Advanced Synthesis of 6-Chloropurine: A Strategic Route for Pharmaceutical Intermediates Manufacturing
Advanced Synthesis of 6-Chloropurine: A Strategic Route for Pharmaceutical Intermediates Manufacturing
The global demand for high-purity purine derivatives continues to surge, driven primarily by the expanding market for antiviral therapeutics and oncology treatments. At the forefront of this chemical landscape is 6-chloropurine, a critical building block for synthesizing life-saving medications such as Tenofovir and Adefovir. A pivotal advancement in the production of this key intermediate is detailed in patent CN102336755B, which outlines a robust and efficient chemical synthesis method. This technology represents a significant departure from traditional methodologies by utilizing acetyl hypoxanthine as the direct starting material, thereby streamlining the manufacturing workflow. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the environmental footprint of API production. The following analysis dissects the technical merits and commercial implications of this innovative process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 6-chloropurine has relied heavily on hypoxanthine as the primary feedstock. While chemically feasible, this conventional pathway introduces significant logistical and environmental bottlenecks that modern manufacturing seeks to eliminate. The traditional route necessitates a multi-step sequence where acetyl hypoxanthine must first undergo hydrolysis to generate hypoxanthine, followed by acidification and purification before it can even enter the chlorination reactor. This preliminary deacetylation step is not only time-consuming but also generates substantial volumes of acidic wastewater, posing severe challenges for waste treatment facilities. Furthermore, the isolation and drying of the intermediate hypoxanthine add unnecessary unit operations, increasing energy consumption and the risk of product loss through mechanical handling. These inefficiencies accumulate, resulting in higher operational expenditures and a larger carbon footprint, which are increasingly scrutinized by regulatory bodies and corporate sustainability mandates.
The Novel Approach
In stark contrast to the legacy processes, the methodology disclosed in the patent leverages acetyl hypoxanthine directly, effectively bypassing the cumbersome deacetylation stage entirely. By reacting acetyl hypoxanthine directly with phosphorus oxychloride in the presence of a tertiary amine catalyst, the synthesis achieves a remarkable simplification of the process flow. This "one-pot" strategy not only accelerates the overall production timeline but also drastically curtails the generation of liquid waste associated with the hydrolysis of the acetyl group. The structural integrity of the acetyl group is maintained until the chlorination event, ensuring that the reaction proceeds with high specificity towards the desired 6-chloro substitution. 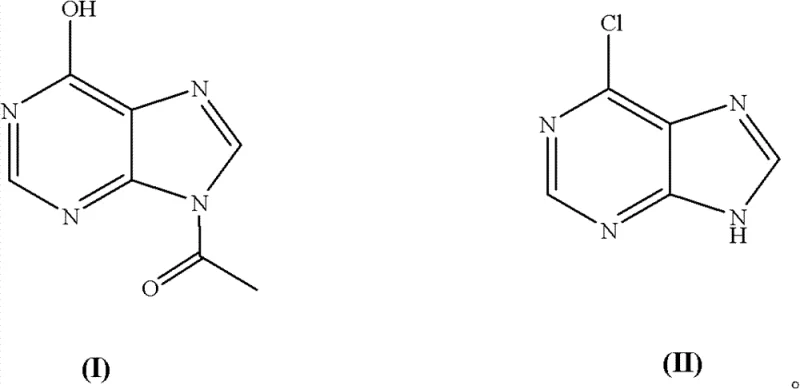 This direct conversion capability transforms the economic model of production, offering a reliable pharmaceutical intermediate supplier a distinct competitive edge through reduced utility costs and simplified effluent management protocols.
This direct conversion capability transforms the economic model of production, offering a reliable pharmaceutical intermediate supplier a distinct competitive edge through reduced utility costs and simplified effluent management protocols.
Mechanistic Insights into POCl3-Mediated Chlorination
The core of this synthetic breakthrough lies in the efficient activation of the hydroxyl group at the C6 position of the purine ring system. In the presence of phosphorus oxychloride (POCl3), the oxygen atom of the acetyl hypoxanthine acts as a nucleophile, attacking the phosphorus center to form a reactive phosphorylated intermediate. This activation renders the C6 position highly susceptible to nucleophilic attack by chloride ions. The role of the tertiary amine catalyst—whether it be DMA, triethylamine, or pyridine—is multifaceted; it serves not only to scavenge the hydrogen chloride byproduct generated during the reaction but also potentially stabilizes the transition state, facilitating the displacement of the phosphorylated leaving group. 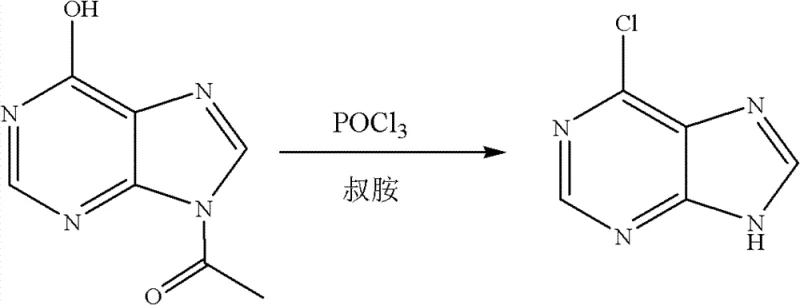 The reaction conditions specified, ranging from 70°C to 105°C, provide sufficient thermal energy to overcome the activation barrier without inducing significant degradation of the sensitive purine scaffold. This precise control over reaction kinetics ensures that the chlorination occurs selectively at the C6 position, minimizing the formation of poly-chlorinated impurities or decomposition products that often plague high-temperature halogenation reactions.
