Advanced Chiral Resolution Technology for High-Purity Linezolid Intermediates and Commercial Scalability
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for the production of critical antibiotic intermediates, particularly for oxazolidinone class drugs like Linezolid. Patent CN102321040A introduces a transformative approach to synthesizing Linezolid and its key chiral intermediate by shifting away from expensive chiral starting materials towards a resolution strategy using racemic precursors. This technology leverages the formation of diastereomeric salts with common chiral acids, such as L-tartrate or D-malic acid, to effectively separate enantiomers. By utilizing this resolution technique, manufacturers can bypass the stringent and costly requirements associated with sourcing enantiomerically pure building blocks, thereby streamlining the supply chain for this essential anti-infective agent.
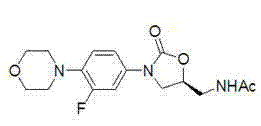
The significance of this patent lies in its ability to produce the biologically active S-isomer with high optical purity through a sequence of salt formation, crystallization, and liberation. The process is designed to be operationally simple, avoiding complex asymmetric catalysis or enzymatic steps that often require specialized equipment or conditions. Instead, it relies on fundamental principles of stereochemistry and solubility differences, making it highly adaptable for large-scale industrial production. This method not only addresses the economic constraints of traditional synthesis but also offers a reliable pathway to minimize impurities related to the wrong enantiomer, ensuring the final API meets rigorous pharmacopeial standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Linezolid has heavily relied on the "chiral pool" strategy, where the chirality is introduced at the very beginning of the synthesis using expensive, enantiomerically pure raw materials. As illustrated in prior art references such as WO2002085849, conventional routes often utilize precursors like S-1-acetamido-3-chloropropane-2-acetate to construct the oxazolidinone ring.
These traditional methods present significant logistical and financial challenges for procurement teams. The starting materials are specialized fine chemicals that command premium prices due to the complexity of their own synthesis. Furthermore, maintaining the stereochemical integrity throughout the multi-step synthesis is critical; any epimerization or racemization during the ring-closing or substitution steps can lead to a decrease in the enantiomeric excess (ee) of the final product. This necessitates tight process controls and often results in lower overall yields, as batches failing to meet strict chiral specifications must be discarded or reworked, driving up the cost of goods sold (COGS).
The Novel Approach
In stark contrast, the methodology disclosed in CN102321040A adopts a "late-stage resolution" or "intermediate resolution" strategy that fundamentally alters the economic model of production. Instead of starting with a chiral reagent, the process begins with a racemic mixture of 5-aminomethyl-3-(3-fluoro-4-morpholinyl phenyl)-oxazolidin-2-one, which is significantly cheaper and easier to source in bulk quantities. The chirality is induced through a classical resolution technique involving the reaction of this racemic amine with a chiral acid.
This approach decouples the cost of the starting material from the cost of chirality. By forming diastereomeric salts, the process exploits the distinct physical properties—specifically solubility—of the resulting salt pairs. One diastereomer crystallizes out of the solution while the other remains in the mother liquor, allowing for physical separation via filtration. This method is inherently more robust because the crystallization step acts as a purification event, simultaneously enriching the optical purity and removing chemical impurities. The result is a streamlined process that transforms a commodity chemical into a high-value chiral intermediate with minimal waste and maximum efficiency.
Mechanistic Insights into Diastereomeric Salt Resolution
The core mechanism driving this technology is the acid-base reaction between the basic nitrogen of the racemic oxazolidinone intermediate and the carboxylic acid group of the chiral resolving agent. When the racemic amine reacts with a single enantiomer of a chiral acid, such as L-tartaric acid, two distinct diastereomeric salts are formed: one comprising the (S)-amine and the (L)-acid, and the other comprising the (R)-amine and the (L)-acid. Unlike enantiomers, which have identical physical properties in an achiral environment, diastereomers possess different melting points, solubilities, and crystallization behaviors.
The patent details a sophisticated optimization of solvent systems to maximize the solubility gap between these two salts. By carefully selecting solvents like ethyl acetate, ether, or dichloromethane, and manipulating parameters such as temperature and concentration, the process ensures that the less soluble diastereomeric salt precipitates selectively. For instance, cooling the reaction mixture to temperatures as low as -20°C or heating to 35°C can dramatically shift the equilibrium and solubility profiles, favoring the crystallization of the desired isomer. This thermodynamic control is crucial for achieving high enantiomeric excess, as demonstrated in the examples where ee values reached up to 99.9% after recrystallization.
