Revolutionizing Hydroxychloroquine Sulfate Production via Palladium-Catalyzed Coupling and Safe Reduction
Introduction to Patent CN112920114B: A Paradigm Shift in Antimalarial API Synthesis
The pharmaceutical industry constantly seeks more efficient, safer, and cost-effective pathways for producing essential medicines, and the synthesis of hydroxychloroquine sulfate is no exception. Patent CN112920114B, published in April 2022, introduces a groundbreaking three-step synthetic methodology that fundamentally reimagines the construction of this critical antimalarial and autoimmune disease therapeutic. Unlike legacy processes that rely on hazardous solvents and extreme conditions, this novel approach leverages advanced transition metal catalysis to achieve high yields with exceptional operational simplicity. The core innovation lies in the strategic assembly of the quinoline core and the amino-alcohol side chain through a sequence of palladium-catalyzed coupling, ring-opening amidation, and selective reduction. This technical breakthrough not only addresses long-standing environmental and safety concerns but also offers a robust framework for reliable hydroxychloroquine sulfate supplier operations aiming to optimize their supply chains.
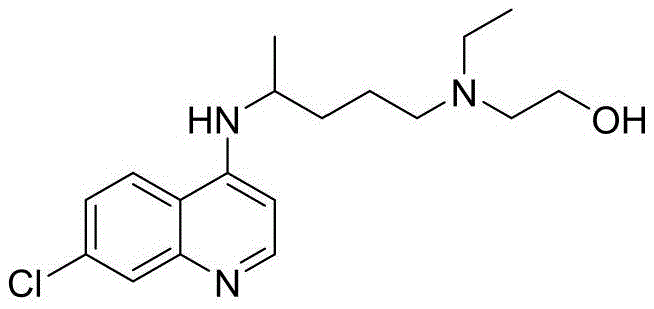
For R&D directors and process chemists, the significance of this patent cannot be overstated, as it provides a clear roadmap for overcoming the impurity profiles and safety hazards associated with traditional manufacturing. By utilizing cheap and easily obtainable raw materials such as 4,7-dichloroquinoline and 5-methyl-2-pyrrolidone, the invention drastically simplifies the material sourcing landscape. Furthermore, the elimination of toxic phenol solvents and high-pressure reactors aligns perfectly with modern green chemistry principles, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing. As we delve deeper into the technical specifics, it becomes evident that this route represents a substantial leap forward in process intensification and sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hydroxychloroquine has been plagued by significant technical and economic bottlenecks, primarily driven by the reliance on archaic reaction conditions. As illustrated in prior art patents such as US2546658, traditional methods often employ phenol as both a solvent and a reactant medium, requiring temperatures exceeding 125°C. Phenol is a solid at room temperature with a melting point of approximately 42°C, necessitating complex heating and feeding systems that complicate scale-up operations and increase energy consumption. Moreover, phenol is highly toxic and corrosive, posing severe health risks to operators and creating a massive burden on wastewater treatment facilities due to the difficulty in degrading phenolic waste streams.
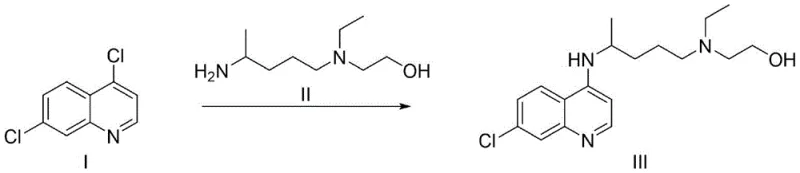
Furthermore, alternative routes disclosed in patents like WO2010027150 attempt to mitigate some issues but introduce new challenges, such as the requirement for high-pressure reactors operating at 5-20 bar. These pressurized conditions demand expensive specialized equipment and rigorous safety protocols, which inherently limit production capacity and increase capital expenditure. Additionally, many conventional routes rely on pre-synthesized side chains that are themselves expensive and difficult to purify, often lacking UV absorption which complicates process control. The cumulative effect of these factors is a manufacturing process that is not only environmentally unfriendly but also economically inefficient, with long reaction times ranging from 18 to 24 hours that severely impact throughput and overall productivity.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the process described in CN112920114B offers a streamlined, three-step solution that elegantly bypasses the need for toxic solvents and high-pressure equipment. The new route initiates with a palladium-catalyzed coupling reaction between 4,7-dichloroquinoline and 5-methyl-2-pyrrolidone, a transformation that proceeds efficiently at atmospheric pressure in common organic solvents like 1,4-dioxane or toluene. This initial step constructs the core carbon-nitrogen bond with high selectivity, avoiding the formation of complex byproduct mixtures typical of non-catalytic nucleophilic substitutions. Subsequent steps involve a controlled ring-opening of the pyrrolidone moiety followed by a mild reduction, ensuring that the sensitive functional groups remain intact while achieving the desired structural architecture.
