Advanced Stereoselective Synthesis of Gemcitabine for Commercial API Production
Advanced Stereoselective Synthesis of Gemcitabine for Commercial API Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology therapeutics, and the synthesis of Gemcitabine represents a pinnacle of complex nucleoside chemistry. Patent CN100475832C discloses a novel, highly stereoselective technical method for synthesizing industrial-grade Gemcitabine and its hydrochloride salt, addressing long-standing challenges in stereocontrol and process scalability. This innovation provides a streamlined route that bypasses the harsh conditions and excessive reagent usage typical of legacy methods, offering a viable solution for reliable pharmaceutical intermediates supplier networks aiming to secure high-purity supply chains. By leveraging specific protecting group strategies, this process achieves superior beta-isomer enrichment, ensuring that the final active pharmaceutical ingredient meets stringent quality specifications required for global regulatory compliance.
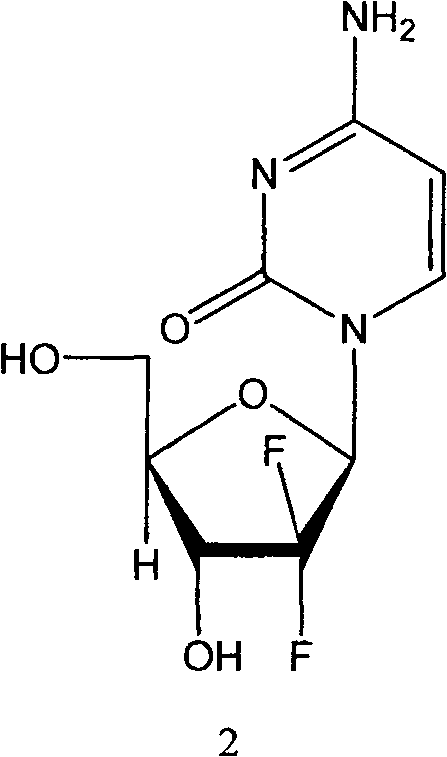
Gemcitabine, chemically known as 2'-deoxy-2',2'-difluorocytidine, functions as a potent antimetabolite that disrupts DNA replication by inhibiting ribonucleotide reductase. The structural complexity of this difluoronucleoside analogue necessitates precise stereochemical control during synthesis to ensure therapeutic efficacy. The disclosed technology not only optimizes the synthetic pathway but also introduces key intermediate compounds that facilitate easier purification and higher overall yields. For procurement and R&D teams, understanding the nuances of this patented route is essential for evaluating cost reduction in API manufacturing and ensuring the continuity of supply for this vital anticancer agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Gemcitabine have been plagued by significant operational deficiencies that hinder large-scale industrial application. Early methods, such as those disclosed by Hertel et al. in U.S. Pat 4808614, relied heavily on silica gel column chromatography to separate stereoisomers, a technique that is notoriously difficult to scale and results in substantial solvent waste and product loss. Furthermore, alternative routes described by Chou Ta-Sen required extremely low temperatures, often down to -78°C, to prepare key methanesulfonate intermediates, imposing severe energy costs and equipment constraints on manufacturing facilities. These legacy processes also frequently demanded a massive excess of cytosine, sometimes up to 20 molar equivalents, to drive stereoselectivity, which drastically inflates raw material costs and complicates downstream purification efforts.
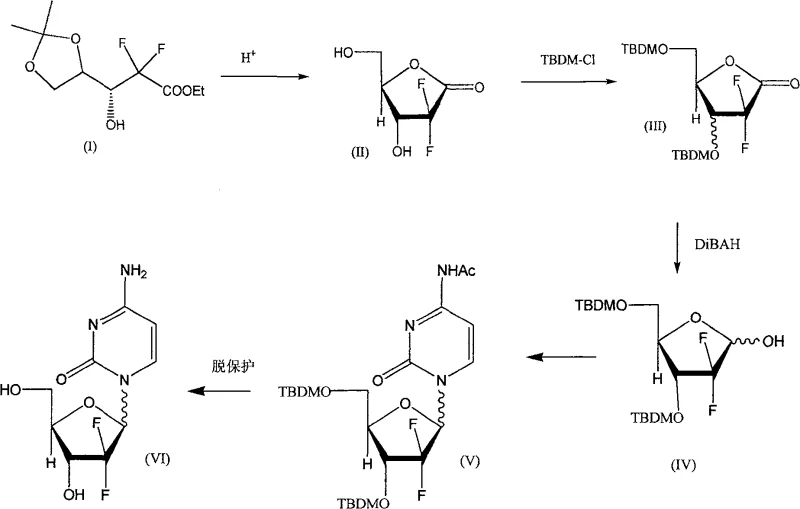
In addition to thermal and stoichiometric inefficiencies, prior art methods often utilized hazardous reagents and unstable intermediates that posed safety risks. For instance, certain international applications disclosed processes requiring quick cryoconcentration of reaction liquids to prevent the decomposition of potassium salt compounds, a step that is operationally fragile and prone to failure in a production environment. The reliance on excessive hydrogen halides for halogenation steps further exacerbated environmental concerns due to toxicity and difficult three-waste treatment. These cumulative factors rendered many existing technologies unsuitable for the commercial scale-up of complex pharmaceutical intermediates, creating a persistent demand for a gentler, more efficient synthetic methodology.
