Advanced Green Synthesis of Ceftriaxone Sodium: Replacing Toxic Solvents for Scalable Production
Advanced Green Synthesis of Ceftriaxone Sodium: Replacing Toxic Solvents for Scalable Production
The pharmaceutical industry is currently undergoing a significant paradigm shift towards greener, more sustainable manufacturing processes, particularly for high-volume antibiotics like Ceftriaxone Sodium. Patent CN103539803A introduces a groundbreaking methodology that fundamentally alters the solvent landscape for synthesizing this critical third-generation cephalosporin. Traditionally, the production of the key intermediate 7-ACT and the final Active Pharmaceutical Ingredient (API) has relied heavily on hazardous organic solvents such as acetonitrile and dichloromethane. This new technical disclosure presents a robust alternative by substituting these toxic media with Dimethyl Carbonate (DMC) and a binary Ethanol-Water system. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediates supplier, this innovation represents a dual opportunity: enhancing worker safety through the elimination of carcinogenic solvents while simultaneously optimizing the cost structure of cost reduction in antibiotic manufacturing. The process demonstrates exceptional reproducibility, achieving molar yields of approximately 90% for the intermediate and up to 95% for the final API, all while maintaining HPLC purities exceeding 99%.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 7-Amino-3-[(2,5-dihydro-6-hydroxy-2-methyl-5-oxo-1,2,4-triazin-3-yl)thiomethyl]cephalosporanic acid (7-ACT), the pivotal precursor to Ceftriaxone Sodium, has been plagued by significant safety and environmental liabilities. Conventional protocols, as cited in various prior art patents, predominantly utilize acetonitrile as the reaction medium in the presence of boron trifluoride catalysts. Acetonitrile is classified as a Class 2 solvent due to its inherent toxicity, potential animal carcinogenicity, and ability to cause severe physiological disturbances including respiratory and circulatory disorders upon chronic exposure. Furthermore, the recovery of acetonitrile is energetically intensive and technically challenging due to its miscibility with water, often leading to substantial solvent loss and increased wastewater treatment burdens. In the final condensation step to form Ceftriaxone Sodium, traditional methods frequently employ dichloromethane or complex ternary solvent systems. These halogenated solvents pose severe occupational health risks and complicate the regulatory compliance landscape for any commercial scale-up of complex pharmaceutical intermediates. The reliance on such hazardous materials not only inflates operational costs through strict containment requirements but also introduces supply chain vulnerabilities associated with the tightening global regulations on volatile organic compounds (VOCs).
The Novel Approach
The methodology disclosed in Patent CN103539803A offers a transformative solution by replacing these problematic solvents with environmentally benign alternatives without compromising reaction efficiency. In the first stage, Dimethyl Carbonate (DMC) serves as the primary reaction solvent for the coupling of 7-ACA and triazine acid (TTZ). DMC is recognized globally as a green solvent, characterized by its non-toxic nature, high flash point, and low vapor pressure, effectively mitigating the explosion risks and health hazards associated with acetonitrile. As illustrated in the reaction scheme below, this substitution maintains the efficacy of the boron trifluoride catalyst while facilitating easier downstream processing.
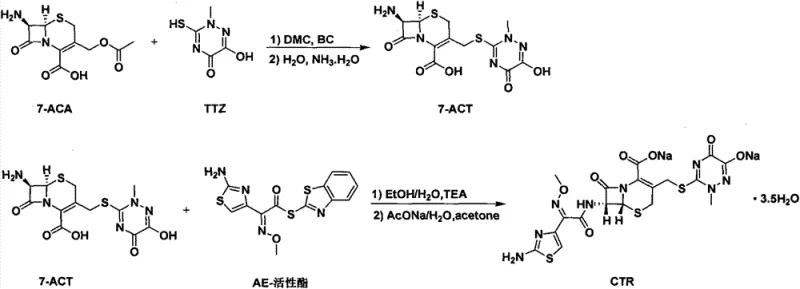
Furthermore, the final condensation step utilizes a binary system of ethanol and water, completely eliminating the need for chlorinated solvents like dichloromethane. This ethanol-water mixture not only provides an excellent medium for the reaction between 7-ACT and the AE-active ester but also simplifies the crystallization process. The result is a streamlined workflow that significantly reduces the environmental footprint of the manufacturing process. By adopting this novel approach, manufacturers can achieve high-purity pharmaceutical intermediates with color grades meeting the stringent ≤YG3 standard, ensuring that the final product is suitable for sensitive clinical applications such as the treatment of meningitis and severe respiratory infections.
