Advanced Purification Strategies for High-Purity Gemcitabine Intermediates
The pharmaceutical industry's relentless pursuit of high-purity oncology therapeutics has placed significant scrutiny on the synthesis of key nucleoside analogues. Patent CN112979723B introduces a transformative purification methodology specifically designed for the production of Gemcitabine intermediates, addressing long-standing challenges in impurity management. This technical disclosure outlines a robust pathway to synthesize 2-deoxy-2,2-difluoro-D-erythropentofuranose-3,5-dibenzoate-1-methanesulfonate, a critical precursor in the Gemcitabine value chain. By integrating a novel Schiff base formation step, the process effectively isolates the target aldehyde from structurally similar diol byproducts that traditionally plague reduction reactions. For R&D directors and procurement specialists, this represents a pivotal shift towards more reliable pharmaceutical intermediate supplier capabilities, ensuring that downstream API synthesis proceeds with minimal risk of contamination or yield loss.
The Limitations of Conventional Methods vs. The Novel Approach
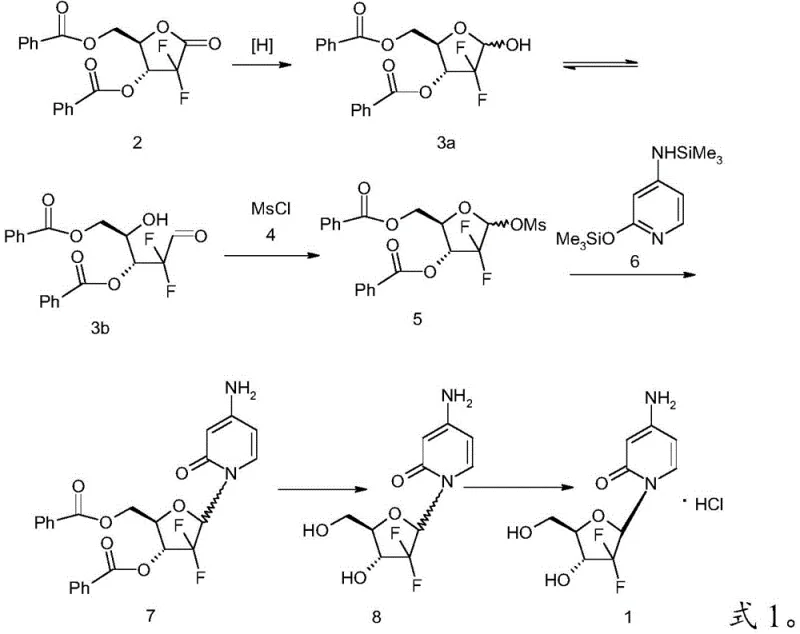
The Limitations of Conventional Methods
Traditional synthetic routes for generating the key furanose intermediate typically rely on the direct reduction of the lactone starting material followed immediately by mesylation. However, this conventional approach suffers from a fundamental chemical limitation: the lack of selectivity during the reduction phase often leads to the formation of over-reduced diol byproducts. As illustrated in the background art, when Compound 2 is reduced, it inevitably generates a mixture containing not only the desired hemiacetal or aldehyde forms but also significant quantities of the diol impurity, designated as Compound 9. This structural similarity makes chromatographic separation economically unviable on a large scale. Consequently, when this impure mixture undergoes sulfonylation, the diol reacts to form a bis-mesylated byproduct, Compound 10, which is notoriously difficult to remove and severely compromises the purity of the final intermediate. This accumulation of impurities directly impacts the cost reduction in pharmaceutical intermediates manufacturing by lowering overall yields and necessitating complex, wasteful purification steps.