The Novel Approach
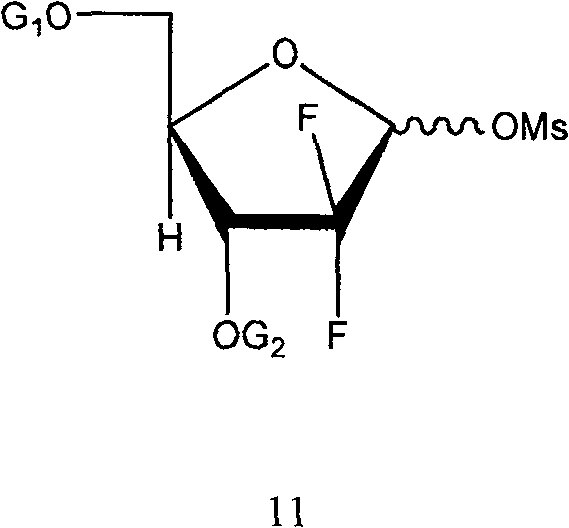
The novel process outlined in CN100475832C fundamentally reengineers the synthetic strategy by introducing specific aryl-based protecting groups that inherently favor the desired stereochemistry. Instead of relying on extreme cold or massive reagent excess, this method utilizes a controlled condensation reaction where an alpha-enriched sugar intermediate (Formula 11) reacts with protected cytosine (Formula 12) in high-boiling solvents like xylene or toluene. By maintaining the protected cytosine in a high-density state through a slow dripping procedure over several hours, the reaction achieves a remarkable beta-isomer ratio of up to 1:40 (alpha:beta) using only 2.5 molar equivalents of cytosine. This dramatic reduction in reagent usage, coupled with reaction temperatures ranging from 100°C to 150°C, signifies a major leap forward in process efficiency and safety.
Moreover, this innovative route eliminates the need for silica gel column chromatography entirely, replacing it with simple recrystallization and washing steps that are easily adaptable to multi-ton production scales. The total synthesis involves only five steps from the key intermediate to the final hydrochloride salt, achieving a total recovery rate of approximately 35.9% with product purity meeting USP and EP standards. The ability to produce high-purity Gemcitabine without the burden of complex chromatographic separations directly translates to reduced operational expenditures and a smaller environmental footprint. This approach effectively resolves the stereoselectivity issues of the past while providing a robust framework for the high-purity API intermediate manufacturing required by modern pharmaceutical standards.
Mechanistic Insights into Stereoselective Glycosylation
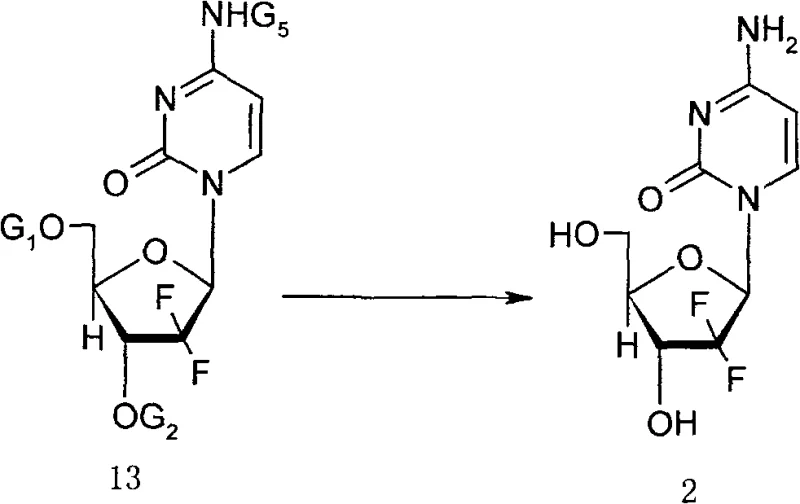
The core of this technological breakthrough lies in the meticulous design of the protecting groups attached to the sugar moiety, specifically the use of biphenyl carbonyl or benzoyl groups at the 3 and 5 positions. These bulky aryl groups exert a profound steric influence on the conformation of the furanose ring, stabilizing the transition state that leads to the desired beta-anomer during the glycosylation step. When the alpha-enriched methanesulfonate intermediate (Formula 11) is introduced to the silylated cytosine system, the electronic and steric properties of the protecting groups guide the nucleophilic attack of the cytosine nitrogen to the anomeric carbon with high fidelity. This mechanistic precision allows the reaction to proceed with high stereoselectivity even at elevated temperatures, overcoming the kinetic barriers that typically necessitate cryogenic conditions in other synthetic routes.