The reaction conditions specified, ranging from 70°C to 105°C, provide sufficient thermal energy to overcome the activation barrier without inducing significant degradation of the sensitive purine scaffold. This precise control over reaction kinetics ensures that the chlorination occurs selectively at the C6 position, minimizing the formation of poly-chlorinated impurities or decomposition products that often plague high-temperature halogenation reactions.
From an impurity control perspective, the choice of molar ratios is critical for maintaining high product quality. The patent specifies a molar ratio of acetyl hypoxanthine to chlorinating agent to tertiary amine of 1:(3.5-20.0):(1.0-1.5). Operating within these parameters ensures that there is a vast excess of the chlorinating agent, driving the equilibrium towards completion and suppressing reversible side reactions. The excess POCl3 also acts as a solvent, maintaining a homogeneous reaction phase which improves heat transfer and mixing efficiency. Upon completion, the removal of unreacted POCl3 via evaporation followed by quenching with ice water allows for the controlled precipitation of the product. Adjusting the pH to a neutral range of 7-9 is a crucial final step that ensures the product precipitates in its free base form while keeping acidic impurities in the aqueous phase, thereby achieving the reported purity levels of approximately 99.0% without the need for complex chromatographic purification.
How to Synthesize 6-Chloropurine Efficiently
Implementing this synthesis route requires careful attention to reagent addition and temperature control to maximize yield and safety. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to large-scale industrial reactors. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational protocol for process engineers looking to adopt this technology. It is imperative to adhere to the specified molar ratios and temperature ranges to replicate the high success rates documented in the intellectual property.
- Charge acetyl hypoxanthine, phosphorus oxychloride, and a tertiary amine catalyst (such as DMA or triethylamine) into a reaction vessel.
- Heat the mixture to 70-105°C and maintain for 4-8 hours until TLC indicates complete consumption of the starting material.
- Remove excess phosphorus oxychloride by evaporation, cool with ice water, adjust pH to 7-9, and filter the precipitated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acetyl hypoxanthine-based route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of the deacetylation step fundamentally alters the cost structure of manufacturing, removing the need for additional reactors, filtration units, and drying equipment associated with the intermediate hypoxanthine isolation. This reduction in capital equipment requirements translates directly into lower fixed costs per kilogram of produced API intermediate. Furthermore, the simplified workflow reduces the total batch cycle time, allowing manufacturing facilities to increase throughput and respond more agilely to fluctuating market demands. The ability to produce high-purity material with fewer unit operations also decreases the probability of cross-contamination and human error, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the drastic simplification of the synthetic route. By avoiding the hydrolysis and subsequent acidification steps required in traditional methods, manufacturers save substantially on reagents, utilities, and labor. The direct use of acetyl hypoxanthine means that the costly and time-intensive purification of hypoxanthine is rendered obsolete. Additionally, the recovery and recycling of excess phosphorus oxychloride further contribute to raw material efficiency, ensuring that the cost of goods sold remains competitive even in volatile chemical markets. This streamlined approach allows for a leaner manufacturing operation with significantly reduced overheads.
- Enhanced Supply Chain Reliability: Sourcing acetyl hypoxanthine is generally more straightforward and stable compared to managing the logistics of multiple precursors and reagents required for the older multi-step routes. The robustness of the reaction conditions, which tolerate a range of tertiary amine catalysts, provides procurement teams with flexibility. If one catalyst becomes scarce or expensive, alternatives like pyridine or triethylamine can be substituted without revalidating the entire process. This flexibility mitigates supply risk and ensures continuity of supply for downstream customers who rely on consistent deliveries of 6-chloropurine for their own API synthesis campaigns.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the reduction of wastewater volume is a critical compliance metric. This process inherently generates less aqueous waste by eliminating the hydrolysis step, simplifying effluent treatment and reducing disposal costs. The reaction is exothermic but manageable within standard glass-lined or stainless steel reactors, making it highly scalable from pilot plant to commercial tonnage. The high yield and purity achieved reduce the burden on downstream purification processes, further minimizing solvent usage and waste generation. This alignment with green chemistry principles enhances the corporate sustainability profile of any manufacturer adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the synthesis of 6-chloropurine via this patented method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route. Understanding these details is crucial for risk assessment and process validation.
Q: What is the primary advantage of using acetyl hypoxanthine over hypoxanthine for 6-chloropurine synthesis?
A: Using acetyl hypoxanthine directly eliminates the need for a preliminary deacetylation and acidification step required when starting from hypoxanthine, significantly reducing wastewater generation and processing time.
Q: Which tertiary amines are suitable catalysts for this chlorination reaction?
A: The patent specifies several effective tertiary amines including N,N-Dimethylaniline (DMA), N,N-Diethylaniline, Triethylamine, and Pyridine, allowing flexibility based on availability and cost.
Q: What purity levels can be achieved with this synthetic method?
A: Experimental data from the patent indicates that this method consistently yields 6-chloropurine with a purity of approximately 99.0% as determined by HPLC analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Chloropurine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. Our technical team has extensively analyzed the potential of the acetyl hypoxanthine chlorination pathway, confirming its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 6-chloropurine meets the exacting standards required for antiviral and oncology drug manufacturing.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your project's economics and timeline. Let us be your strategic partner in delivering high-performance chemical solutions.