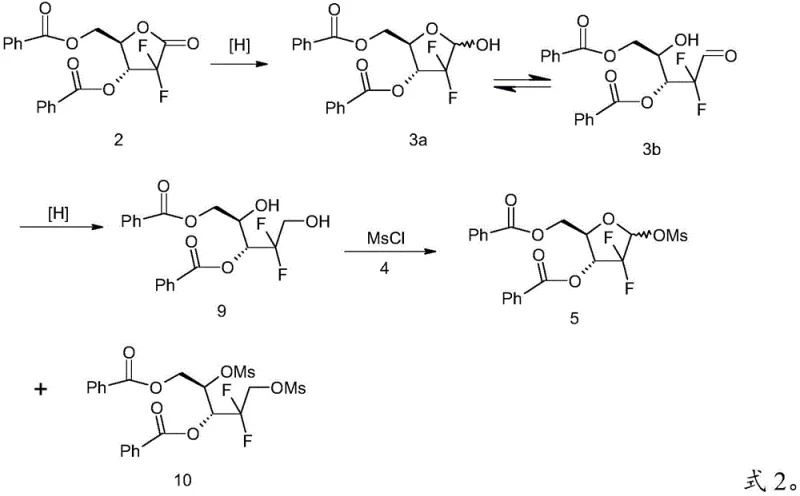
The Novel Approach
The innovative strategy disclosed in the patent circumvents these separation hurdles by introducing a chemoselective derivatization step prior to final functionalization. Instead of attempting to separate the aldehyde from the diol directly, the process converts the target aldehyde into a Schiff base through a dehydration condensation reaction with aniline. This transformation drastically alters the physical properties of the target molecule, enabling its separation from the unreacted diol impurity via simple filtration or crystallization. Once isolated, the pure Schiff base can be hydrolyzed back to the high-purity aldehyde, which is then subjected to mesylation. This decoupling of the reduction and mesylation steps ensures that the diol impurity never enters the final sulfonylation reactor, thereby preventing the formation of the problematic bis-mesylated side product. This approach significantly enhances the commercial scale-up of complex pharmaceutical intermediates by simplifying the workflow and improving the robustness of the purification profile.
Mechanistic Insights into Schiff Base-Mediated Purification
The core of this technological advancement lies in the precise exploitation of carbonyl reactivity differences between the target intermediate and its byproducts. The mechanism begins with the controlled reduction of the lactone ring using lithium tri-tert-butoxyaluminum hydride at cryogenic temperatures (-78°C). While this step minimizes over-reduction, it cannot entirely eliminate the formation of the diol Compound 9. The breakthrough occurs in the subsequent step where aniline acts as a specific trapping agent. The primary amine of aniline reacts selectively with the aldehyde functionality of Compound 3 (in its open-chain form) to form an imine linkage, creating the Schiff base Compound 12. This reaction is thermodynamically driven by the removal of water, typically facilitated by azeotropic distillation in solvents like toluene. Crucially, the diol Compound 9 lacks the reactive carbonyl group required for this condensation, rendering it inert under these specific conditions. This chemical orthogonality allows for a clean physical separation, where the solidified Schiff base can be filtered off, leaving the soluble diol impurities in the mother liquor.
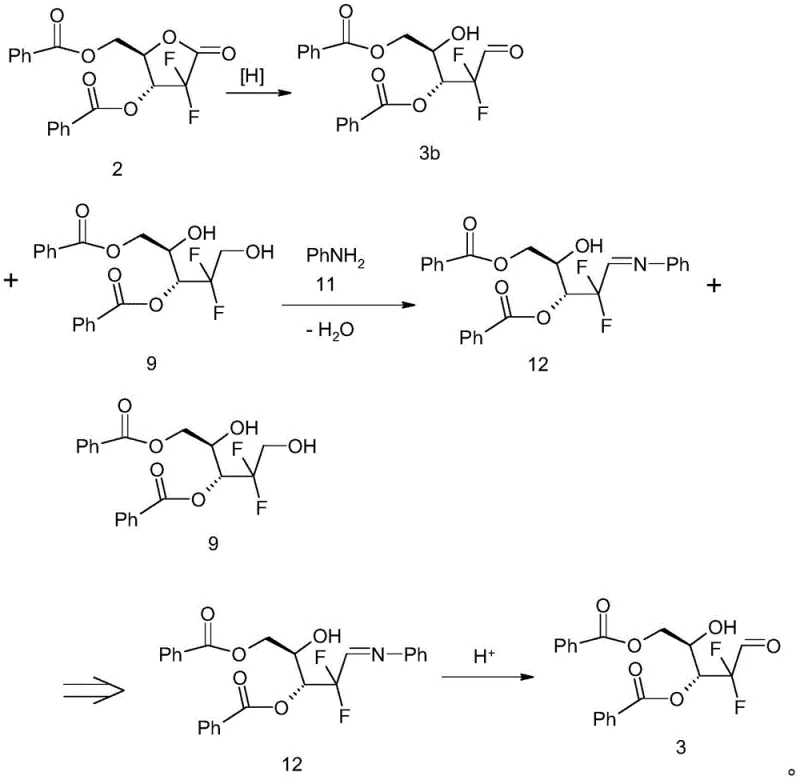
Following the isolation of the Schiff base, the process employs a mild acidic hydrolysis to regenerate the free aldehyde. This step is critical for restoring the reactive center needed for the final mesylation without reintroducing impurities. The use of concentrated hydrochloric acid in a biphasic system ensures complete hydrolysis while maintaining the integrity of the sensitive fluorinated sugar backbone. The resulting Compound 3 exhibits exceptional purity, often exceeding 99%, which serves as an ideal substrate for the subsequent reaction with methanesulfonyl chloride. By ensuring that only the mono-alcohol form of the sugar is present during mesylation, the process guarantees the exclusive formation of the mono-mesylate Compound 5. This mechanistic precision eliminates the need for extensive column chromatography, relying instead on scalable crystallization and filtration techniques that are preferred in GMP manufacturing environments for high-purity pharmaceutical intermediates.