Mechanistic Insights into DMC-Mediated Electrophilic Substitution and Binary Solvent Crystallization
The core chemical innovation lies in the utilization of Dimethyl Carbonate as a polar aprotic solvent that effectively solubilizes both the beta-lactam substrate (7-ACA) and the triazine acid (TTZ) while stabilizing the boron trifluoride catalyst complex. Mechanistically, the reaction proceeds via an electrophilic substitution where the activated triazine moiety attacks the C-3 position of the cephalosporin nucleus. The patent data indicates that the mass-to-volume ratio of 7-ACA to DMC is a Critical Process Parameter (CPP), with an optimal range of 1:5 to 1:8. Specifically, a ratio of 1:5.71 was found to balance reaction kinetics and solvent economy, ensuring complete conversion within 50 minutes at 30°C. Deviating from this ratio leads to either prolonged reaction times, which increase energy consumption, or poor process control, which compromises the purity of the resulting 7-ACT. The structure of the intermediate 7-ACT, shown below, highlights the sensitive beta-lactam ring which requires mild conditions to prevent degradation.
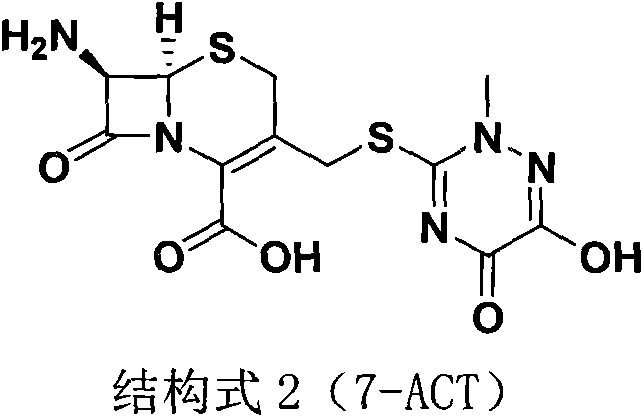
In the subsequent step, the transition to an ethanol-water binary system for the condensation with AE-active ester is equally critical. The polarity of this mixture facilitates the nucleophilic attack of the 7-amino group on the active ester carbonyl. The patent specifies an optimal ethanol-to-water volume ratio of 15:1. This specific ratio is crucial; excessive water slows the reaction kinetics and degrades product quality, while insufficient water hinders the complete dissolution of reagents and subsequent salt formation. The reaction is initiated at low temperatures (1-3°C) to control exothermicity and minimize epimerization, followed by a controlled warm-up to 20°C for crystallization. This precise thermal management, combined with the green solvent system, ensures the formation of the stable disodium salt structure of Ceftriaxone Sodium, depicted below, with minimal impurity profiles.
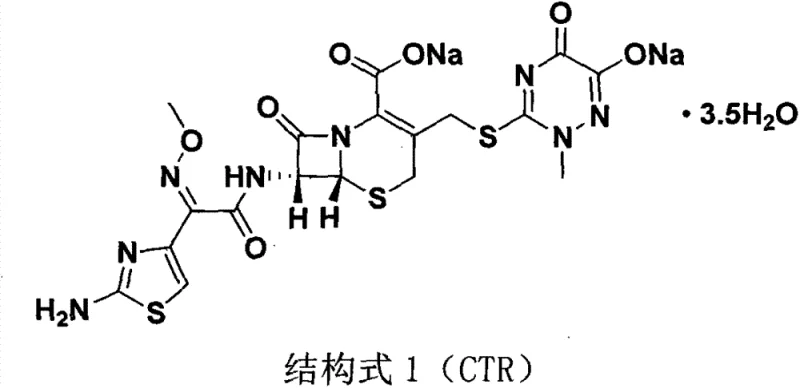
How to Synthesize Ceftriaxone Sodium Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to industrial production. The process is divided into two distinct stages: the preparation of the 7-ACT intermediate and the final condensation to form the API. Each stage requires precise control over stoichiometry, temperature, and pH to ensure consistent quality. The following guide summarizes the critical operational parameters derived from the patent embodiments, serving as a foundation for process engineers looking to implement this green technology. For detailed standard operating procedures and safety guidelines, please refer to the technical documentation provided below.