Furthermore, the deprotection strategy employed in the final stages ensures the integrity of the sensitive difluoro structure. The conversion of the protected nucleoside (Formula 13) to the free base (Formula 2) is achieved using anhydrous methanol ammonia solution under mild conditions, typically between 20°C and 50°C. This gentle deprotection prevents the degradation of the glycosidic bond, which is a common failure point in nucleoside synthesis. Following deprotection, the free base is converted to the hydrochloride salt by adjusting the pH to 1.5–2.5 using concentrated hydrochloric acid in organic solvents like isopropanol. This final crystallization step not only isolates the product but also serves as a final purification stage, ensuring that the resulting Gemcitabine HCl possesses the requisite chemical purity and enantiomeric excess for clinical use.
How to Synthesize Gemcitabine Efficiently
The synthesis of Gemcitabine via this novel route involves a sequence of carefully orchestrated reactions designed to maximize yield and stereocontrol while minimizing waste. The process begins with the preparation of the key alpha-enriched intermediate, followed by a high-temperature condensation with protected cytosine, and concludes with deprotection and salt formation. Each step has been optimized to avoid the pitfalls of prior art, such as the need for chromatography or hazardous reagents. For detailed operational parameters, including specific solvent volumes, temperature gradients, and stirring rates, operators should refer to the standardized protocols derived from the patent examples. The following guide outlines the critical phases of this streamlined manufacturing process.
- Preparation of key intermediate Formula 11 (alpha-enriched methanesulfonate) via reduction and sulfonation of protected furanose precursors.
- Condensation of Formula 11 with protected cytosine (Formula 12) in high-boiling solvents like xylene to yield beta-enriched Formula 13.
- Deprotection of Formula 13 using methanol ammonia followed by salt formation with hydrochloric acid to obtain Gemcitabine HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant simplification of the purification workflow, which directly impacts the cost of goods sold. By eliminating silica gel column chromatography, manufacturers can drastically reduce solvent consumption and waste disposal costs, two of the largest variable expenses in API production. Additionally, the reduction in cytosine usage from 20 molar equivalents in older methods to just 2.5 equivalents in this new process represents a substantial saving in raw material costs, enhancing the overall economic viability of the project without compromising on yield or quality.
- Cost Reduction in Manufacturing: The streamlined nature of this process allows for significant cost optimization through the removal of expensive and time-consuming purification steps. The avoidance of column chromatography means that production capacity is not bottlenecked by slow separation techniques, allowing for faster batch turnover. Furthermore, the use of common, high-boiling solvents like xylene and toluene, which are easily recoverable and recyclable, contributes to a lower environmental compliance burden and reduced operational expenditure. The qualitative improvement in process efficiency ensures that the cost reduction in pharmaceutical intermediates manufacturing is realized through both material savings and improved throughput.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which operate at moderate to high temperatures rather than cryogenic levels, makes the supply chain less vulnerable to equipment failures or utility fluctuations. The intermediates generated in this process, particularly the alpha-enriched methanesulfonates, are stable enough to be handled without the need for immediate cryoconcentration, reducing the risk of batch loss due to decomposition. This stability enhances the reliability of supply, ensuring that production schedules can be met consistently. For buyers, this translates to a more dependable source of high-purity pharmaceutical intermediates with reduced lead times and fewer disruptions.
- Scalability and Environmental Compliance: The design of this synthetic route is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production. The absence of toxic hydrogen halides and the minimization of hazardous waste streams align with increasingly strict global environmental regulations. The ability to achieve high purity through recrystallization rather than chromatography simplifies the engineering requirements for scale-up, making it easier to validate the process for GMP manufacturing. This scalability ensures that the technology can support the growing global demand for Gemcitabine while maintaining a sustainable and compliant production footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Gemcitabine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations or for procurement specialists assessing supplier capabilities.
Q: How does this novel process improve stereoselectivity compared to prior art?
A: The process utilizes specific aryl protecting groups (such as biphenyl carbonyl) on the sugar moiety which sterically direct the nucleophilic attack of cytosine. This allows for an alpha:beta ratio of 2.4:1 in the intermediate to convert into a highly beta-enriched product (1:40 alpha:beta) without needing excessive cytosine equivalents.
Q: What are the critical reaction conditions for the condensation step?
A: The condensation is performed in high-boiling organic solvents like xylene or toluene at temperatures between 100°C and 150°C. The protected cytosine is maintained in a high-density state by dripping the sugar intermediate solution over several hours, ensuring optimal stereochemical outcome.
Q: Does this method require column chromatography for purification?
A: No, a major advantage of this technology is the elimination of silica gel column chromatography. Purification is achieved through simple recrystallization and washing steps, making it highly suitable for large-scale industrial manufacturing and significantly reducing solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for life-saving oncology drugs like Gemcitabine. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in a practical manufacturing setting. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that verify every batch against international pharmacopeia standards. Our capability to implement complex stereoselective syntheses allows us to offer a reliable supply of high-quality intermediates and APIs to our global partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this novel route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