How to Synthesize 2-Deoxy-2,2-difluoro-D-erythropentofuranose Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for replicating this high-efficiency purification method in a pilot or production setting. The procedure emphasizes strict temperature control during the initial reduction and precise stoichiometric balance during the Schiff base formation to maximize yield. Operators must ensure that the dehydration condensation is driven to completion, typically monitored by HPLC, to prevent residual aldehyde from carrying over. The following guide summarizes the critical operational parameters required to achieve the reported purity standards.
- Perform selective reduction of Compound 2 using lithium tri-tert-butoxyaluminum hydride at -78°C to generate a mixture of Compound 3 and byproduct Compound 9.
- React the crude mixture with aniline in toluene at 60-65°C to form a separable Schiff base (Compound 12), leaving the diol impurity in solution.
- Isolate the Schiff base via filtration, hydrolyze with acid to recover pure Compound 3, and proceed to mesylation to obtain Compound 5.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the purification train. By replacing complex chromatographic separations with crystallization and filtration, the process reduces solvent consumption and processing time, leading to substantial cost savings in raw material and utility usage. Furthermore, the elimination of the bis-mesylated byproduct ensures a cleaner reaction profile, which minimizes the burden on downstream API synthesis teams who would otherwise have to manage these persistent impurities. This reliability is crucial for maintaining continuous supply chains for critical oncology drugs, where batch failures can have severe market consequences.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through yield enhancement and waste reduction. By preventing the formation of inseparable byproducts early in the synthesis, the overall material throughput is improved. The ability to recycle solvents like toluene and dichloromethane, combined with the avoidance of expensive silica gel columns, significantly lowers the cost of goods sold (COGS). Additionally, the high purity of the intermediate reduces the need for reprocessing or re-crystallization cycles, streamlining the manufacturing timeline and reducing labor costs associated with extended batch times.
- Enhanced Supply Chain Reliability: Consistency in intermediate quality is the backbone of a reliable supply chain. This method delivers a product with consistently high purity, reducing the variability that often plagues multi-step organic syntheses. For supply chain planners, this means more predictable lead times and a lower risk of batch rejection during quality control testing. The robustness of the chemistry allows for scaling from kilogram to tonnage quantities without significant re-engineering of the process, ensuring that suppliers can meet fluctuating market demands for Gemcitabine APIs without interruption.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process favors safer unit operations. The replacement of hazardous chromatographic solvents with standard crystallization solvents aligns better with green chemistry principles and regulatory expectations for waste management. The reduced generation of hazardous waste streams simplifies disposal compliance and lowers environmental fees. Moreover, the scalability of filtration and crystallization equipment is well-established in the fine chemical industry, facilitating a smoother technology transfer from R&D labs to commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this purification strategy. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework.
Q: How does the Schiff base method improve purity compared to direct reduction?
A: Direct reduction often yields a difficult-to-separate mixture of the target aldehyde and over-reduced diol. The Schiff base method chemically modifies only the aldehyde, allowing physical separation of the diol impurity before the final mesylation step.
Q: What is the critical temperature control for the reduction step?
A: The reduction reaction utilizing lithium tri-tert-butoxyaluminum hydride must be strictly maintained at -78°C to minimize over-reduction to the diol byproduct.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes standard unit operations like filtration and crystallization, making it highly suitable for commercial scale-up without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of fluorinated nucleoside synthesis requires a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped to verify the absence of critical impurities like the bis-mesylated byproduct, guaranteeing that every batch meets the exacting standards required for oncology drug manufacturing.
We invite you to collaborate with us to leverage this advanced purification technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your production efficiency and secure your supply of high-quality Gemcitabine intermediates.