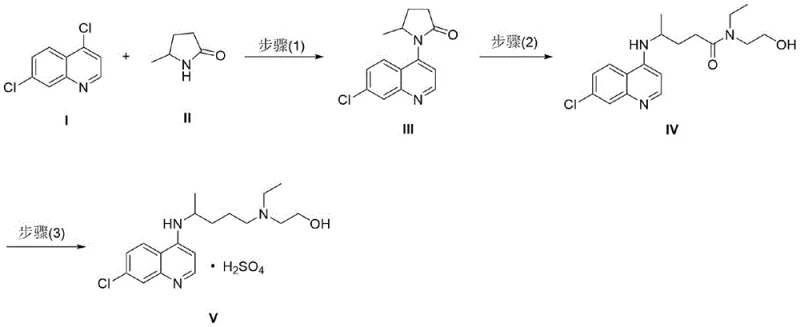
This innovative strategy effectively decouples the complexity of the synthesis from the harshness of the conditions, allowing for operation at moderate temperatures between 50°C and 150°C. By replacing the problematic phenol solvent with safer alternatives and eliminating the need for pressurized vessels, the novel approach significantly lowers the barrier to entry for commercial scale-up of complex pharmaceutical intermediates. The result is a process that is not only safer and more environmentally compliant but also inherently more flexible, enabling manufacturers to respond rapidly to market demands without the constraints imposed by specialized high-pressure infrastructure or hazardous waste disposal limitations.
Mechanistic Insights into Palladium-Catalyzed Coupling and Selective Reduction
The heart of this new synthetic methodology lies in the sophisticated application of transition metal catalysis, specifically the palladium-catalyzed cross-coupling in the first step. This reaction likely proceeds via a Buchwald-Hartwig type mechanism, where the palladium catalyst, coordinated with bulky phosphine ligands such as Xantphos or dppf, facilitates the oxidative addition of the aryl chloride bond in 4,7-dichloroquinoline. The choice of ligand is critical here, as it modulates the electronic and steric environment around the metal center, promoting the subsequent coordination and insertion of the nitrogen nucleophile from the 5-methyl-2-pyrrolidone. The presence of a strong base, such as cesium carbonate or triethylamine, serves to deprotonate the nucleophile and regenerate the active catalytic species, driving the cycle forward to completion with high turnover numbers.
Following the coupling, the second step involves the nucleophilic attack of N-ethyl-2-hydroxyethylamine on the lactam carbonyl of the intermediate, effectively opening the five-membered ring to form an amide linkage. This transformation can be accelerated by Lewis acids like aluminum trichloride, which activate the carbonyl group towards nucleophilic attack, ensuring high conversion rates even at lower temperatures. The final step is a chemoselective reduction of the amide bond to the corresponding amine, a notoriously difficult transformation that requires precise control to avoid over-reduction or side reactions. The patent highlights the use of borane-tetrahydrofuran complexes or sodium borohydride systems, which provide the necessary hydride source to reduce the amide carbonyl to a methylene group while preserving the integrity of the quinoline ring and the chloro substituent.
How to Synthesize Hydroxychloroquine Sulfate Efficiently
Implementing this novel synthetic route requires careful attention to reaction parameters and reagent quality to maximize yield and purity. The process is designed to be modular, allowing each step to be optimized independently before being telescoped into a continuous flow or batch process. For R&D teams looking to adopt this technology, the key lies in selecting the appropriate catalyst system and solvent combination that balances reaction rate with ease of workup. The detailed standardized synthesis steps below outline the specific conditions and stoichiometry required to replicate the high performance reported in the patent examples.
