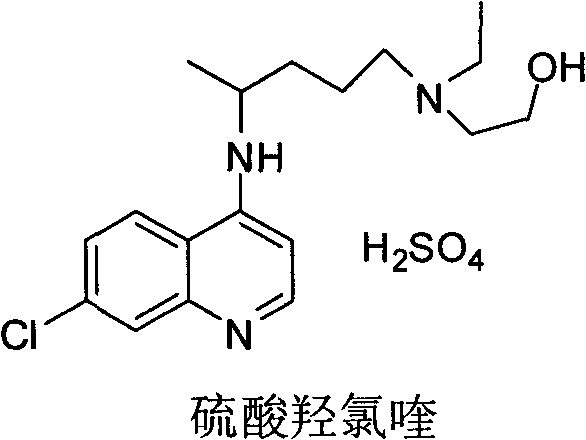
Advanced Industrial Preparation of Hydroxychloroquine Sulfate for Global Pharmaceutical Supply Chains
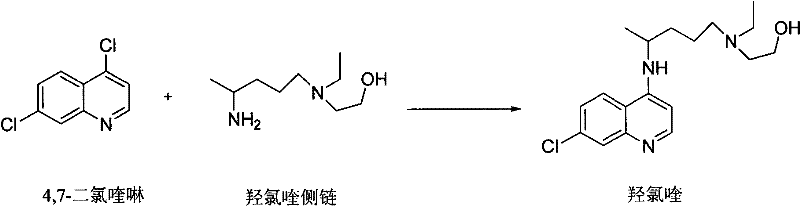
The global demand for high-quality antimalarial and autoimmune disease treatments has placed intense scrutiny on the supply chain integrity of key active pharmaceutical ingredients. Patent CN102050781B represents a significant technological breakthrough in the industrial preparation of hydroxychloroquine sulfate, addressing critical bottlenecks related to purity, yield, and environmental safety. This proprietary methodology shifts away from the hazardous and inefficient practices of the past, establishing a new benchmark for reliable pharmaceutical intermediates supplier standards. By optimizing the condensation reaction between 4,7-dichloroquinoline and the specific side chain, the process achieves a final product purity exceeding 99.5% with single impurities controlled below 0.1%. This level of quality is not merely a regulatory checkbox but a fundamental requirement for ensuring patient safety and therapeutic efficacy in sensitive populations. The innovation lies in the precise thermal management of the reaction trajectory, which fundamentally alters the impurity profile compared to traditional batch processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxychloroquine has been plagued by methodologies that introduce significant operational risks and environmental burdens. For instance, legacy processes described in patents such as CA2561987 often rely on extended reaction times of 20 to 24 hours at elevated temperatures, which paradoxically leads to higher impurity content necessitating complex downstream purification. These older routes frequently employ methyl isobutyl ketone (MIBK) and acetic anhydride in multi-step workups that are both costly and time-consuming. Furthermore, methods like those in US2546658 utilize phenol as a catalyst, introducing severe toxicity and corrosion issues that complicate equipment maintenance and wastewater treatment. The handling of phenolic wastewater is a major environmental liability, requiring specialized treatment facilities to meet discharge regulations. Additionally, high-pressure synthesis routes disclosed in documents like WO2010027150 necessitate the use of autoclaves operating at 5-20 bar, creating substantial safety hazards and limiting the scalability of production in standard chemical plants.
