are volatile and subject to geopolitical supply constraints; by removing this dependency, manufacturers can stabilize their raw material costs and insulate themselves from market shocks. Furthermore, the removal of heavy metals simplifies the regulatory compliance landscape, as there is no need for expensive scavenging resins or additional purification stages to meet strict ICH Q3D guidelines for elemental impurities. This streamlining of the downstream processing workflow directly correlates to reduced manufacturing cycle times and lower operational expenditures.
- Cost Reduction in Manufacturing: The replacement of low-yielding Palladium-catalyzed steps with high-yielding base-metal or reagent-driven transformations fundamentally alters the cost structure of the intermediate. The use of commodity chemicals like sodium nitrite, acetic anhydride, and phosphorus oxychloride ensures that raw material costs remain low and predictable. Additionally, the high yields reported in the patent examples (often exceeding 90% per step) mean that less starting material is wasted, maximizing the output per batch and significantly lowering the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The starting material, 3-oxo-3-(4-phenoxyphenyl) propionate, is structurally simple and can be sourced from multiple suppliers or synthesized via established etherification protocols. This reduces the risk of single-source bottlenecks that often occur with specialized, complex building blocks. The robustness of the reaction conditions—avoiding cryogenic temperatures or high-pressure hydrogenation—means that production can be executed in a wider range of manufacturing facilities, increasing the available capacity and ensuring continuity of supply even during regional disruptions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. It avoids the use of highly toxic reagents like carbon monoxide (used in some carbonylation routes) or explosive diazo compounds. The waste streams are primarily aqueous acids and organic solvents which are well-understood and manageable within standard wastewater treatment protocols. The ability to run the reaction at atmospheric pressure and moderate temperatures reduces the energy footprint of the plant, aligning with modern green chemistry initiatives and sustainability goals that are increasingly important for corporate procurement policies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for feasibility assessments.
Q: How does this new synthesis route compare to traditional Palladium-catalyzed methods?
A: Unlike traditional routes that rely on expensive Palladium catalysts for methylation with yields as low as 33%, this novel method utilizes cost-effective reagents like sodium nitrite and phosphorus oxychloride, achieving yields exceeding 90% without heavy metal contamination risks.
Q: What are the purity specifications achievable with this process?
A: The patented process demonstrates exceptional control over impurities, consistently delivering product purity above 98.8% as verified by HPLC analysis in multiple experimental examples, minimizing the need for complex downstream purification.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the route is designed for scalability, utilizing mild reaction conditions (0°C to reflux) and avoiding hazardous high-pressure hydrogenation steps found in other methods, making it ideal for commercial scale-up in standard reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
As the demand for Roxadustat continues to grow globally, securing a reliable supply of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in heterocyclic chemistry to bring complex patents like CN111499572A to life. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to optimize your supply chain for Roxadustat production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data, discuss route feasibility assessments, and discover how our advanced manufacturing capabilities can drive efficiency and reliability in your drug development pipeline.