- Step 1: Perform a palladium-catalyzed coupling between 4,7-dichloroquinoline and 5-methyl-2-pyrrolidone using ligands like Xantphos or dppf in solvents such as 1,4-dioxane or toluene at 80-150°C.
- Step 2: React the resulting quinolinyl-pyrrolidone intermediate with N-ethyl-2-hydroxyethylamine, optionally using aluminum trichloride or thermal conditions, to open the lactam ring and form the amide linkage.
- Step 3: Reduce the amide intermediate using borane complexes or sodium borohydride systems, followed by salification with sulfuric acid to yield the final hydroxychloroquine sulfate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this new synthetic route offers profound advantages for procurement managers and supply chain heads who are tasked with minimizing costs and ensuring continuity of supply. The most immediate benefit is the drastic simplification of the raw material portfolio; by utilizing commodity chemicals like 4,7-dichloroquinoline and avoiding expensive, custom-synthesized side chains, companies can significantly reduce their exposure to volatile raw material markets. Furthermore, the elimination of phenol removes the need for specialized corrosion-resistant equipment and complex waste treatment protocols, leading to substantial cost savings in both capital expenditure and operational overhead. This shift towards safer, more benign chemistry translates directly into a more resilient and cost-efficient supply chain.
- Cost Reduction in Manufacturing: The new process eliminates the use of toxic phenol and high-pressure reactors, which are major cost drivers in traditional manufacturing. By operating at atmospheric pressure and using standard glass-lined or stainless steel reactors, facilities can avoid the high capital costs associated with specialized pressure vessels and the ongoing maintenance they require. Additionally, the shorter reaction times and higher yields mean that less energy is consumed per kilogram of product, and the reduced formation of impurities lowers the cost of downstream purification and waste disposal, resulting in a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Reliance on complex, multi-step side chain syntheses often creates bottlenecks in the supply chain, as any disruption in the production of these intermediates can halt the entire API manufacturing process. The novel route simplifies the supply chain by using readily available starting materials that are produced by multiple vendors globally, reducing the risk of single-source dependency. The robustness of the catalytic steps also ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification batches and ensuring a steady flow of product to meet market demand.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the ability to demonstrate a green and sustainable process is becoming a key competitive advantage. This new method aligns perfectly with environmental regulations by eliminating hazardous solvents and reducing the generation of toxic waste. The absence of high-pressure steps makes the process inherently safer and easier to scale from pilot plant to commercial production, allowing manufacturers to ramp up capacity quickly without the need for extensive safety reviews or infrastructure upgrades, thereby ensuring long-term compliance and operational flexibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel hydroxychloroquine sulfate synthesis. These answers are derived directly from the technical disclosures in patent CN112920114B and are intended to provide clarity on the feasibility and benefits of adopting this advanced manufacturing route. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or licensing.
Q: How does the new catalytic route improve safety compared to traditional phenol-based methods?
A: The novel process eliminates the use of molten phenol, a toxic and corrosive solid that requires complex heating systems and generates hazardous waste. By switching to safer solvents like 1,4-dioxane or toluene and operating at atmospheric pressure, the new method significantly reduces operator exposure risks and waste treatment costs.
Q: What are the key advantages of the palladium-catalyzed coupling step?
A: This step allows for the direct introduction of the side chain precursor onto the quinoline core under mild conditions (80-150°C) without the need for high-pressure equipment. The use of specific ligands ensures high selectivity and conversion, minimizing impurities and simplifying downstream purification.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids high-pressure reactors and toxic solvents, uses commercially available raw materials, and features short reaction times. The robust nature of the catalytic system and the straightforward workup procedures make it highly operable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxychloroquine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and a proven track record in process development. Our team of experienced chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical industrial setting. We are committed to delivering high-purity hydroxychloroquine sulfate that meets stringent purity specifications, utilizing our rigorous QC labs to monitor every step of the synthesis and guarantee product consistency.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative technology for their supply chains. By working together, we can conduct a Customized Cost-Saving Analysis tailored to your specific production needs, identifying opportunities to optimize costs and improve efficiency. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior synthetic method into your manufacturing portfolio.