- React 7-ACA and Triazine Acid (TTZ) in Dimethyl Carbonate (DMC) with Boron Trifluoride-DMC catalyst at 30°C for 50 minutes to form 7-ACT.
- Terminate reaction with sodium bisulfite, adjust pH to 3.8-4.0 with ammonia water, and crystallize at 6-7°C to isolate high-purity 7-ACT.
- Condense 7-ACT with AE-active ester in an Ethanol-Water binary system (15: 1 ratio) at low temperature, followed by sodium salt formation and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this green synthesis route offers compelling strategic advantages beyond mere regulatory compliance. The shift away from high-cost, high-toxicity solvents like acetonitrile and dichloromethane directly impacts the bottom line by reducing the costs associated with hazardous waste disposal and solvent recovery. Dimethyl carbonate and ethanol are commodity chemicals with stable supply chains and significantly lower price volatility compared to specialized halogenated solvents. This stability contributes to substantial cost savings in raw material procurement and inventory management.
- Cost Reduction in Manufacturing: The elimination of toxic solvents removes the need for expensive containment infrastructure and complex abatement systems required for VOC emissions. Additionally, the ease of recovering Dimethyl Carbonate and Ethanol through standard distillation techniques reduces solvent loss, leading to a more efficient material balance. The process operates under mild conditions (30°C for the first step), which lowers energy consumption for heating and cooling compared to more aggressive traditional methods. These factors collectively drive down the Cost of Goods Sold (COGS) without sacrificing yield or purity.
- Enhanced Supply Chain Reliability: Relying on green solvents like DMC and Ethanol mitigates the risk of supply disruptions caused by environmental crackdowns on toxic chemical production. Since these solvents are widely produced for various industries, sourcing is more resilient. Furthermore, the improved safety profile of the manufacturing process reduces the likelihood of unplanned shutdowns due to safety incidents or regulatory inspections, ensuring a continuous and reliable flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been optimized for mass production with specific attention to solvent ratios that prevent bottlenecks in filtration and drying. The use of non-halogenated solvents simplifies the handling of mother liquors and washings, making wastewater treatment more straightforward and less costly. This alignment with green chemistry principles future-proofs the manufacturing site against increasingly stringent environmental regulations, securing long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis method. These answers are derived directly from the experimental data and comparative analysis presented in Patent CN103539803A, providing clarity on yield expectations, solvent performance, and quality metrics. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is Dimethyl Carbonate preferred over Acetonitrile for 7-ACT synthesis?
A: Dimethyl Carbonate (DMC) is classified as a non-toxic green solvent with a high flash point and low vapor pressure, whereas Acetonitrile is a Class 2 toxic solvent with carcinogenic potential. DMC also offers easier recovery and lower environmental impact.
Q: What is the optimal solvent ratio for the final Ceftriaxone Sodium condensation step?
A: The patent specifies an optimal volume ratio of Anhydrous Ethanol to Water of 15:1. This ratio balances reaction kinetics and product quality, achieving yields up to 95% while maintaining high purity.
Q: How does this process impact the color grade of the final API?
A: By utilizing green solvents like DMC and Ethanol-Water systems, the process minimizes side reactions and impurity formation, consistently achieving a color grade of ≤YG3, which meets stringent pharmacopoeia standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftriaxone Sodium Supplier
The technological advancements detailed in Patent CN103539803A underscore the immense potential for optimizing the production of critical antibiotics through green chemistry. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of such innovations, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Ceftriaxone Sodium or 7-ACT we produce adheres to the highest international standards. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our commitment to green manufacturing aligns perfectly with the evolving needs of global healthcare providers.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific supply chain requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this greener process. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to deliver high-quality, cost-effective solutions for the global market.