The Novel Approach
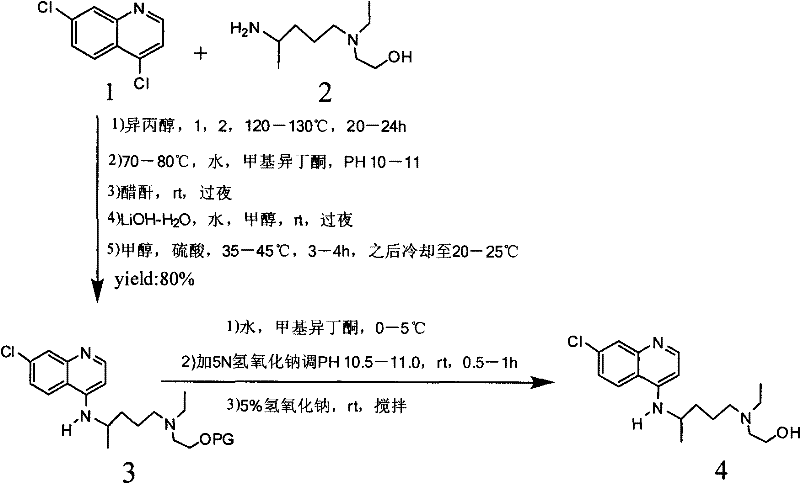
In stark contrast, the method disclosed in CN102050781B streamlines the entire production workflow into a highly efficient two-step sequence that eliminates the need for hazardous reagents and extreme conditions. The core innovation involves a solvent-steaming technique where the reaction mixture is gradually heated, allowing for the continuous removal of low-boiling components while driving the condensation forward. This approach effectively suppresses the formation of the problematic 7-chloro-4-(4-N-hydroxyethyl-1-methyl-tert amino) quinoline impurity without requiring expensive extraction solvents. By avoiding the use of chloroform, methylene chloride, and phenol, the process drastically reduces the ecological footprint and simplifies the regulatory compliance burden for manufacturing sites. The elimination of high-pressure equipment further enhances the economic viability of the process, allowing for implementation in standard glass-lined or stainless steel reactors found in most fine chemical facilities. This transition from complex, hazardous chemistry to a streamlined, ambient-pressure process represents a paradigm shift in cost reduction in API manufacturing.
Mechanistic Insights into Thermal-Controlled Condensation
The heart of this technological advancement lies in the precise kinetic control of the nucleophilic substitution reaction between the quinoline ring and the amine side chain. Traditional methods often apply heat rapidly or maintain static high temperatures, which accelerates side reactions and degradation pathways. The patented process employs a graduated heating profile, taking 7 to 12 hours to ramp the temperature from the initial reflux point up to the optimal range of 120-125°C. This slow ramping period allows the reaction to proceed through lower-energy pathways initially, maximizing the conversion of the starting material before the system reaches the thermal threshold where degradation becomes significant. Once the target temperature of 120-125°C is achieved, the reaction is maintained for a specific window of 13 to 18 hours. This duration is critical; it is long enough to ensure complete consumption of the 4,7-dichloroquinoline but short enough to prevent the thermal decomposition of the product or the formation of polymeric byproducts. The simultaneous removal of solvent during this heating phase helps to shift the equilibrium towards the product, adhering to Le Chatelier's principle without the need for excessive molar excesses of the expensive side chain.
Impurity control is further enhanced by the choice of recrystallization solvents, which are selected based on their differential solubility profiles for the target molecule versus the specific byproducts generated during the condensation. The use of ethyl acetate and isopropanol mixtures creates a solvent environment where the desired hydroxychloroquine base crystallizes with high selectivity, leaving the trace impurities in the mother liquor. This physical purification step is far more robust than the chemical scavenging methods used in prior art, which often introduce new contaminants. The subsequent salt formation with sulfuric acid is conducted under mild conditions, ensuring that the crystal lattice of the final sulfate salt forms with high integrity. This results in a material with excellent flow properties and stability, which are crucial parameters for downstream tablet formulation. The mechanistic understanding of how thermal gradients influence the impurity spectrum allows process chemists to fine-tune the operation for maximum yield, ensuring that the high-purity hydroxychloroquine sulfate meets the stringent specifications required by global pharmacopeias.
How to Synthesize Hydroxychloroquine Sulfate Efficiently
The implementation of this synthesis route requires careful attention to the heating ramp and solvent management to replicate the high yields reported in the patent data. The process begins with the charging of 4,7-dichloroquinoline and the side chain in isopropanol, followed by a controlled temperature increase that serves as the primary driver for reaction efficiency. Detailed operational parameters regarding stirring speeds, specific addition rates, and cooling curves are essential for maintaining batch-to-batch consistency. The following guide outlines the standardized protocol derived from the patent examples, providing a clear roadmap for process engineers to follow during technology transfer. For the complete step-by-step laboratory and pilot plant instructions, please refer to the detailed synthesis guide below.
- Condense 4,7-dichloroquinoline with the hydroxychloroquine side chain in isopropanol, gradually heating to 120-125°C over 7-12 hours to minimize byproduct formation.