Following the isolation of the pure diastereomeric salt, the chiral intermediate is liberated by treating the salt with an aqueous base, such as sodium carbonate or sodium hydroxide. This neutralization step converts the ammonium salt back into the free amine, which is then extracted into an organic phase. The final step involves acetylation of the primary amine to yield Linezolid. This mechanistic pathway is advantageous because the chiral acid can theoretically be recovered from the aqueous layer after basification, allowing for recycling and further reducing the environmental footprint and material costs of the overall process.
How to Synthesize Linezolid Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from racemic mixtures to optically pure intermediates suitable for API production. The process is characterized by its flexibility in solvent choice and resolving agents, allowing manufacturers to tailor the conditions to their specific facility capabilities. Detailed standard operating procedures regarding molar ratios, solvent volumes, and temperature ramps are essential for reproducibility.
- Dissolve racemic 5-aminomethyl-3-(3-fluoro-4-morpholinyl phenyl)-oxazolidin-2-one and a chiral acid (e.g., L-tartrate) in an organic solvent like ethyl acetate or dichloromethane to form diastereomeric salts.
- Induce crystallization by adjusting temperature (e.g., cooling to -20°C or heating to 35°C) to precipitate the desired single diastereomeric salt, then filter the solid.
- Dissolve the filtered salt in water, adjust pH to alkaline using sodium carbonate to free the base, extract with dichloromethane, and finally acetylate to obtain Linezolid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution-based technology offers profound strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic reduction in raw material costs. By switching from specialized chiral building blocks to bulk racemic amines and commodity chiral acids, companies can insulate themselves from the price volatility often seen in the market for fine chiral reagents. This shift transforms a high-cost input into a low-cost input, directly improving the gross margin of the final API.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials creates a substantial opportunity for cost optimization. Traditional routes require purchasing precursors that have already undergone resolution or asymmetric synthesis, meaning the manufacturer pays a premium for chirality before the process even begins. In this new method, the cost of chirality is limited to the stoichiometric amount of cheap resolving agents like tartaric acid, which are produced on a massive industrial scale for the food and pharma industries. Additionally, the potential to recover and recycle the resolving agent further amplifies these savings, creating a circular economy within the manufacturing process that minimizes waste disposal costs.
- Enhanced Supply Chain Reliability: Relying on racemic starting materials significantly de-risks the supply chain. Racemic amines are simpler to synthesize and are available from a broader range of global suppliers compared to their enantiomerically pure counterparts. This diversification of the supplier base reduces the risk of shortages and provides greater negotiating leverage. Furthermore, the robustness of the crystallization step ensures consistent quality output even if there are minor fluctuations in the quality of the incoming racemic feedstock, as the crystallization acts as a powerful purification barrier.
- Scalability and Environmental Compliance: The process is inherently scalable because it relies on unit operations that are standard in any chemical plant, such as crystallization, filtration, and extraction. There is no need for specialized reactors for asymmetric catalysis or sensitive handling of air-sensitive reagents. From an environmental perspective, the use of benign chiral acids and the avoidance of heavy metal catalysts align with green chemistry principles. The ability to recycle solvents and resolving agents reduces the volume of hazardous waste generated, simplifying compliance with increasingly stringent environmental regulations and lowering the total cost of ownership for the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of using chiral acid resolution over chiral pool synthesis for Linezolid?
A: The resolution method utilizes inexpensive, readily available racemic starting materials and bulk chiral acids like tartaric acid, significantly reducing raw material costs compared to purchasing high-value enantiomerically pure precursors.
Q: How is high optical purity achieved in this process?
A: High optical purity is achieved through the selective crystallization of diastereomeric salts, where solubility differences are maximized by optimizing solvent systems (e.g., ethyl acetate/ether) and temperature controls, potentially reaching >99% ee.
Q: Can the chiral resolving agents be recovered?
A: Yes, the chiral acids used in the salt formation can be recovered after hydrolysis and recycling, further enhancing the economic efficiency and environmental profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linezolid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the methodology described in CN102321040A and possesses the technical capability to implement this resolution strategy at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and compliant with cGMP standards. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Linezolid intermediate we produce meets the highest global regulatory requirements.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective technology for their API supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this resolution route for your specific project. We encourage you to contact us today to discuss your requirements,索取 specific COA data, and review our comprehensive route feasibility assessments to secure a reliable and competitive supply of high-quality Linezolid intermediates.