- Maintain the reaction temperature at 120-125°C for 13-18 hours to ensure complete conversion while preserving product integrity.
- Purify the crude hydroxychloroquine via recrystallization using ethyl acetate and isopropanol, then react with sulfuric acid to form the final sulfate salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing method offers profound advantages that directly impact the bottom line and supply security. By eliminating the need for specialized high-pressure reactors and corrosive phenol handling systems, capital expenditure for new production lines is significantly reduced, while existing facilities can be retrofitted with minimal downtime. The simplification of the workup procedure, removing multiple extraction and overnight stirring steps, translates directly into shorter cycle times and increased throughput capacity per reactor vessel. This efficiency gain allows suppliers to respond more agilely to market demand spikes, ensuring continuity of supply for critical medications. Furthermore, the reduction in hazardous waste generation lowers the operational costs associated with waste disposal and environmental monitoring, contributing to a more sustainable and cost-effective supply chain model.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by optimizing the stoichiometric ratio of the expensive side chain, reducing it from the excessive multiples used in older methods to a near-equimolar ratio of 1:1.2. This reduction in raw material consumption is a primary driver of lower variable costs. Additionally, the replacement of expensive and difficult-to-recycle solvents like MIBK and chloroform with commodity solvents like isopropanol and ethyl acetate further drives down the cost of goods sold. The simplified purification process also reduces labor hours and utility consumption, as there are fewer unit operations and less energy required for solvent recovery. These cumulative efficiencies result in a highly competitive pricing structure for the final API without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available, non-controlled solvents and reagents mitigates the risk of supply disruptions caused by regulatory restrictions on toxic chemicals. Since the process does not require phenol or high-pressure equipment, it can be manufactured in a wider range of qualified facilities, diversifying the supplier base and reducing single-point-of-failure risks. The robustness of the crystallization step ensures consistent product quality, minimizing the risk of batch failures and rejected shipments that can disrupt downstream formulation schedules. This stability is crucial for procurement managers who need to secure long-term contracts with guaranteed delivery performance for essential medicines.
- Scalability and Environmental Compliance: The absence of high-pressure steps and highly toxic reagents makes this process inherently safer and easier to scale from pilot batches to multi-ton commercial production. The environmental profile is significantly improved by avoiding the generation of phenolic wastewater and chlorinated organic waste, which are subject to increasingly strict global regulations. This alignment with green chemistry principles facilitates faster regulatory approvals and reduces the administrative burden of environmental reporting. The ability to scale up without encountering the heat transfer and mass transfer limitations typical of viscous, high-pressure reactions ensures that production volumes can be increased seamlessly to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy routes. Understanding these distinctions is vital for technical teams evaluating process changes and for commercial teams negotiating supply agreements. The focus remains on the tangible improvements in purity, safety, and operational simplicity that define this new standard.
Q: How does the new preparation method improve impurity control compared to conventional routes?
A: The novel method utilizes a controlled gradual heating profile (7-12 hours to reach 120-125°C) which significantly reduces the formation of the 7-chloro-4-(4-N-hydroxyethyl-1-methyl-tert amino) quinoline impurity, achieving single impurity levels ≤0.1% without complex extraction steps.
Q: What are the safety advantages of this industrial process over high-pressure methods?
A: Unlike prior art methods requiring autoclaves at 5-20 bar pressure, this process operates at atmospheric pressure with gentle reflux conditions, eliminating the need for specialized pressure-resistant equipment and reducing potential safety hazards associated with high-pressure operations.
Q: Does this synthesis route eliminate the use of toxic solvents like phenol or chloroform?
A: Yes, the process avoids the use of highly toxic and corrosive reagents such as phenol and chlorinated solvents like chloroform or methylene chloride in the final purification, relying instead on safer solvents like isopropanol and ethyl acetate, which simplifies waste treatment and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxychloroquine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior manufacturing processes is key to maintaining a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102050781B are fully realized in large-scale operations. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of hydroxychloroquine sulfate meets the highest international standards. Our commitment to quality assurance means that clients receive a product that is not only chemically pure but also physically consistent, facilitating smooth formulation and tablet compression processes.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can reduce your overall procurement costs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in commercial scale-up of complex quinoline derivatives can support your long-term strategic goals.
